The demand for functional imaging in clinical medicine is comprehensive. Although the gold standard for the functional imaging of human bones in clinical settings is still radionuclide-based imaging modalities, nonionizing noninvasive imaging technology in small animals has greatly advanced in recent decades, especially the diffuse optical imaging to which Britton Chance made tremendous contributions. The evolution of imaging probes, instruments, and computation has facilitated exploration in the complicated biomedical research field by allowing longitudinal observation of molecular events in live cells and animals. These research-imaging tools are being used for clinical applications in various specialties, such as oncology, neuroscience, and dermatology. The Bone, a deeply located mineralized tissue, presents a challenge for noninvasive functional imaging in humans. Using nanoparticles (NP) with multiple favorable properties as bioimaging probes has provided orthopedics an opportunity to benefit from these noninvasive bone-imaging techniques. This review highlights the historical evolution of radionuclide-based imaging, computed tomography, positron emission tomography, and magnetic resonance imaging, diffuse optics–enabled in vivo technologies, vibrational spectroscopic imaging, and a greater potential for using NPs for biomedical imaging.
The Evolution of Structural Imaging to Functional and Molecular Imaging of Bone
Bone is a composite material consisting primarily of hydroxyapatite crystal and type I collagen, which accounts for ∼90% of the organic component. Because bone has high mineral content and higher attenuation of x-rays than the surrounding soft tissue, conventional roentgenography was, when introduced in 1895, the first noninvasive structural imaging modality for bone. Because of the advances in physics, digital geometric processing, and computational power, computed tomography (CT) and magnetic resonance imaging (MRI) can process a large array of digital data to reconstruct virtual three-dimensional (3D) images of bone and soft tissue . However, functional changes usually precede structural changes in disease process, and molecular signaling aberrations are fundamental to most diseases. Therefore, clinical demands for advanced imaging technology capable of providing functional and molecular information are urgent.
From the functional point of view, physiological bone homeostasis, repair, and pathologic changes are mediated by a balance of positive and negative remodeling processes. Noninvasive functional imaging of bone includes in vivo imaging of bone formation, resorption, inflammation, vascularization, and mechanical properties. At the molecular level, the bone-remodeling kinetics involves a complex molecular interaction network between the cells and the cell–matrix. Appropriate functional and molecular imaging systems with sufficient spatial and temporal resolution provide valuable information to advance bone research and clinical frontiers.
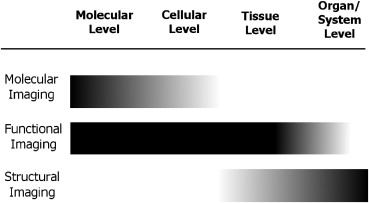
Functional imaging, compared to structural imaging, unravels physiological and pathologic activities, and it has been widely used both in basic medical research and in clinical settings. The scope of functional imaging is shown in Figure 1 . Functional imaging senses signals from biological tissue and reconstructs the information into registered images that reflect regional changes in the blood supply, metabolism, chemical constituents, or physical properties. Decades ago, the introduction of intravascular contrast agents not only allowed physicians to distinguish vessels from other tubular structures in the human body but also shed a light on functional imaging . Contrast-enhanced CT has been used to assess vascularity and permeability to distinguish between benign and malignant lesions in orthopedics and other clinical specialties .
Molecular imaging , an integral part of functional imaging, is recognized as an important medical advance toward personalized medicine to optimize disease treatment. Detecting subtle functional changes at the molecular level provides advanced diagnoses and clues to therapeutic strategies. The development of molecular probes and new sensing and image processing technologies has contributed to the emergence of molecular imaging. To map the molecular events in vivo, the imaging probes consist of a targeting unit that directs the probe to the site of interest and a reporter unit that emits signals spontaneously or on specific stimulation.
The Targeting Unit of Bone-imaging Probes
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
The Reporter Unit of Bone-imaging Probes: From Radionuclides to Near-infrared Fluorophores to Nanomaterials
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Current Noninvasive Functional Imaging Modalities in Orthopedic Clinics and Research
Modality 99m Tc-MDP SPECT 18 F-NaF PET Contrast-enhanced CT Dual-energy CT Proton MRI Doppler Ultrasound Image acquisition time Minutes Shorter than SPECT Minutes Minutes Minutes to hours 10–30 milliseconds Spatial resolution ∗ 7–15 mm 6–10 mm 0.5–2.0 mm <1 mm 10–100 μm 50 μm Detection depth Whole body Whole body Whole body Whole body Whole body Centimeters Sensitivity † 10 −10 to 10 −11 mol 10 −11 to 10 −12 mol
Greater than CT 10 −3 to 10 −5 mol for Gd contrast agent
10 −6 to 10 −9 mol for selective nanoparticles — Specificity Low Greater than SPECT
High High — Volumetric quantification Relative Absolute Absolute Absolute Absolute — Cost of infrastructure High Higher than SPECT Moderate Moderate High Low Cost of each use Moderate High Low Low Low Low Detection applications
Major advantages
Major disadvantages
CT, computed tomography; MDP, methylenediphosphonate; MRI, magnetic resonance imaging; NaF, sodium fluoride; PET, positron emission tomography; SPECT, single photon emission computed tomography.
Data are integrated from the previous studies .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Current Noninvasive Functional Imaging Modalities in Orthopedic Research Only
Modality Bioluminescence Imaging (BLI) Fluorescence Reflectance Imaging (FRI) ∗ Fluorescence Molecular Tomography (FMT) Photoacoustic Imaging (PAI) Energy source Chemical luminescence
Near-infrared Near-infrared laser Image acquisition time Seconds to minutes Seconds to minutes Minutes Milliseconds Spatial resolution 3–5 mm 1–3 mm <1 mm ∼100 μm Detection depth <3 cm <1 cm <20 cm ∼5 cm Sensitivity 10 −15 to 10 −17 mol 10 −9 to 10 −12 mol 10 −13 mol
Specificity High High High High with targeting contrast Volumetric quantification Relative Relative Absolute Relative Cost of infrastructure Moderate Moderate Moderate Low Cost of each use Low Low Low Low Detection capability Molecular
Major advantages
Major disadvantages
Longer image processing time than BLI and FRI Relatively lower sensitivity
NIR, near-infrared; 2D, two-dimensional; 3D, three-dimensional.
