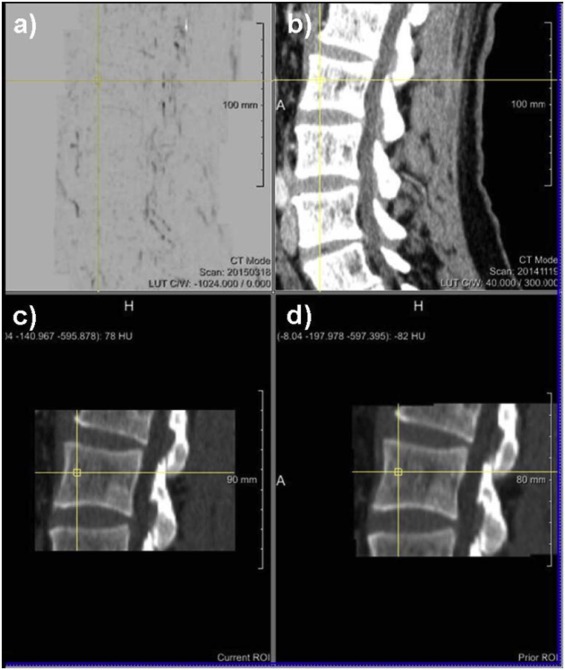
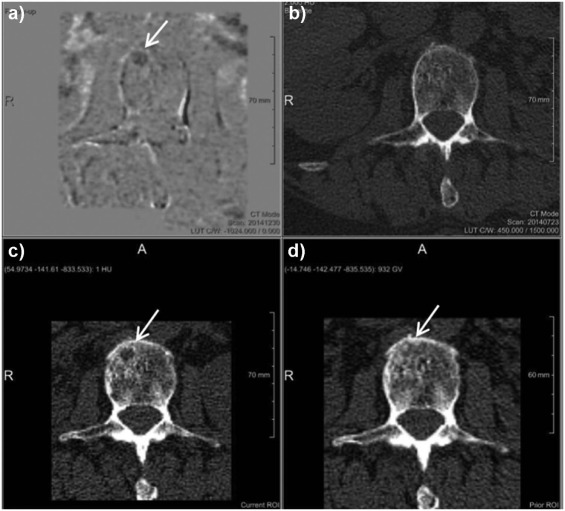
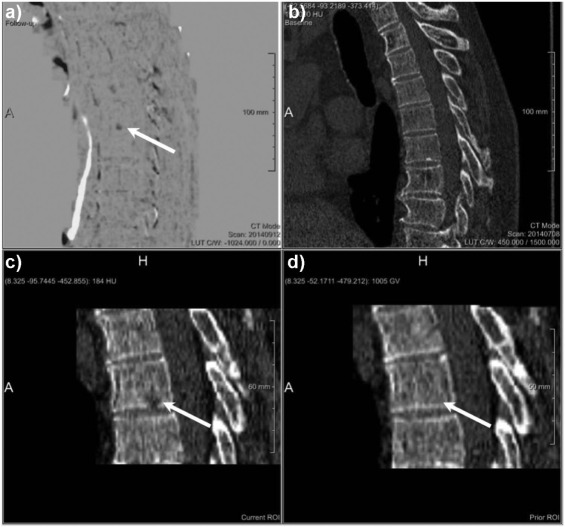
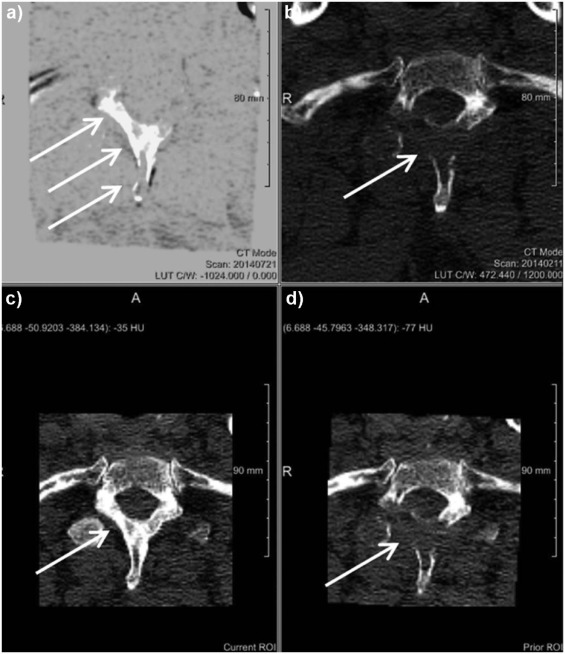
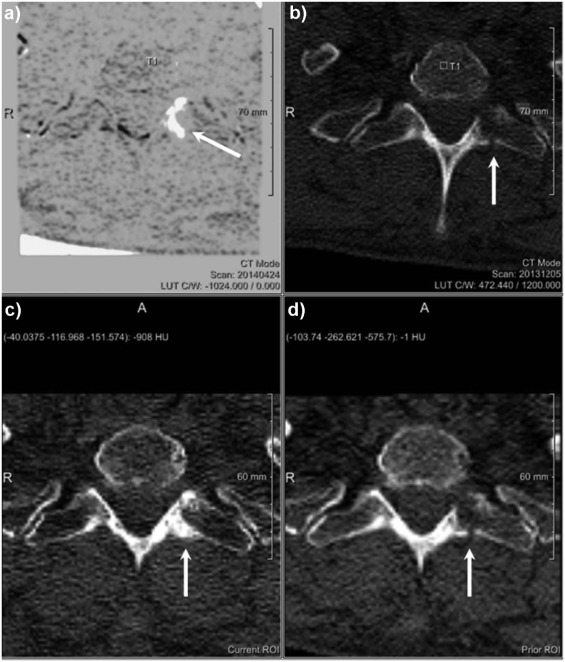
Rationale and Objectives
The study aimed to assess the diagnostic benefit of a novel computed tomography (CT) post-processing software generating subtraction maps of longitudinal non-enhanced CT examinations for monitoring the course of myeloma bone disease in the spine.
