Rationale and Objectives
This study was designed to test the feasibility of contrast pulse sequencing imaging for contrast-enhanced grayscale ultrasound in assessing the effects of antiangiogenic therapy.
Materials and Methods
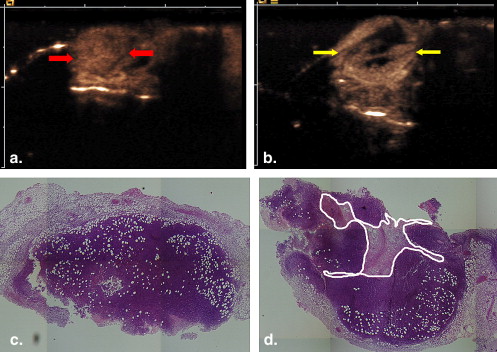
Mice with subcutaneously implanted H22 mouse hepatoma were treated with thalidomide or placebo by oral gavage over 7 days, starting at 24 hours after implantation. Contrast pulse sequencing ultrasound imaging was performed on day 8 to evaluate maximal cross-sectional area and nonenhanced area. Immediately after imaging, mice were euthanized, and tumor tissue was removed for fixation in a 10% formalin solution. The section equivalent to the ultrasound imaging plane was stained with hematoxylin and eosin to allow for the assessment of the largest cross-sectional area and necrotic area.
Results
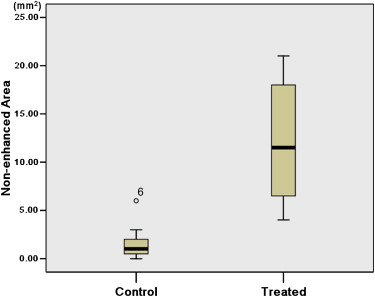
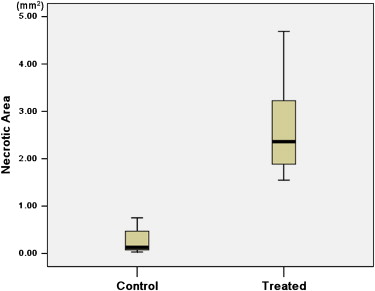
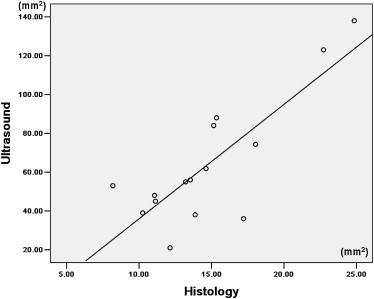
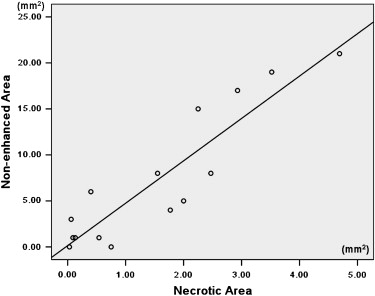
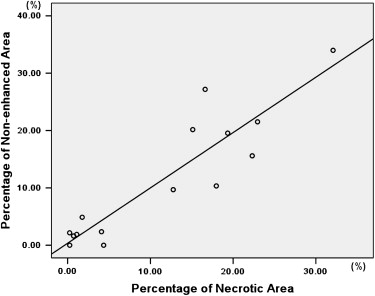
There was no significant difference in tumor volume between the two groups. The difference of largest cross-sectional area determined by the two methods was not significant between control and treated tumors ( P > .05). The nonenhanced area and its percentage evaluated by ultrasound were significantly larger in treated tumors than in control tumors ( P < .05). The necrotic area and its percentage estimated by pathology slice were also significantly larger in treated tumors than in control tumors ( P < .05). The largest cross-sectional area determined by the two methods was well correlated ( r = 0.815, P < .001). There was good correlation between the nonenhanced area on ultrasound and the necrotic area on pathology slides ( r = 0.909, P < .001). The percentage of nonenhanced area was well correlated with the percentage of necrotic area ( r = 0.910, P < .001).
Conclusion
Contrast-enhanced grayscale ultrasound with contrast pulse sequencing imaging provides a tool for early monitoring of antiangiogenic treatment of tumors, before apparent change in tumor size.