Data are integrated from the previous studies .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Imaging modalities for the skeletal system
Radionuclide Imaging of Bone Metabolic Activity
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Fluorescence Reflectance Imaging and Fluorescence Molecular Tomography
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Optical Imaging of Osteoblastic Activity
Get Radiology Tree app to read full this article<
Optical Imaging of Osteoclastic Activity
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Raman Spectroscopic Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 3
Current Noninvasive “Bone Quality” Imaging Modalities
Modality Dual-energy X-ray Absorptiometry Quantitative Computed Tomography Quantitative Ultrasound High-resolution Magnetic Resonance Imaging Raman Spectroscopic Imaging Variable derived from bone tissue Bone mineral density per unit projection body surface area
Skeleton assessed Central (hip and spine) Central and peripheral Peripheral (calcaneus, radius, phalanges, and tibia) Central and peripheral Peripheral Image acquisition time Minutes Minutes Seconds to minutes Minutes to hours Minutes Ionizing radiation exposure Low (0.08–4.6 μSv for pencil beam; 6.7–31 μSv for fan beam)
No No No Portability Poor Poor Good Poor Moderate Application Clinical standard Clinical Clinical Research Research
Data are integrated from the previous studies .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Magnetic Resonance Imaging and Magnetic Resonance Spectroscopy for Functional Imaging of Bone
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Ultrasound for Functional Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Nanomedicine-enabled imaging in orthopedics
Get Radiology Tree app to read full this article<
Nanomaterials for Magnetic Resonance Imaging Probes
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Gold NPs in Computed Tomography and Photoacoustic Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Other Optically Active Nanomaterials for Optical Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Organic Materials for Bone Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Upconversion NPs
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Developing functional imaging technologies
The Modular Design in Targeting
Get Radiology Tree app to read full this article<
Multimodal Imaging Contrast Agents
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 4
Comparison of Nanoparticle (NP) Imaging Probes for Functional Imaging in Clinical and Research Use
Probes Imaging Modality Mechanism Advantages/Disadvantages Clinical and Research Implications References Gold nanoparticles Fluorescence microscopy or FRI Plasmon-based photoluminescence
Dark-field microspectroscopy Strong and characteristic light scattering Optical coherence tomography Strong light scattering CT Photon absorption MRI contrast enhancer
PAI Strong light absorption Surface-enhanced Raman spectroscopy Huge increase of the Raman signal, by a factor of about 10 14 to 10 15 to allow single molecular detection Superparamagnetic iron oxide NP MRI Disturbance of magnetic field
Quantum dot (QD) Fluorescence reflectance imaging Quantum confinement effect
Lanthanide-doped upconversion nanoparticle (UCNP) Optical imaging
CT, computed tomography; FRI, fluorescence reflectance imaging; MRI, magnetic resonance imaging; NIR, near-infrared; PAI, photoacoustic imaging.
Common charateristics for NPs listed in the table: (1) surface modification for multiple biological and chemical functions, (2) integrated properties for multimodality imaging, (3) good photostability, (4) adaptive physicochemical and biological properties for various imaging modalities.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Holliday R.A.: Inflammatory diseases of the temporal bone: evaluation with CT and MR. Semin Ultrasound CT MR 1989; 10: pp. 213-235.
2. Huckman M.S.: Clinical experience with the intravenous infusion of iodinated contrast material as an adjunct to computed tomography. Surg Neurol 1975; 4: pp. 297-318.
3. Thornton M.M.: Multi-modality imaging of musculoskeletal disease in small animals. J Musculoskelet Neuronal Interact 2004; 4: pp. 364.
4. Hill J.H., Mafee M.F., Lygizos N.A., et. al.: Dynamic computed tomography. Its use in the assessment of vascular malformations and angiofibroma. Arch Otolaryngol 1985; 111: pp. 62-65.
5. Hasegawa M., Fujisawa H., Hayashi Y., et. al.: CT arteriography for orbital tumors: diagnostic and surgical value. J Clin Neurosci 2005; 12: pp. 548-552.
6. Okada M., Kondo H., Sou H., et. al.: The efficacy of contrast protocol in hepatic dynamic computed tomography: multicenter prospective study in community hospitals. Springerplus 2013; 2: pp. 367.
7. Levine E., Neff J.R.: Dynamic computed tomography scanning of benign bone lesions: preliminary results. Skeletal Radiol 1983; 9: pp. 238-245.
8. Reumann M.K., Weiser M.C., Mayer-Kuckuk P.: Musculoskeletal molecular imaging: a comprehensive overview. Trends Biotechnol 2010; 28: pp. 93-101.
9. Wilmot A., Gieschler S., Behera D., et. al.: Molecular imaging: an innovative force in musculoskeletal radiology. AJR Am J Roentgenol 2013; 201: pp. 264-277.
10. Mayer-Kuckuk P., Boskey A.L.: Molecular imaging promotes progress in orthopedic research. Bone 2006; 39: pp. 965-977.
11. Hokugo A., Sun S., Park S., et. al.: Equilibrium-dependent bisphosphonate interaction with crystalline bone mineral explains anti-resorptive pharmacokinetics and prevalence of osteonecrosis of the jaw in rats. Bone 2013; 53: pp. 59-68.
12. Sato M., Grasser W., Endo N., et. al.: Bisphosphonate action. Alendronate localization in rat bone and effects on osteoclast ultrastructure. J Clin Invest 1991; 88: pp. 2095-2105.
13. Ozcan I., Bouchemal K., Segura-Sanchez F., et. al.: Synthesis and characterization of surface-modified PBLG nanoparticles for bone targeting: in vitro and in vivo evaluations. J Pharm Sci 2011; 100: pp. 4877-4887.
14. Laketic-Ljubojevic I., Suva L.J., Maathuis F.J., et. al.: Functional characterization of N-methyl-D-aspartic acid-gated channels in bone cells. Bone 1999; 25: pp. 631-637.
15. Panahifar A., Mahmoudi M., Doschak M.R.: Synthesis and in vitro evaluation of bone-seeking superparamagnetic iron oxide nanoparticles as contrast agents for imaging bone metabolic activity. ACS Appl Mater Interfaces 2013; 5: pp. 5219-5226.
16. Zhang Z., Ross R.D., Roeder R.K.: Preparation of functionalized gold nanoparticles as a targeted X-ray contrast agent for damaged bone tissue. Nanoscale 2010; 2: pp. 582-586.
17. Sheldon A.L., Robinson M.B.: The role of glutamate transporters in neurodegenerative diseases and potential opportunities for intervention. Neurochem Int 2007; 51: pp. 333-355.
18. Ross R.D., Roeder R.K.: Binding affinity of surface functionalized gold nanoparticles to hydroxyapatite. J Biomed Mater Res A 2011; 99: pp. 58-66.
19. Bernhard J.M., Blanks J.K., Hintz C.J., et. al.: Use of the fluorescent calcite marker calcein to label foraminiferal tests. J Foraminiferal Res 2004; 34: pp. 96-101.
20. Pautke C., Vogt S., Tischer T., et. al.: Polychrome labeling of bone with seven different fluorochromes: enhancing fluorochrome discrimination by spectral image analysis. Bone 2005; 37: pp. 441-445.
21. Suzuki H.K., Mathews A.: Two-color fluorescent labeling of mineralizing tissues with tetracycline and 2,4-bis[N,N’-di-(carbomethyl)aminomethyl] fluorescein. Stain Technol 1966; 41: pp. 57-60.
22. Milch R.A., Rall D.P., Tobie J.E.: Fluorescence of tetracycline antibiotics in bone. J Bone Joint Surg Am 1958; 40-A: pp. 897-910.
23. Dhem A., Piret N., Fortunati D.: Tetracyclines, doxycycline and calcified tissues. Scand J Infect Dis Suppl 1976; pp. 42-46.
24. Rahn B.A., Perren S.M.: [Alizarin complexon-fluorochrome for bone and dentine labeling]. Experientia 1972; 28: pp. 180.
