Rationale and Objectives
Near-infrared spectroscopy (NIRS) of breast can provide functional information on the vascular and structural compartments of tissues in regions identified during simultaneous magnetic resonance imaging (MRI). NIRS can be acquired during dynamic contrast-enhanced MRI (DCE-MRI) to accomplish image-guided spectroscopy of the enhancing regions, potentially increasing the diagnostic specificity of the examination and reducing the number of biopsies performed as a result of inconclusive MRI breast imaging studies.
Materials and Methods
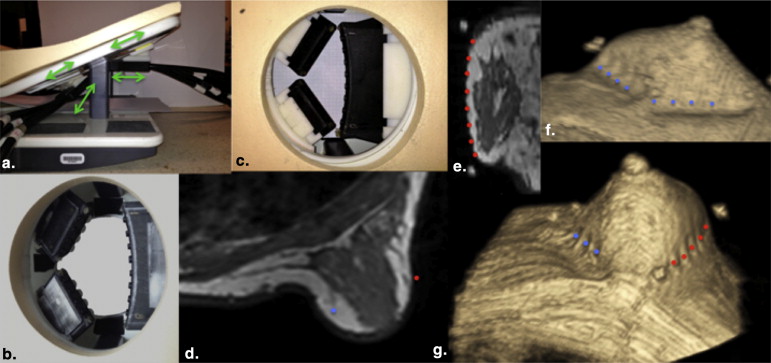
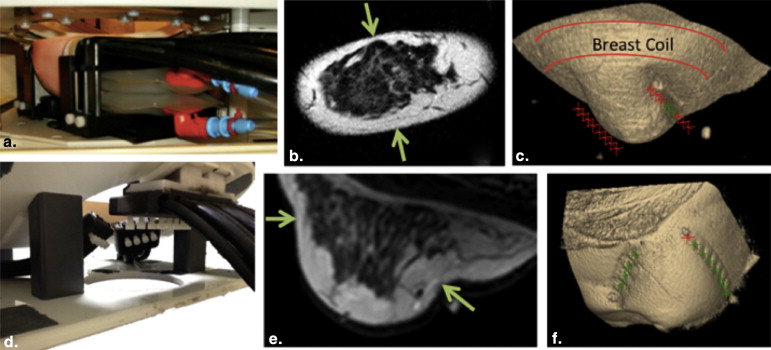
We combine synergistic attributes of concurrent DCE-MRI and NIRS with a new design of the clinical NIRS breast interface that couples to a standard MR breast coil and allows imaging of variable breast sizes. Spectral information from healthy volunteers and cancer patients is recovered, providing molecular information in regions defined by the segmented MR image volume.
Results
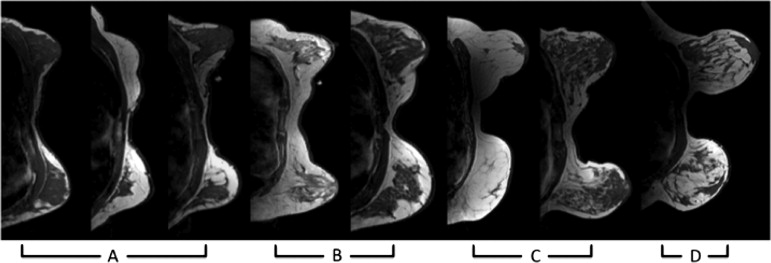
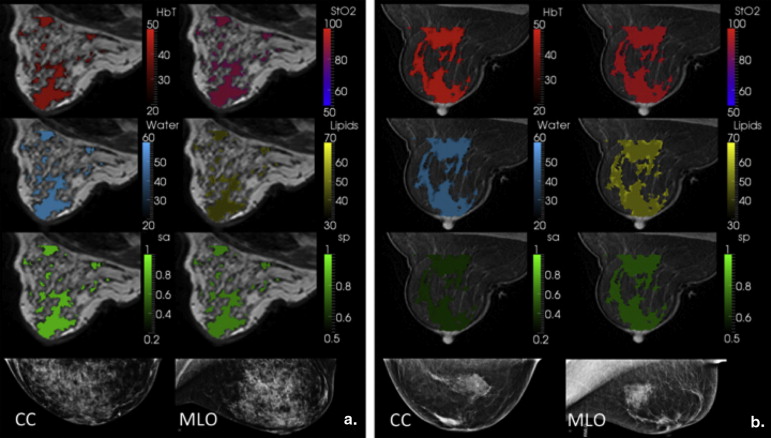
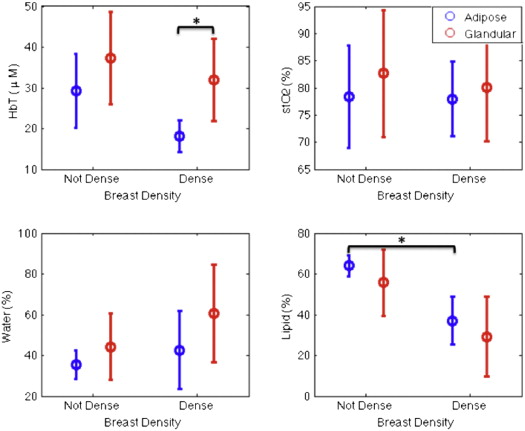
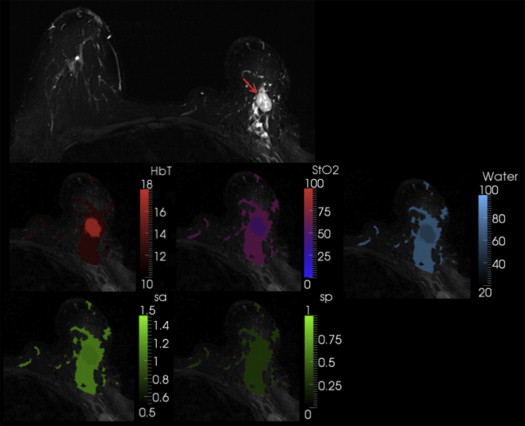
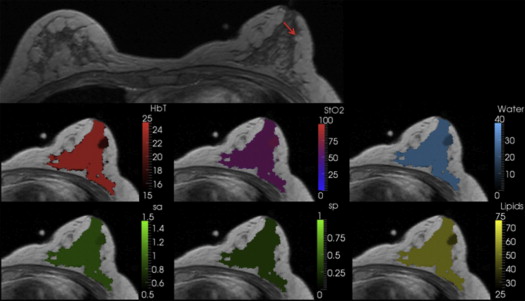
The new coupling system significantly improves examination utility by allowing improved coupling of the NIR fibers to breasts of all cup sizes and lesion locations. This improvement is demonstrated over a range of breast sizes (cup size A through D) and normal tissue heterogeneity using a group of eight healthy volunteers and two cancer patients. Lesions located in the axillary region and medial-posterior breast are now accessible to NIRS optodes. Reconstructed images were found to have biologically plausible hemoglobin content, oxygen saturation, and water and lipid fractions.