One of the most important recent developments in the field of oncology is the introduction of vascular targeting agents into clinical medicine. Contrary to cytotoxic agents, angiogenic inhibitors do not act directly on tumor cells but on endothelial cells, which very frequently induce lesion necrosis with no change in the volume of the initial tumor . The morphologic criteria normally used (World Health Organization or Response Evaluation Criteria in Solid Tumors) may not be the most appropriate endpoints for evaluating antiangiogenic agents . Additionally, these treatments have important side effects and high prices. As a consequence, animal and clinical trials will urgently require the development of reliable and noninvasive methods for the early evaluation of therapeutic efficacy.
Currently, histologic determination of the mean intratumoral microvessel density is the most commonly used method for assessing angiogenesis , but it is also the most invasive procedure. An additional limitation of this approach is that it does not provide information on effective perfusion through a vessel. Moreover, it reflects only a single point in time and cannot be used as a way of monitoring in vivo changes induced by antiangiogenic therapy. Importantly, although microvessel density can be used to predict disease progression, it has no value in monitoring or predicting response to antiangiogenic therapy .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Animal Model
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Ultrasound Imaging Protocol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Histologic Analysis
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
B-mode Imaging
Get Radiology Tree app to read full this article<
Contrast-Enhanced Grayscale Ultrasound
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Histology
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Comparison of Contrast-Enhanced Grayscale Ultrasound and Histology
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Hlatky L., Hahnfeldt P., Folkman J.: Clinical application of antiangiogenic therapy: microvessel density, what it does and doesn’t tell us. J Natl Cancer Inst 2002; 94: pp. 883-893.
2. Padhani A.R., Neeman M.: Challenges for imaging angiogenesis. Br J Radiol 2001; 74: pp. 886-890.
3. Lassau M., Chebil M., Chami L., et. al.: A new functional imaging technique for the early functional evaluation of antiangiogenic treatment: dynamic contrast-enhanced ultrasonography (DCE-US). Targ Oncol 2008; 3: pp. 111-117.
4. Kerbel R., Folkman J.: Clinical translation of angiogenesis inhibitors. Nat Rev Cancer 2002; 2: pp. 727-739.
5. Tatum J.L., Hoffman J.M.: Role of imaging in clinical trials of antiangiogenesis therapy in oncology. Acad Radiol 2000; 7: pp. 798-799.
6. Miller J.C., Pien H.H., Sahani D., et. al.: Imaging angiogenesis: applications and potential for drug development. J Nat Cancer lnst 2005; 97: pp. 172-187.
7. Galbraith S.M.: Antivascular cancer treatments: imaging biomarkers in pharmaceutical drug development. Br J Radiol 2003; 76: pp. s83-s86.
8. Provenzale J.M.: Imaging of angiogenesis: clinical techniques and novel imaging methods. AJR Am J Roentgenol 2007; 188: pp. 11-23.
9. Gee M.S., Saunders H.M., Lee J.C., et. al.: Doppler ultrasound imaging detects changes in tumor perfusion during antivascular therapy associated with vascular anatomic alterations. Cancer Res 2001; 61: pp. 2974-2982.
10. Donnelly E.F., Geng L., Wojcicki W.E., et. al.: Quantified power Doppler US of tumor blood flow correlates with microscopic quantification of tumor blood vessels. Radiology 2001; 219: pp. 166-170.
11. Delorme S., Haberkorn U., Kinscherf R., et. al.: Changes of tumor vascularity during gene therapy monitored with color Doppler US. Ultrasound Med Biol 2001; 27: pp. 1595-1603.
12. Iordanescu I., Becker C., Zetter B., et. al.: Tumor vascularity evaluation in a murine model with contrast-enhanced color Doppler US-effect of angiogenesis inhibitors. Radiology 2002; 222: pp. 460-467.
13. Delorme S., Peschke P., Zuna I., et. al.: Imaging the smallest tumor vessels using color Doppler ultrasound in an experiment [article in German]. Radiologe 2001; 41: pp. 168-172.
14. Phillips P., Gardner E.: Contrast-agent detection and quantification. Eur Radiol 2004; 14: pp. 4-10.
15. Bleuzen A., Tranquart F.: Incidental liver lesions: diagnostic value of cadence contrast pulse sequencing (CPS) and SonoVue. Eur Radiol 2004; 14: pp. 53-62.