25. Rahn B.A., Perren S.M.: Xylenol orange, a fluorochrome useful in polychrome sequential labeling of calcifying tissues. Stain Technol 1971; 46: pp. 125-129.
26. Choe R., Konecky S.D., Corlu A., et. al.: Differentiation of benign and malignant breast tumors by in-vivo three-dimensional parallel-plate diffuse optical tomography. J Biomed Opt 2009; 14: pp. 024020.
27. Guven M., Yazici B., Intes X., et. al.: Diffuse optical tomography with a priori anatomical information. Phys Med Biol 2005; 50: pp. 2837-2858.
28. Intes X., Ntziachristos V., Culver J.P., et. al.: Projection access order in algebraic reconstruction technique for diffuse optical tomography. Phys Med Biol 2002; 47: pp. N1-N10.
29. Patterson M.S., Chance B., Wilson B.C.: Time resolved reflectance and transmittance for the non-invasive measurement of tissue optical properties. Appl Opt 1989; 28: pp. 2331-2336.
30. Chen Y., Mu C., Intes X., et. al.: Adaptive calibration for object localization in turbid media with interfering diffuse photon density waves. Appl Opt 2002; 41: pp. 7325-7333.
31. Yodh A., Chance B.: Spectroscopy and imaging with diffusing light. Physics Today 1995; 48: pp. 34-40.
32. Quek C.H., Leong K.W.: Near-infrared fluorescent nanoprobes for in vivo optical imaging. Nanomaterials 2012; 2: pp. 92-112.
33. Cohn S.M.: Near-infrared spectroscopy: potential clinical benefits in surgery. J Am Coll Surg 2007; 205: pp. 322-332.
34. Murkin J.M., Arango M.: Near-infrared spectroscopy as an index of brain and tissue oxygenation. Br J Anaesth 2009; 103: pp. i3-i13.
35. Smith A.M., Mancini M.C., Nie S.: Bioimaging: second window for in vivo imaging. Nat Nanotechnol 2009; 4: pp. 710-711.
36. Lim Y.T., Kim S., Nakayama A., et. al.: Selection of quantum dot wavelengths for biomedical assays and imaging. Mol Imaging 2003; 2: pp. 50-64.
37. Hong G., Lee J.C., Robinson J.T., et. al.: Multifunctional in vivo vascular imaging using near-infrared II fluorescence. Nat Med 2012; 18: pp. 1841-1846.
38. Won N., Jeong S., Kim K., et. al.: Imaging depths of near-infrared quantum dots in first and second optical windows. Mol Imaging 2012; 11: pp. 338-352.
39. Welsher K., Liu Z., Sherlock S.P., et. al.: A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat Nanotechnol 2009; 4: pp. 773-780.
40. Zhang Y., Hong G., Zhang Y., et. al.: Ag2S quantum dot: a bright and biocompatible fluorescent nanoprobe in the second near-infrared window. ACS Nano 2012; 6: pp. 3695-3702.
41. Hu K.W., Liu T.M., Chung K.Y., et. al.: Efficient near-IR hyperthermia and intense nonlinear optical imaging contrast on the gold nanorod-in-shell nanostructures. J Am Chem Soc 2009; 131: pp. 14186-14187.
42. Balandin A.A.: Nanophononics: phonon engineering in nanostructures and nanodevices. J Nanosci Nanotechnol 2005; 5: pp. 1015-1022.
43. Liao H., Nehl C.L., Hafner J.H.: Biomedical applications of plasmon resonant metal nanoparticles. Nanomedicine (Lond) 2006; 1: pp. 201-208.
44. Kaur R., Badea I.: Nanodiamonds as novel nanomaterials for biomedical applications: drug delivery and imaging systems. Int J Nanomedicine 2013; 8: pp. 203-220.
45. Zhang G., Zeng X., Li P.: Nanomaterials in cancer-therapy drug delivery system. J Biomed Nanotechnol 2013; 9: pp. 741-750.
46. Coto-Garcia A.M., Sotelo-Gonzalez E., Fernandez-Arguelles M.T., et. al.: Nanoparticles as fluorescent labels for optical imaging and sensing in genomics and proteomics. Anal Bioanal Chem 2011; 399: pp. 29-42.
47. Melendez-Alafort L., Muzzio P.C., Rosato A.: Optical and multimodal peptide-based probes for in vivo molecular imaging. Anticancer Agents Med Chem 2012; 12: pp. 476-499.
48. Jokerst J.V., Gambhir S.S.: Molecular imaging with theranostic nanoparticles. Acc Chem Res 2011; 44: pp. 1050-1060.
49. Souris J.S.: Seeing the light in bone metabolism imaging. Trends Biotechnol 2002; 20: pp. 364-366.
50. Einhorn T.A., Vigorita V.J., Aaron A.: Localization of technetium-99m methylene diphosphonate in bone using microautoradiography. J Orthop Res 1986; 4: pp. 180-187.
51. Grynpas M.D.: Fluoride effects on bone crystals. J Bone Miner Res 1990; 5: pp. S169-S175.
52. Piert M., Zittel T.T., Becker G.A., et. al.: Assessment of porcine bone metabolism by dynamic. J Nucl Med 2001; 42: pp. 1091-1100.
53. Messa C., Goodman W.G., Hoh C.K., et. al.: Bone metabolic activity measured with positron emission tomography and [18F]fluoride ion in renal osteodystrophy: correlation with bone histomorphometry. J Clin Endocrinol Metab 1993; 77: pp. 949-955.
54. Blau M., Nagler W., Bender M.A.: Fluorine-18: a new isotope for bone scanning. J Nucl Med 1962; 3: pp. 332-334.
55. Wong K.K., Piert M.: Dynamic bone imaging with 99mTc-labeled diphosphonates and 18F-NaF: mechanisms and applications. J Nucl Med 2013; 54: pp. 590-599.
56. Blake G.M., Park-Holohan S.J., Cook G.J., et. al.: Quantitative studies of bone with the use of 18F-fluoride and 99mTc-methylene diphosphonate. Semin Nucl Med 2001; 31: pp. 28-49.
57. Czernin J., Satyamurthy N., Schiepers C.: Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med 2010; 51: pp. 1826-1829.
58. Grant F.D., Fahey F.H., Packard A.B., et. al.: Skeletal PET with 18F-fluoride: applying new technology to an old tracer. J Nucl Med 2008; 49: pp. 68-78.
59. Hetzel M., Arslandemir C., Konig H.H., et. al.: F-18 NaF PET for detection of bone metastases in lung cancer: accuracy, cost-effectiveness, and impact on patient management. J Bone Miner Res 2003; 18: pp. 2206-2214.
60. Schirrmeister H., Glatting G., Hetzel J., et. al.: Prospective evaluation of the clinical value of planar bone scans, SPECT, and (18)F-labeled NaF PET in newly diagnosed lung cancer. J Nucl Med 2001; 42: pp. 1800-1804.
61. Schirrmeister H., Guhlmann A., Elsner K., et. al.: Sensitivity in detecting osseous lesions depends on anatomic localization: planar bone scintigraphy versus 18F PET. J Nucl Med 1999; 40: pp. 1623-1629.