Conclusions
In summary, a new NIRS/MRI breast interface was developed to accommodate the variation in breast sizes and lesion locations that can be expected in clinical practice. DCE-MRI–guided NIRS quantifies total hemoglobin, oxygenation, and scattering in MR-enhancing regions, increasing the diagnostic information acquired from MR examinations.
Breast cancer is a complex biological disease that presents challenges for detection and diagnosis. Mammography, the current gold standard for breast screening, has an overall sensitivity and specificity reported to be 77% and 97%, respectively, in a randomized multicenter trial . However, the technique is much less effective in women with mammographically dense breasts, in which the sensitivity and specificity fall considerably to 63% and 89%, respectively . Women with more dense breasts have both higher incidence of and mortality from breast cancer. They are also the most difficult group to screen with mammography . Therefore, current clinical care includes breast dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) for screening of high-risk patients . DCE-MRI is recommended for screening this population in combination with mammography because it has greater sensitivity than standard mammography, reported to be 93%–100% . Screening specificity with DCE-MRI is less consistent. It generally causes 3- to 5-fold more false-positive findings than mammography, leading to more unnecessary biopsies and invasive procedures that are stressful for patients. In this study, the coupling of near-infrared spectroscopy (NIRS) with breast MRI is examined with a new interface that allows substantially improved flexibility in delivering the combined examination to the breast than previous versions.