16. Wink M.H., de la Rosette J.J., Laguna P., et. al.: Ultrasonography of renal masses using contrast pulse sequence imaging: a pilot study. J Endourol 2007; 21: pp. 466-472.
17. Rissanen T.T., Korpisalo P., Karvinen H., et. al.: High-resolution ultrasound perfusion imaging of therapeutic angiogenesis. JACC: Cardiovasc Imaging 2008; 1: pp. 83-91.
18. Zhu B.D., Yuan S.J., Zhao Q.C., et. al.: Antitumor effect of Gefitinib, an epidermal growth factor receptor tyrosine kinase inhibitor, combined with cytotoxic agent on murine hepatocellular carcinoma. World J Gastroenterol 2005; 11: pp. 1382-1386.
19. Kumarv V., Abbas A.K., Fausto , Aater J.C.: Robbins and Cotran pathologic basis of disease.8th ed.2007.SaundersPhiladelphia
20. Folkman J.: Tumor angiogenesis: therapeutic implications. N Engl J Med 1971; 285: pp. 1182-1186.
21. D’Amato R.J., Loughnan M.S., Flynn E., et. al.: Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci U S A 1994; 91: pp. 4082-4085.
22. Kenyon B.M., Browne F., D’Amato R.J.: Effects of thalidomide and related metabolites in a mouse corneal model of neovascularization. Exp Eye Res 1997; 64: pp. 971-978.
23. Teo S.K.: Properties of thalidomide and its analogues: implications for anticancer therapy. AAPS J 2005; 7: pp. E14-E19.
24. Rissanen T.T., Korpisalo P., Markkanen J.E., et. al.: Blood flow remodels growing vasculature during vascular endothelial growth factor gene therapy and determines between capillary arterialization and sprouting angiogenesis. Circulation 2005; 112: pp. 3937-3946.
25. Cosgrove D.: Angiogenesis imaging ultrasound. Br J Radiol 2003; 76: pp. S43-S49.
26. Pollard R.E., Broumas A.R., Wisner E.R., et. al.: Quantitative contrast enhanced ultrasound and CT assessment of tumor response to antiangiogenic therapy in rats. Ultrasound Med Biol 2007; 33: pp. 235-245.
27. Mendel D.B., Laird A.D., Xin X., et. al.: In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res 2003; 9: pp. 327-337.
28. Broumas A.R., Pollard R.E., Bloch S.H., et. al.: Contrast-enhanced computed tomography and ultrasound for the evaluation of tumor blood flow. Invest Radiol 2005; 40: pp. 134-147.
29. De Giorgi U., Aliberti C., Benea G., et. al.: Effect of angiosonography to monitor response during imatinib treatment in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res 2005; 11: pp. 6171-6176.
30. Lassau N., Lamuraglia M., Chami L., et. al.: Gastrointestinal stromal tumors treated with imatinib: monitoring response with contrast-enhanced sonography. AJR Am J Roentgenol 2006; 187: pp. 1267-1273.
31. Lamuraglia M., Le Cesne A., Chami L., et. al.: Dynamic contrast-enhanced Doppler ultrasound (DCE-US) is a useful radiological assessment to early predict the outcome of patients with gastrointestinal stromal tumors (GIST) treated with imatinib (IM) [abstract]. J Clin Oncol 2006; 24: pp. 9539.
32. Du G.J., Lin H.H., Xu Q.T., et. al.: Thalidomide inhibits growth of tumors through COX-2 degradation independent of antiangiogenesis. Vascul Pharmacol 2005; 43: pp. 112-119.
33. Kotoh T., Dhar D.K., Masunaga R., et. al.: Antiangiogenic therapy of human esophageal cancers with thalidomide in nude mice. Surgery 1999; 125: pp. 536-544.
34. Benjamin R.S., Choi H., Macapinlac H.A., et. al.: We should desist using RECIST, at least in GIST. J Clin Oncol 2007; 25: pp. 1760-1764.
35. Choi H., Charnsangavej C., Faria S.C., et. al.: Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 2007; 25: pp. 1753-1759.