62. Schirrmeister H., Guhlmann A., Kotzerke J., et. al.: Early detection and accurate description of extent of metastatic bone disease in breast cancer with fluoride ion and positron emission tomography. J Clin Oncol 1999; 17: pp. 2381-2389.
63. Temmerman O.P., Raijmakers P.G., Heyligers I.C., et. al.: Bone metabolism after total hip revision surgery with impacted grafting: evaluation using H2 15O and [18F]fluoride PET; a pilot study. Mol Imaging Biol 2008; 10: pp. 288-293.
64. Cook G.J., Blake G.M., Marsden P.K., et. al.: Quantification of skeletal kinetic indices in Paget’s disease using dynamic 18F-fluoride positron emission tomography. J Bone Miner Res 2002; 17: pp. 854-859.
65. Schiepers C., Nuyts J., Bormans G., et. al.: Fluoride kinetics of the axial skeleton measured in vivo with fluorine-18-fluoride PET. J Nucl Med 1997; 38: pp. 1970-1976.
66. Installe J., Nzeusseu A., Bol A., et. al.: (18)F-fluoride PET for monitoring therapeutic response in Paget’s disease of bone. J Nucl Med 2005; 46: pp. 1650-1658.
67. Nicolaou S., Liang T., Murphy D.T., et. al.: Dual-energy CT: a promising new technique for assessment of the musculoskeletal system. AJR Am J Roentgenol 2012; 199: pp. S78-S86.
68. Barber P.A., Rushforth D., Agrawal S., et. al.: Infrared optical imaging of matrix metalloproteinases (MMPs) up regulation following ischemia reperfusion is ameliorated by hypothermia. BMC Neurosci 2012; 13: pp. 76.
69. Rasmussen J.C., Tan I.C., Marshall M.V., et. al.: Human lymphatic architecture and dynamic transport imaged using near-infrared fluorescence. Transl Oncol 2010; 3: pp. 362-372.
70. Massoud T.F., Gambhir S.S.: Molecular imaging in living subjects: seeing fundamental biological processes in a new light. Genes Dev 2003; 17: pp. 545-580.
71. Kobayashi H., Ogawa M., Alford R., et. al.: New strategies for fluorescent probe design in medical diagnostic imaging. Chem Rev 2010; 110: pp. 2620-2640.
72. Das B.B., Liu F., Alfano R.R.: Time-resolved fluorescence and photon migration studies in biomedical and model random media. Rep Prog Phys 1997; 60: pp. 227-292.
73. Xu Y., Iftimia N., Jiang H., et. al.: Imaging of in vitro and in vivo bones and joints with continuous-wave diffuse optical tomography. Opt Express 2001; 8: pp. 447-451.
74. Xu Y., Iftimia N., Jiang H., et. al.: Three-dimensional diffuse optical tomography of bones and joints. J Biomed Opt 2002; 7: pp. 88-92.
75. Ntziachristos V., Ripoll J., Wang L.V., et. al.: Looking and listening to light: the evolution of whole-body photonic imaging. Nat Biotechnol 2005; 23: pp. 313-320.
76. Ntziachristos V., Tung C.H., Bremer C., et. al.: Fluorescence molecular tomography resolves protease activity in vivo. Nat Med 2002; 8: pp. 757-760.
77. Kozloff K.M., Volakis L.I., Marini J.C., et. al.: Near-infrared fluorescent probe traces bisphosphonate delivery and retention in vivo. J Bone Miner Res 2010; 25: pp. 1748-1758.
78. Kozloff K.M., Weissleder R., Mahmood U.: Noninvasive optical detection of bone mineral. J Bone Miner Res 2007; 22: pp. 1208-1216.
79. Lambers F.M., Stuker F., Weigt C., et. al.: Longitudinal in vivo imaging of bone formation and resorption using fluorescence molecular tomography. Bone 2013; 52: pp. 587-595.
80. Zaheer A., Lenkinski R.E., Mahmood A., et. al.: In vivo near-infrared fluorescence imaging of osteoblastic activity. Nat Biotechnol 2001; 19: pp. 1148-1154.
81. Kovar J.L., Xu X., Draney D., et. al.: Near-infrared-labeled tetracycline derivative is an effective marker of bone deposition in mice. Anal Biochem 2011; 416: pp. 167-173.
82. Drake F.H., Dodds R.A., James I.E., et. al.: Cathepsin K, but not cathepsins B, L, or S, is abundantly expressed in human osteoclasts. J Biol Chem 1996; 271: pp. 12511-12516.
83. Teitelbaum S.L.: Bone resorption by osteoclasts. Science 2000; 289: pp. 1504-1508.
84. Gowen M., Lazner F., Dodds R., et. al.: Cathepsin K knockout mice develop osteopetrosis due to a deficit in matrix degradation but not demineralization. J Bone Miner Res 1999; 14: pp. 1654-1663.
85. Sprague J.E., Kitaura H., Zou W., et. al.: Noninvasive imaging of osteoclasts in parathyroid hormone-induced osteolysis using a 64Cu-labeled RGD peptide. J Nucl Med 2007; 48: pp. 311-318.
86. Kozloff K.M., Quinti L., Patntirapong S., et. al.: Non-invasive optical detection of cathepsin K-mediated fluorescence reveals osteoclast activity in vitro and in vivo. Bone 2009; 44: pp. 190-198.
87. Lecaille F., Weidauer E., Juliano M.A., et. al.: Probing cathepsin K activity with a selective substrate spanning its active site. Biochem J 2003; 375: pp. 307-312.
88. Kowada T., Kikuta J., Kubo A., et. al.: In vivo fluorescence imaging of bone-resorbing osteoclasts. J Am Chem Soc 2011; 133: pp. 17772-17776.
89. Pelletier M.J.: Analytical applications of Raman spectroscopy.1st ed.1999.Blackwell Publishing
90. Diem M.: Introduction to modern vibrational spectroscopy.1st ed.1993.Wiley-InterscienceHoboken, NJ
91. Carden A., Morris M.D.: Application of vibrational spectroscopy to the study of mineralized tissues (review). J Biomed Opt 2000; 5: pp. 259-268.
92. Sahar N.D., Hong S.I., Kohn D.H.: Micro- and nano-structural analyses of damage in bone. Micron 2005; 36: pp. 617-629.
93. Tarnowski C.P., Ignelzi M.A., Morris M.D.: Mineralization of developing mouse calvaria as revealed by Raman microspectroscopy. J Bone Miner Res 2002; 17: pp. 1118-1126.
94. Matousek P., Clark I.P., Draper E.R., et. al.: Subsurface probing in diffusely scattering media using spatially offset Raman spectroscopy. Appl Spectrosc 2005; 59: pp. 393-400.
95. Matousek P., Morris M.D., Everall N., et. al.: Numerical simulations of subsurface probing in diffusely scattering media using spatially offset Raman spectroscopy. Appl Spectrosc 2005; 59: pp. 1485-1492.
96. Matousek P., Draper E.R., Goodship A.E., et. al.: Noninvasive Raman spectroscopy of human tissue in vivo. Appl Spectrosc 2006; 60: pp. 758-763.
97. Matousek P., Stone N.: Recent advances in the development of Raman spectroscopy for deep non-invasive medical diagnosis. J Biophotonics 2013; 6: pp. 7-19.