Combined NIRS and MRI is an emerging imaging approach that could benefit patients after screening by increasing the specificity of DCE-MRI before biopsy . The technique can be used to noninvasively quantify oxy- and deoxy-hemoglobin, water and lipid content, and scattering parameters in adipose and fibroglandular tissues. Combined NIRS and MRI systems have been developed in the United States and Germany for human imaging , but they have been slow to progress because of design challenges and cost. However, incentives exist to develop MRI/NIRS based on promising results from stand-alone NIRS. Hundreds of patients have undergone stand-alone NIRS breast imaging at multiple academic centers in the United States and Europe . In addition, several commercial systems have been developed . The sensitivity and specificity of breast cancer detection with NIRS alone varies depending on the system geometry but has been reported to be in the range of 91%–96% and 93%–95%, respectively . Because MRI information can be used to guide image reconstruction in MRI/NIRS, the technique may improve on the stand-alone NIRS results, especially when lesions are smaller than 1–2 cm. The spatial correlation between the data streams of MRI and NIRS has the potential to provide complementary information .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Human Subject Imaging
Get Radiology Tree app to read full this article<
Instrumentation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Optical Image Reconstruction
Get Radiology Tree app to read full this article<
−∇⋅D∇Φ(r,ω)+(μa+iωc)Φ(r,ω)=S(r,ω), −
∇
·
D
∇
Φ
(
r
,
ω
)
+
(
μ
a
+
i
ω
c
)
Φ
(
r
,
ω
)
=
S
(
r
,
ω
)
,
discretized on finite elements. Here, a source, S , with frequency, ω , describes light fluence, Φ, through the turbid media. We also use a modified Tikhonov regularization routine with regularization parameter, λ , using a Levenberg–Marquardt iterative update, which stabilizes the estimation process by reducing the effects of noise on the image reconstruction, and eliminating improbable solutions . The image formation algorithm (Eq. 2 ) is nonlinear and solved with a Newton-type minimization method for Δ c within the matrix equation :
(JTJ+λI)Δc=JTΔδ, (
J
T
J
+
λ
I
)
Δ
c
=
J
T
Δ
δ
,
that optimizes the estimation of the physiological parameters, c , which include oxygenated and deoxygenated hemoglobin concentrations, water fraction, scatter amplitude, and scatter power . We typically report total hemoglobin, HbT = HbO + Hb, and oxygen saturation, StO2=HbOHbT StO
2
=
HbO
HbT , from these parameters. Here, J is the Jacobian matrix, I is the identity matrix, and δ is the model-data misfit. Selection of λ influences the resulting solutions , and it is chosen based on inherent system noise and fiber coupling errors, which are difficult to quantify in general because they can be case specific .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Triangular Interface Performance
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Human Subject Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Recovered Values from Optical Imaging of Abnormal Breast Tissue in Two Patients
Tissue Total Hemoglobin (μM) Oxygen Saturation (%) Water Content (%) Lipid Content (%) Scatter Amplitude Scatter Power Patient 1 Adipose 8.7 46.1 33.4 n/a 0.76 0.3 Glandular 12.6 60.2 44.5 n/a 1.2 0.3 Tumor 17.9 44.5 61.4 n/a 1.1 0.3 Tumor/Glandular1.4× 0.7× 0.8× n/a 0.9× 1.0× Patient 2 Adipose 12.0 64.6 27.7 72.3 1.2 0.1 Glandular 17.8 54.1 68.6 31.4 1.1 0.1 Tumor 12.7 68.4 31.2 68.8 0.85 0.1 Tumor/Glandular0.7× 1.26× 0.5× 2.2× 0.8× 1.0×
Patient 1 was a malignant case, and the region of interest (ROI)–total hemoglobin (HbT) was 1.4 times the glandular HbT. Patient 2 was a benign case with ROI-HbT of 0.7× the glandular HbT (contrasts in bold type).
Get Radiology Tree app to read full this article<
Discussion
Triangular Interface Development
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Human Image Interpretation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Skaane P., Hofvind S., Skjennald A.: Randomized trial of screen-film versus full-field digital mammography with soft-copy reading in population-based screening program: follow-up and final results of Oslo II study. Radiology 2007; 244: pp. 708-717.
2. Carney P.A., Miglioretti D.L., Yankaskes B.C., et. al.: Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med 2003; 138: pp. 168-175.
3. Lam P.B., Vacek P.M., Geller B.M., et. al.: The association of increased weight, body mass index, and tissue density with the risk of breast carcinoma in Vermont. Cancer 2000; 89: pp. 369-375.
4. Boyd N.F., Lockwood G.A., Byng J.W., et. al.: Mammographic densities and breast cancer risk. Cancer Epidemiol Biomarkers Prev 1998; 7: pp. 1133-1144.
5. Chiu S.Y.-H., Duffy S., Yen A.M., et. al.: Effect of baseline breast density on breast cancer incidence, stage, mortality, and screening parameters: 25-year follow-up of a Swedish mammographic screening. Cancer Epidemiol Biomarkers Prev 2010; 19: pp. 1219-1228.
6. Kuhl C.K.: Current status of breast MR imaging. Part 2. Clinical applications. Radiology 2007; 244: pp. 672-691.
7. Lee J.M., Halpern E.F., Rafferty E.A., et. al.: Evaluating the correlation between film mammography and MRI for screening women with increased breast cancer risk. Acad Radiol 2009; 16: pp. 1323-1328.