98. Paschalis E.P., Shane E., Lyritis G., et. al.: Bone fragility and collagen cross-links. J Bone Miner Res 2004; 19: pp. 2000-2004.
99. Paschalis E.P., Verdelis K., Doty S.B., et. al.: Spectroscopic characterization of collagen cross-links in bone. J Bone Miner Res 2001; 16: pp. 1821-1828.
100. Draper E.R., Morris M.D., Camacho N.P., et. al.: Novel assessment of bone using time-resolved transcutaneous Raman spectroscopy. J Bone Miner Res 2005; 20: pp. 1968-1972.
101. Schulmerich M.V., Dooley K.A., Morris M.D., et. al.: Transcutaneous fiber optic Raman spectroscopy of bone using annular illumination and a circular array of collection fibers. J Biomed Opt 2006; 11: pp. 060502.
102. Maher J.R., Inzana J.A., Awad H.A., et. al.: Overconstrained library-based fitting method reveals age- and disease-related differences in transcutaneous Raman spectra of murine bones. J Biomed Opt 2013; 18: pp. 077001.
103. Reisz R.R., Huang T.D., Roberts E.M., et. al.: Embryology of Early Jurassic dinosaur from China with evidence of preserved organic remains. Nature 2013; 496: pp. 210-214.
104. Boskey A., Pleshko Camacho N.: FT-IR imaging of native and tissue-engineered bone and cartilage. Biomaterials 2007; 28: pp. 2465-2478.
105. Massiot D., Messinger R.J., Cadars S., et. al.: Topological, geometric, and chemical order in materials: insights from solid-state NMR. Acc Chem Res 2013; 46: pp. 1975-1984.
106. Young I.R., Bydder G.M.: Magnetic resonance: new approaches to imaging of the musculoskeletal system. Physiol Meas 2003; 24: pp. R1-R23.
107. Wehrli F.W.: Magnetic resonance of calcified tissues. J Magn Reson 2013; 229: pp. 35-48.
108. Noseworthy M.D., Bulte D.P., Alfonsi J.: BOLD magnetic resonance imaging of skeletal muscle. Semin Musculoskelet Radiol 2003; 7: pp. 307-315.
109. Bierry G., Dietemann J.L.: Imaging evaluation of inflammation in the musculoskeletal system: current concepts and perspectives. Skeletal Radiol 2013; 42: pp. 1347-1359.
110. Jasanoff A.: Functional MRI using molecular imaging agents. Trends Neurosci 2005; 28: pp. 120-126.
111. Gade T.P., Motley M.W., Beattie B.J., et. al.: Imaging of alkaline phosphatase activity in bone tissue. PLoS One 2011; 6: pp. e22608.
112. Werner E.J., Datta A., Jocher C.J., et. al.: High-relaxivity MRI contrast agents: where coordination chemistry meets medical imaging. Angew Chem Int Ed Engl 2008; 47: pp. 8568-8580.
113. Hao D., Ai T., Goerner F., et. al.: MRI contrast agents: basic chemistry and safety. J Magn Reson Imaging 2012; 36: pp. 1060-1071.
114. Xue S., Qiao J., Pu F., et. al.: Design of a novel class of protein-based magnetic resonance imaging contrast agents for the molecular imaging of cancer biomarkers. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2013; 5: pp. 163-179.
115. Basilion J.P., Yeon S., Botnar R.: Magnetic resonance imaging: utility as a molecular imaging modality. Curr Top Dev Biol 2005; 70: pp. 1-33.
116. Matsumoto Y., Jasanoff A.: Metalloprotein-based MRI probes. FEBS Lett 2013; 587: pp. 1021-1029.
117. Chance B., Im J., Nioka S., et. al.: Skeletal muscle energetics with PNMR: personal views and historic perspectives. NMR Biomed 2006; 19: pp. 904-926.
118. Akki A., Gupta A., Weiss R.G.: Magnetic resonance imaging and spectroscopy of the murine cardiovascular system. Am J Physiol Heart Circ Physiol 2013; 304: pp. H633-H648.
119. Scheau C., Preda E.M., Popa G.A., et. al.: Magnetic resonance spectroscopy–a non-invasive method in evaluating focal and diffuse central nervous system disease. J Med Life 2012; 5: pp. 423-427.
120. Maris J.M., Chance B.: Magnetic resonance spectroscopy of neoplasms. Magn Reson Annu 1986; pp. 213-235.
121. Kuhnt D., Bauer M.H., Ganslandt O., et. al.: Functional imaging: where do we go from here?. J Neurosurg Sci 2013; 57: pp. 1-11.
122. Ramm Sander P., Hau P., Koch S., et. al.: Stem cell metabolic and spectroscopic profiling. Trends Biotechnol 2013; 31: pp. 204-213.
123. Leonard J.C., Younkin D.P., Chance B., et. al.: Nuclear magnetic resonance: an overview of its spectroscopic and imaging applications in pediatric patients. J Pediatr 1985; 106: pp. 756-761.
124. Sapega A.A., Sokolow D.P., Graham T.J., et. al.: Phosphorus nuclear magnetic resonance: a non-invasive technique for the study of muscle bioenergetics during exercise. Med Sci Sports Exerc 1987; 19: pp. 410-420.
125. Heppenstall R.B., Sapega A.A., Scott R., et. al.: The compartment syndrome. An experimental and clinical study of muscular energy metabolism using phosphorus nuclear magnetic resonance spectroscopy. Clin Orthop Relat Res 1988; pp. 138-155.
126. Tamura M., Hazeki O., Nioka S., et. al.: In vivo study of tissue oxygen metabolism using optical and nuclear magnetic resonance spectroscopies. Annu Rev Physiol 1989; 51: pp. 813-834.
127. Chance B., Smith D.S., Delivoria-Papadopoulos M., et. al.: New techniques for evaluating metabolic brain injury in newborn infants. Crit Care Med 1989; 17: pp. 465-471.
128. Sapega A.A., Sokolow D.P., Graham T.J., et. al.: Phosphorus nuclear magnetic resonance: a non-invasive technique for the study of muscle bioenergetics during exercise. Med Sci Sports Exerc 1993; 25: pp. 656-666.
129. Cao H., Ackerman J.L., Hrovat M.I., et. al.: Quantitative bone matrix density measurement by water- and fat-suppressed proton projection MRI (WASPI) with polymer calibration phantoms. Magn Reson Med 2008; 60: pp. 1433-1443.
130. Scheel A.K., Hermann K.G., Kahler E., et. al.: A novel ultrasonographic synovitis scoring system suitable for analyzing finger joint inflammation in rheumatoid arthritis. Arthritis Rheum 2005; 52: pp. 733-743.
131. Taylor P.C.: VEGF and imaging of vessels in rheumatoid arthritis. Arthritis Res 2002; 4: pp. S99-S107.
132. Klauser A.S., Peetrons P.: Developments in musculoskeletal ultrasound and clinical applications. Skeletal Radiol 2010; 39: pp. 1061-1071.
133. Unnikrishnan S., Klibanov A.L.: Microbubbles as ultrasound contrast agents for molecular imaging: preparation and application. AJR Am J Roentgenol 2012; 199: pp. 292-299.
134. Wei K., Jayaweera A.R., Firoozan S., et. al.: Quantification of myocardial blood flow with ultrasound-induced destruction of microbubbles administered as a constant venous infusion. Circulation 1998; 97: pp. 473-483.
135. Pochon S., Tardy I., Bussat P., et. al.: BR55: a lipopeptide-based VEGFR2-targeted ultrasound contrast agent for molecular imaging of angiogenesis. Invest Radiol 2010; 45: pp. 89-95.
136. Koenig S.H., Brown R.D.: Relaxometry of magnetic resonance imaging contrast agents. Magn Reson Annu 1987; pp. 263-286.
137. Shieh D.B., Cheng F.Y., Su C.H., et. al.: Aqueous dispersions of magnetite nanoparticles with NH3+ surfaces for magnetic manipulations of biomolecules and MRI contrast agents. Biomaterials 2005; 26: pp. 7183-7191.
138. Cheng F.Y., Su C.H., Yang Y.S., et. al.: Characterization of aqueous dispersions of Fe(3)O(4) nanoparticles and their biomedical applications. Biomaterials 2005; 26: pp. 729-738.
139. Wang Y.X., Hussain S.M., Krestin G.P.: Superparamagnetic iron oxide contrast agents: physicochemical characteristics and applications in MR imaging. Eur Radiol 2001; 11: pp. 2319-2331.
140. Rosen J.E., Chan L., Shieh D.B., et. al.: Iron oxide nanoparticles for targeted cancer imaging and diagnostics. Nanomedicine 2012; 8: pp. 275-290.
141. Barja B.C., Herszage J., dos Santos Afonso M.: Iron(III)–phosphonate complexes. Polyhedron 2001; 20: pp. 1821-1830.
142. Lalatonne Y., Monteil M., Jouni H., et. al.: Superparamagnetic bifunctional bisphosphonates nanoparticles: a potential MRI contrast agent for osteoporosis therapy and diagnostic. J Osteoporos 2010; 2010: pp. 747852.
143. Jendelova P., Herynek V., DeCroos J., et. al.: Imaging the fate of implanted bone marrow stromal cells labeled with superparamagnetic nanoparticles. Magn Reson Med 2003; 50: pp. 767-776.
144. Balakumaran A., Pawelczyk E., Ren J., et. al.: Superparamagnetic iron oxide nanoparticles labeling of bone marrow stromal (mesenchymal) cells does not affect their “stemness”. PLoS One 2010; 5: pp. e11462.
145. Jing X.H., Yang L., Duan X.J., et. al.: In vivo MR imaging tracking of magnetic iron oxide nanoparticle labeled, engineered, autologous bone marrow mesenchymal stem cells following intra-articular injection. Joint Bone Spine 2008; 75: pp. 432-438.
146. Jasmin , Torres A.L., Nunes H.M., et. al.: Optimized labeling of bone marrow mesenchymal cells with superparamagnetic iron oxide nanoparticles and in vivo visualization by magnetic resonance imaging. J Nanobiotechnology 2011; 9: pp. 4.
147. Hainfeld J.F., Slatkin D.N., Focella T.M., et. al.: Gold nanoparticles: a new X-ray contrast agent. Br J Radiol 2006; 79: pp. 248-253.
148. Au J.T., Craig G., Longo V., et. al.: Gold nanoparticles provide bright long-lasting vascular contrast for CT imaging. AJR Am J Roentgenol 2013; 200: pp. 1347-1351.
149. Kim D., Jeong Y.Y., Jon S.: A drug-loaded aptamer-gold nanoparticle bioconjugate for combined CT imaging and therapy of prostate cancer. ACS Nano 2010; 4: pp. 3689-3696.
150. Kim D., Park S., Lee J.H., et. al.: Antibiofouling polymer-coated gold nanoparticles as a contrast agent for in vivo X-ray computed tomography imaging. J Am Chem Soc 2007; 129: pp. 7661-7665.
151. Li J., Chaudhary A., Chmura S.J., et. al.: A novel functional CT contrast agent for molecular imaging of cancer. Phys Med Biol 2010; 55: pp. 4389-4397.
152. Kojima C., Umeda Y., Ogawa M., et. al.: X-ray computed tomography contrast agents prepared by seeded growth of gold nanoparticles in PEGylated dendrimer. Nanotechnology 2010; 21: pp. 245104.
153. Kattumuri V., Katti K., Bhaskaran S., et. al.: Gum arabic as a phytochemical construct for the stabilization of gold nanoparticles: in vivo pharmacokinetics and X-ray-contrast-imaging studies. Small 2007; 3: pp. 333-341.
154. Manohar S., Ungureanu C., Van Leeuwen T.G.: Gold nanorods as molecular contrast agents in photoacoustic imaging: the promises and the caveats. Contrast Media Mol Imaging 2011; 6: pp. 389-400.
155. Li P.C., Wang C.R., Shieh D.B., et. al.: In vivo photoacoustic molecular imaging with simultaneous multiple selective targeting using antibody-conjugated gold nanorods. Opt Express 2008; 16: pp. 18605-18615.
156. Li P.C., Wei C.W., Liao C.K., et. al.: Photoacoustic imaging of multiple targets using gold nanorods. IEEE Trans Ultrason Ferroelectr Freq Control 2007; 54: pp. 1642-1647.
157. Seekell K., Crow M.J., Marinakos S., et. al.: Hyperspectral molecular imaging of multiple receptors using immunolabeled plasmonic nanoparticles. J Biomed Opt 2011; 16: pp. 116003.
158. Talbot C.B., Patalay R., Munro I., et. al.: Application of ultrafast gold luminescence to measuring the instrument response function for multispectral multiphoton fluorescence lifetime imaging. Opt Express 2011; 19: pp. 13848-13861.
159. Ha S., Carson A., Agarwal A., et. al.: Detection and monitoring of the multiple inflammatory responses by photoacoustic molecular imaging using selectively targeted gold nanorods. Biomed Opt Express 2011; 2: pp. 645-657.
160. Tong L., Wei Q., Wei A., et. al.: Gold nanorods as contrast agents for biological imaging: optical properties, surface conjugation and photothermal effects. Photochem Photobiol 2009; 85: pp. 21-32.
161. Oldenburg A.L., Hansen M.N., Zweifel D.A., et. al.: Plasmon-resonant gold nanorods as low backscattering albedo contrast agents for optical coherence tomography. Opt Express 2006; 14: pp. 6724-6738.
162. Troutman T.S., Barton J.K., Romanowski M.: Optical coherence tomography with plasmon resonant nanorods of gold. Opt Lett 2007; 32: pp. 1438-1440.
163. Chou C.H., Chen C.D., Wang C.R.: Highly efficient, wavelength-tunable, gold nanoparticle based optothermal nanoconvertors. J Phys Chem B 2005; 109: pp. 11135-11138.
164. Liao C.K., Huang S.W., Wei C.W., et. al.: Nanorod-based flow estimation using a high-frame-rate photoacoustic imaging system. J Biomed Opt 2007; 12: pp. 064006.