8. Lord S.J., Lei W., Craft P., et. al.: A systematic review of the effectiveness of magnetic resonance imaging (MRI) as an addition to mammography and ultrasound in screening young women at high risk of breast cancer. Eur J Cancer 2007; 43: pp. 1905-1917.
9. Warner E., Messersmith H., Causer P., et. al.: Systematic review: using magnetic resonance imaging to screen women at high risk for breast cancer. Ann Intern Med 2008; 148: pp. 671-679.
10. Carpenter C.M., Pogue B.W., Jiang S., et. al.: Image-guided spectroscopy provides molecular specific information in vivo: MRI-guided spectroscopy of breast cancer hemoglobin, water, and scatterer size. Opt Lett 2007; 32: pp. 933-935.
11. Tromberg B.J., Pogue B.W., Paulsen K.D., et. al.: Assessing the future of diffuse optical imaging technologies for breast cancer management. Med Phys 2008; 35: pp. 2443-2451.
12. Ntziachristos V., Yodh A.G., Schnall M., et. al.: Concurrent MRI and diffuse optical tomography of breast after indocyanine green enhancement. Proc Natl Acad Sci U S A 2000; 97: pp. 2767-2772.
13. Brooksby B., Pogue B.W., Jiang S., et. al.: Imaging breast adipose and fibroglandular tissue molecular signatures using hybrid MRI-guided near-infrared spectral tomography. Proc Nat Acad Sci U S A 2006; 103: pp. 8828-8833.
14. Poellinger A., Burock S., Grosenick D., et. al.: Breast cancer: early- and late-fluorescence near-infrared imaging with indocyanine green—a preliminary study. Radiology 2011; 258: pp. 409-416.
15. Rinneberg H., Grosenick D., Moesta T.K., et. al.: Scanning time-domain optical mammography: detection and characterization of breast tumors in vivo. Technol Cancer Res Treat 2005; 4: pp. 483-496.
16. Intes X.: Time-domain optical mammography SoftScan initial results. Acad Radiol 2005; 12: pp. 934-947.
17. Poellinger A., Martin J.C., Ponder S.L., et. al.: Near-infrared laser computed tomography of the breast first clinical experience. Acad Radiol 2008; 15: pp. 1545-1553.
18. Chance B., Nioka S., Zhang J., et. al.: Breast cancer detection based on incremental biochemical and physiological properties of breast cancers: a six-year, two-site study. Acad Radiol 2005; 12: pp. 925-933.
19. Kukreti S., Cerussi A.E., Tanamai W., et. al.: Characterization of metabolic differences between benign and malignant tumors: high-spectral-resolution diffuse optical spectroscopy. Radiology 2010; 254: pp. 277-284.
20. Srinivasan S., Pogue B.W., Jiang S., et. al.: Interpreting hemoglobin and water concentration, oxygen saturation, and scattering measured by near-infrared tomography of normal breast in vivo. Proc Nat Acad Sci U S A 2003; 100: pp. 12349-12354.
21. Choe R., Konecky S.D., Corlu A., et. al.: Differentiation of benign and malignant breast lesions by in-vivo three-dimensional diffuse optical tomography. Cancer Res 2009; 69: 102S-102S
22. Carpenter C.M., Srinivasan S., Pogue B.W., et. al.: Methodology development for three-dimensional MR-guided near infrared spectroscopy of breast tumors. Opt Express 2008; 16: pp. 17903-17914.
23. Mastanduno M.A., Jiang S., DiFlorio-Alexander R., et. al.: Remote positioning optical breast magnetic resonance coil for slice-selection during image-guided near-infrared spectroscopy of breast cancer. J Biomed Opt 2011; 16: pp. 066001.
24. Brooksby B., Jiang S., Dehghani H., et. al.: Magnetic resonance-guided near-infrared tomography of the breast. Rev Sci Instrum 2004; 75: pp. 5262-5270.
25. Dehghani H., Eames M.E., Yalavarthy P.K., et. al.: Near infrared optical tomography using NIRFAST: Algorithm for numerical model and image reconstruction. Commun Numer Methods Eng 2008; 25: pp. 711-732.
26. Dehghani H., Pogue B.W., Shudong J., et. al.: Three-dimensional optical tomography: resolution in small-object imaging. Appl Opt 2003; 42: pp. 3117-3128.
27. McBride T.O., Pogue B.W., Gerety E., et. al.: Spectroscopic diffuse optical tomography for the quantitative assessment of hemoglobin concentration and oxygen saturation in breast tissue.. Appl Opt 1999; 38: pp. 5480-5490.