165. Chamberland D.L., Agarwal A., Kotov N., et. al.: Photoacoustic tomography of joints aided by an Etanercept-conjugated gold nanoparticle contrast agent – an ex vivo preliminary rat study. Nanotechnology 2008; 19: pp. 095101.
166. Xia Y., Li W., Cobley C.M., et. al.: Gold nanocages: from synthesis to theranostic applications. Acc Chem Res 2011; 44: pp. 914-924.
167. Wang Y., Liu Y., Luehmann H., et. al.: Evaluating the pharmacokinetics and in vivo cancer targeting capability of Au nanocages by positron emission tomography imaging. ACS Nano 2012; 6: pp. 5880-5888.
168. Wang Y., Liu Y., Luehmann H., et. al.: Radioluminescent gold nanocages with controlled radioactivity for real-time in vivo imaging. Nano Lett 2013; 13: pp. 581-585.
169. Petryayeva E., Algar W.R., Medintz I.L.: Quantum dots in bioanalysis: a review of applications across various platforms for fluorescence spectroscopy and imaging. Appl Spectrosc 2013; 67: pp. 215-252.
170. Larson D.R., Zipfel W.R., Williams R.M., et. al.: Water-soluble quantum dots for multiphoton fluorescence imaging in vivo. Science 2003; 300: pp. 1434-1436.
171. Michalet X., Pinaud F.F., Bentolila L.A., et. al.: Quantum dots for live cells, in vivo imaging, and diagnostics. Science 2005; 307: pp. 538-544.
172. Mai W.X., Meng H.: Mesoporous silica nanoparticles: a multifunctional nano therapeutic system. Integr Biol (Camb) 2013; 5: pp. 19-28.
173. Tang F., Li L., Chen D.: Mesoporous silica nanoparticles: synthesis, biocompatibility and drug delivery. Adv Mater 2012; 24: pp. 1504-1534.
174. Huang D.M., Hung Y., Ko B.S., et. al.: Highly efficient cellular labeling of mesoporous nanoparticles in human mesenchymal stem cells: implication for stem cell tracking. FASEB J 2005; 19: pp. 2014-2016.
175. Chung T.H., Wu S.H., Yao M., et. al.: The effect of surface charge on the uptake and biological function of mesoporous silica nanoparticles in 3T3-L1 cells and human mesenchymal stem cells. Biomaterials 2007; 28: pp. 2959-2966.
176. Hsiao J.K., Tsai C.P., Chung T.H., et. al.: Mesoporous silica nanoparticles as a delivery system of gadolinium for effective human stem cell tracking. Small 2008; 4: pp. 1445-1452.
177. van Schooneveld M.M., Cormode D.P., Koole R., et. al.: A fluorescent, paramagnetic and PEGylated gold/silica nanoparticle for MRI, CT and fluorescence imaging. Contrast Media Mol Imaging 2010; 5: pp. 231-236.
178. Beck G.R., Ha S.W., Camalier C.E., et. al.: Bioactive silica-based nanoparticles stimulate bone-forming osteoblasts, suppress bone-resorbing osteoclasts, and enhance bone mineral density in vivo. Nanomedicine 2012; 8: pp. 793-803.
179. Nabeshi H., Yoshikawa T., Akase T., et. al.: Effect of amorphous silica nanoparticles on in vitro RANKL-induced osteoclast differentiation in murine macrophages. Nanoscale Res Lett 2011; 6: pp. 464.
180. Park Y.J., Nah S.H., Lee J.Y., et. al.: Surface-modified poly(lactide-co-glycolide) nanospheres for targeted bone imaging with enhanced labeling and delivery of radioisotope. J Biomed Mater Res A 2003; 67: pp. 751-760.
181. Zhang H.F., Maslov K., Stoica G., et. al.: Functional photoacoustic microscopy for high-resolution and noninvasive in vivo imaging. Nat Biotechnol 2006; 24: pp. 848-851.
182. Zhang H.F., Maslov K., Li M.L., et. al.: In vivo volumetric imaging of subcutaneous microvasculature by photoacoustic microscopy. Opt Express 2006; 14: pp. 9317-9323.
183. Hu S., Wang L.V.: Photoacoustic imaging and characterization of the microvasculature. J Biomed Opt 2010; 15: pp. 011101.
184. Chu S.W., Chen I.H., Liu T.M., et. al.: Nonlinear bio-photonic crystal effects revealed with multimodal nonlinear microscopy. J Microsc 2002; 208: pp. 190-200.
185. Tsai M.R., Chiu Y.W., Lo M.T., et. al.: Second-harmonic generation imaging of collagen fibers in myocardium for atrial fibrillation diagnosis. J Biomed Opt 2010; 15: pp. 026002.
186. Chen S.Y., Yu H.C., Wang I.J., et. al.: Infrared-based third and second harmonic generation imaging of cornea. J Biomed Opt 2009; 14: pp. 044012.
187. Sun C.K., Yu C.H., Tai S.P., et. al.: In vivo and ex vivo imaging of intra-tissue elastic fibers using third-harmonic-generation microscopy. Opt Express 2007; 15: pp. 11167-11177.
188. Hsieh C.S., Chen S.U., Lee Y.W., et. al.: Higher harmonic generation microscopy of in vitro cultured mammal oocytes and embryos. Opt Express 2008; 16: pp. 11574-11588.
189. Tai S.P., Tsai T.H., Lee W.J., et. al.: Optical biopsy of fixed human skin with backward-collected optical harmonics signals. Opt Express 2005; 13: pp. 8231-8242.
190. Sivakumar S., van Veggel F.C., May P.S.: Near-infrared (NIR) to red and green up-conversion emission from silica sol-gel thin films made with La(0.45)Yb(0.50)Er(0.05)F(3) nanoparticles, hetero-looping-enhanced energy transfer (Hetero-LEET): a new up-conversion process. J Am Chem Soc 2007; 129: pp. 620-625.
191. Wang F., Banerjee D., Liu Y., et. al.: Upconversion nanoparticles in biological labeling, imaging, and therapy. Analyst 2010; 135: pp. 1839-1854.
192. Zhang F., Li J., Shan J., et. al.: Shape, size, and phase-controlled rare-Earth fluoride nanocrystals with optical up-conversion properties. Chemistry 2009; 15: pp. 11010-11019.
193. Cheng L., Wang C., Liu Z.: Upconversion nanoparticles and their composite nanostructures for biomedical imaging and cancer therapy. Nanoscale 2013; 5: pp. 23-37.
194. Hasna K., Kumar S.S., Komath M., et. al.: Synthesis of chemically pure, luminescent Eu3+ doped HAp nanoparticles: a promising fluorescent probe for in vivo imaging applications. Phys Chem Chem Phys 2013; 15: pp. 8106-8111.
195. Lin M., Zhao Y., Wang S., et. al.: Recent advances in synthesis and surface modification of lanthanide-doped upconversion nanoparticles for biomedical applications. Biotechnol Adv 2012; 30: pp. 1551-1561.
196. Chatterjee D.K., Rufaihah A.J., Zhang Y.: Upconversion fluorescence imaging of cells and small animals using lanthanide doped nanocrystals. Biomaterials 2008; 29: pp. 937-943.
197. Idris N.M., Li Z., Ye L., et. al.: Tracking transplanted cells in live animal using upconversion fluorescent nanoparticles. Biomaterials 2009; 30: pp. 5104-5113.
198. Park Y.I., Kim J.H., Lee K.T., et. al.: Nonblinking and nonbleaching upconverting nanoparticles as an optical imaging nanoprobe and T1 magnetic resonance imaging contrast agent. Adv Mater 2009;
199. Yang J., Deng Y., Wu Q., et. al.: Mesoporous silica encapsulating upconversion luminescence rare-earth fluoride nanorods for secondary excitation. Langmuir 2010; 26: pp. 8850-8856.
200. Zhou J., Sun Y., Du X., et. al.: Dual-modality in vivo imaging using rare-earth nanocrystals with near-infrared to near-infrared (NIR-to-NIR) upconversion luminescence and magnetic resonance properties. Biomaterials 2010; 31: pp. 3287-3295.
201. Zijlmans H.J., Bonnet J., Burton J., et. al.: Detection of cell and tissue surface antigens using up-converting phosphors: a new reporter technology. Anal Biochem 1999; 267: pp. 30-36.
202. Lim S.F., Riehn R., Ryu W.S., et. al.: In vivo and scanning electron microscopy imaging of up-converting nanophosphors in Caenorhabditis elegans. Nano Lett 2006; 6: pp. 169-174.
203. Chen G., Shen J., Ohulchanskyy T.Y., et. al.: (α-NaYbF4:Tm(3+))/CaF2 core/shell nanoparticles with efficient near-infrared to near-infrared upconversion for high-contrast deep tissue bioimaging. ACS Nano 2012; 6: pp. 8280-8287.
204. Wu P.C., Su C.H., Cheng F.Y., et. al.: Modularly assembled magnetite nanoparticles enhance in vivo targeting for magnetic resonance cancer imaging. Bioconjug Chem 2008; 19: pp. 1972-1979.
205. Hammer B.E., Christensen N.L., Heil B.G.: Use of a magnetic field to increase the spatial resolution of positron emission tomography. Med Phys 1994; 21: pp. 1917-1920.
206. Xu G., Rajian J.R., Girish G., et. al.: Photoacoustic and ultrasound dual-modality imaging of human peripheral joints. J Biomed Opt 2013; 18: pp. 10502.
207. Rajian J.R., Girish G., Wang X.: Photoacoustic tomography to identify inflammatory arthritis. J Biomed Opt 2012; 17: 96013–96011
208. Jennings L.E., Long N.J.: ‘Two is better than one’–probes for dual-modality molecular imaging. Chem Commun (Camb) 2009; pp. 3511-3524.
209. Wang Y.H., Liao A.H., Chen J.H., et. al.: Photoacoustic/ultrasound dual-modality contrast agent and its application to thermotherapy. J Biomed Opt 2012; 17: pp. 045001.
210. Acharya A.: Luminescent magnetic quantum dots for in vitro/in vivo imaging and applications in therapeutics. J Nanosci Nanotechnol 2013; 13: pp. 3753-3768.
211. Chou S.W., Shau Y.H., Wu P.C., et. al.: In vitro and in vivo studies of FePt nanoparticles for dual modal CT/MRI molecular imaging. J Am Chem Soc 2010; 132: pp. 13270-13278.
212. Madru R., Kjellman P., Olsson F., et. al.: 99mTc-labeled superparamagnetic iron oxide nanoparticles for multimodality SPECT/MRI of sentinel lymph nodes. J Nucl Med 2012; 53: pp. 459-463.
213. Lai S.-M., Tsai T.-Y., Hsu C.-Y., et. al.: Bifunctional silica-coated superparamagnetic FePt nanoparticles for fluorescence/MR dual imaging. J Nanomaterials 2012; 2012: pp. 7.
214. Agarwal A., Shao X., Rajian J.R., et. al.: Dual-mode imaging with radiolabeled gold nanorods. J Biomed Opt 2011; 16: pp. 051307.
215. Shao X., Zhang H., Rajian J.R., et. al.: 125I-labeled gold nanorods for targeted imaging of inflammation. ACS Nano 2011; 5: pp. 8967-8973.
216. Ventura M., Sun Y., Rusu V., et. al.: Dual contrast agent for computed tomography and magnetic resonance hard tissue imaging. Tissue Eng Part C Methods 2013; 19: pp. 405-416.
217. Bouchard L.S., Anwar M.S., Liu G.L., et. al.: Picomolar sensitivity MRI and photoacoustic imaging of cobalt nanoparticles. Proc Natl Acad Sci U S A 2009; 106: pp. 4085-4089.
218. Minchin R.F., Martin D.J.: Nanoparticles for molecular imaging–an overview. Endocrinology 2010; 151: pp. 474-481.
219. Rahmim A., Zaidi H.: PET versus SPECT: strengths, limitations and challenges. Nucl Med Commun 2008; 29: pp. 193-207.
220. Speck U.: Contrast agents: X-ray contrast agents and molecular imaging – a contradiction?.Semmler W.Schwaiger M.Molecular imaging I.2008.Springer-VerlagBerlin Heidelberg:pp. 167-175.
221. Jasanoff A.: MRI contrast agents for functional molecular imaging of brain activity. Curr Opin Neurobiol 2007; 17: pp. 593-600.
222. Chen I.Y., Wu J.C.: Cardiovascular molecular imaging: focus on clinical translation. Circulation 2011; 123: pp. 425-443.
223. Nunn A.D., Linder K.E., Tweedle M.F.: Can receptors be imaged with MRI agents?. Q J Nucl Med 1997; 41: pp. 155-162.
224. Fumita M., Innis R.B.: In vivo molecular imaging: ligand development and research applications.Davis K.L.Charney D.Coyle J.T. et. al.Neuropsychopharmacology: the fifth generation of progress.2002.American College of Neuropsychopharmacologypp. 411.
225. Weissleder R., Ntziachristos V.: Shedding light onto live molecular targets. Nat Med 2003; 9: pp. 123-128.
226. Adams J.E.: Advances in bone imaging for osteoporosis. Nat Rev Endocrinol 2013; 9: pp. 28-42.
227. Baroncelli G.I.: Quantitative ultrasound methods to assess bone mineral status in children: technical characteristics, performance, and clinical application. Pediatr Res 2008; 63: pp. 220-228.
228. Chappard D., Basle M.F., Legrand E., et. al.: New laboratory tools in the assessment of bone quality. Osteoporos Int 2011; 22: pp. 2225-2240.
229. Morris M.D., Mandair G.S.: Raman assessment of bone quality. Clin Orthop Relat Res 2011; 469: pp. 2160-2169.
230. Donnelly E.: Methods for assessing bone quality: a review. Clin Orthop Relat Res 2011; 469: pp. 2128-2138.
231. Boisselier E., Astruc D.: Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem Soc Rev 2009; 38: pp. 1759-1782.
232. Thorek D.L., Chen A.K., Czupryna J., et. al.: Superparamagnetic iron oxide nanoparticle probes for molecular imaging. Ann Biomed Eng 2006; 34: pp. 23-38.