28. Srinivasan S., Pogue B.W., Jiang S., et. al.: In vivo hemoglobin and water concentrations, oxygen saturation, and scattering estimates from near-infrared breast tomography using spectral reconstruction. Acad Radiol 2006; 13: pp. 195-202.
29. Arridge S.R.: Photon-measurement density-functions. Part I: Analytical forms. Appl Opt 1995; 34: pp. 7395-7409.
30. Arridge S.R., Schweiger M.: Photon-measurement density functions. Part 2: Finite-element-method calculations. Appl Opt 1995; 34: pp. 8026-8037.
31. McBride T.O., Pogue B.W., Jiang S., et. al.: Development and calibration of a parallel modulated near-infrared tomography system for hemoglobin imaging in vivo. Rev Sci Instrum 2001; 72: pp. 1817-1824.
32. Arridge S.R., Schweiger M., Delpy D.T.: Iterative reconstruction of near infrared absorption images. Proc SPIE 1992; 1767: pp. 372-383.
33. Yalavarthy P. A generalized least-squares estimation minimization method for near infrared diffuse optical tomography, (2007).
34. Schweiger M., Nissilä I., Boas D.A., et. al.: Image reconstruction in optical tomography in the presence of coupling errors. Appl Opt 2007; 46: pp. 2743-2756.
35. Brooksby B., Jiang S., Dehghani H., et. al.: Combining near-infrared tomography and magnetic resonance imaging to study in vivo breast tissue: implementation of a Laplacian-type regularization to incorporate magnetic resonance structure. J Biomed Opt 2005; 10: pp. 051504.
36. El-Ghussein F., Mastanduno M.A., Jiang S., et. al.: Hybrid PMT and photodiode parallel detection array for wideband optical spectroscopy of the breast guided by MRI. Prep 2013;
37. Mastanduno, MA, Jiang, S, diFlorio-Alexander, R, et al. Nine-wavelength spectroscopy guided by magnetic resonance imaging improves breast cancer characterization. in BW3A.3 (Optical Society of America, 2012). Available at: http://www.opticsinfobase.org/abstract.cfm?URI=BIOMED-2012-BW3A.3
38. Girardi V., Carbognin G., Camera L., et. al.: Multifocal, multicentric and contralateral breast cancers: breast MR imaging in the preoperative evaluation of patients with newly diagnosed breast cancer. Radiol Med (Torino) 2011; 116: pp. 1226-1238.
39. Boyd N.F., Rommens J.M., Vogt K., et. al.: Mammographic breast density as an intermediate phenotype for breast cancer. Lancet Oncol 2005; 6: pp. 798-808.
40. Cutress R.I., Simoes T., Gill J., et. al.: Modification of the Wise pattern breast reduction for oncological mammaplasty of upper outer and upper inner quadrant breast tumours: a technical note and case series. J Plast Reconstr Aesthet Surg 2013; 66: pp. e31-e36.
41. Jiang S., Pogue B.W., Carpenter C.M., et. al.: Evaluation of breast tumor response to neoadjuvant chemotherapy with tomographic diffuse optical spectroscopy: case studies of tumor region-of-interest changes. Radiology 2009; 252: pp. 551-560.
42. Brooksby B, Jiang S, Dehghani H, et al. “Quantifying adipose and fibroglandular breast tissue properties using MRI-guided NIR tomography”, Proc. SPIE 2005; 5693:255.
43. Poplack S.P., Paulsen K.D., Hartov A., et. al.: Electromagnetic breast imaging: results of a pilot study in women with abnormal mammograms. Radiology 2007; 243: pp. 350-359.
44. Cerussi A., Shah N., Hsiang D., et. al.: In vivo absorption, scattering, and physiologic properties of 58 malignant breast tumors determined by broadband diffuse optical spectroscopy. J. Biomed Opt 2006; 11:
45. Choe R., Konecky S.D., Corlu A., et. al.: Differentiation of benign and malignant breast tumors by in-vivo three-dimensional parallel-plate diffuse optical tomography. J Biomed Opt 2009; 14: pp. 024020.
46. Cerussi A.E., Tanamai V.W., Hsiang D., et. al.: Diffuse optical spectroscopic imaging correlates with final pathological response in breast cancer neoadjuvant chemotherapy. Philos Trans A Math Phys Eng Sci 2011; 369: pp. 4512-4530.
47. Mastanduno M.A., Jiang S., Diflorio-Alexander R., et. al.: Automatic and robust calibration of optical detector arrays for biomedical diffuse optical spectroscopy. Biomed Opt Express 2012; 3: pp. 2339-2352.