Materials and Methods
The local institutional review board approved the retrospective data evaluation. Included were 82 consecutive myeloma patients (46 male; mean age, 65.08 ± 9.76) who underwent 188 repeated whole-body reduced-dose Multislice Detector Computed Tomography (MDCT) at our institution between December 2013 and January 2016. Lytic bone lesions were categorized as new or enlarging versus stable. Bone subtraction maps were read in combination with corresponding 1-mm source images comparing results to those of standard image reading of 5-mm axial and 2-mm multiplanar reformat reconstructions (MPR) scans and hematologic markers, and classified as either progressive disease (PD) or stable disease (SD or remission). The standard of reference was 1-mm axial CT image reading + hematologic response both confirmed at follow-up. For statistical purposes, we subgrouped the hematologic response categories similarly to those applied for CT imaging (progression vs stable/response).
Results
According to the standard of reference, 16 patients experienced PD and 66 SD at follow-up. Th sensitivity, specificity, and accuracy for axial 5 mm + 2 mm MPR image versus bone subtraction maps in a “lesion-by-lesion” reading were 97.6%, 92.3%, and 97.2% versus 97.8%, 96.7%, and 97.7%, respectively. The use of bone subtraction maps resulted in a change of response classification in 9.7% of the patients ( n = 8) versus 5 mm + 2 mm MPR image reading from SD to PD. Bone sclerosis lesions were detected in 52 out of 82 patients (63.4%). The reading time was significantly lower with the software bone subtraction compared to standard reading ( P < 0.01) and 1-mm image reading ( P < 0.001).
Conclusion
Accuracy of bone subtraction maps reading for monitoring multiple myeloma is slightly increased over that of conventional axial + MPR image reading and significantly speeds up the reading time.
Introduction
Multiple myeloma is a malignant hematologic disease of mature B cells with an often guarded prognosis. The disease is characterized by an overproduction of monoclonal immunoglobulins or light-chain molecules. One major feature is myeloma bone disease (MBD), which manifests as focal or diffuse generalized bone destruction. A misbalance of osteoblasts and osteoclasts function results in an overall increased osteoclast activity and subsequent bone resorption as well as disabled osteoblasts . As a consequence, these osteolyses do not heal in the majority of patients .On first diagnosis, almost 80% of the patients with multiple myeloma present with lytic bone lesions , which due to an often high number and small size can pose a challenge for radiological detection. In particular at follow-up and during treatment, the determination of MBD progression can be time-consuming and potentially inaccurate.
In the last decades, post-processing techniques, such as multiplanar reconstructions and the use of thin-slice reconstructions, significantly improved lesion detection. In multiple myeloma, typically the axial skeleton is involved first as the disease predominates in regions of predominant red marrow. Typically, myeloma first involves the bone marrow, which follows bone destruction as the tumor nests enlarge and the dense trabecular architecture of the cancellous bone limits their expansion. Before bone destruction becomes evident on radiographs or even computed tomography (CT), magnetic resonance imaging proves superior for the detection of infiltrative medullary lesions. Nevertheless, CT can provide helpful information during or after therapy, specifically for the detection of residual abnormalities, which are concerning for localized relapse . Although many myeloma patients can be monitored reliably with hematologic parameters such as M-gradient, the non-secretory group of patients does not secrete paraproteins and is therefore difficult to monitor. In this group of patients, imaging plays an important diagnostic role. Moreover, progression of MBD is often not affected by chemotherapy even in responding patients . For this reason, the detection of changes occurring in the axial skeleton at follow-up is critical for accurate assessment of progressive disease. CT is the most accurate technique for imaging assessment of bone. Several new techniques have been tested in the last years with the goal to increase detection of bone changes during disease surveillance. Some researchers applied automatic detection of bone metastases in the thoracolumbar spine and visualization of bone metastases with maximum intensity projection and surface-shaded display techniques . In a small animal myeloma model, Evans et al. tested a fully automatic image analysis method created to detect osteolyses in the cortical bone of the tibia .
Get Radiology Tree app to read full this article<
Materials and Methods
Get Radiology Tree app to read full this article<
Subjects
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Patients’ Demographics
No. of Patients_n_ = 82 100% Age, years, mean ± SD 65.08 ± 9.76 – Range 38–81 – Age at initial diagnosis, mean ± SD 61.21 ± 10.36 – Male 46 56.09 Female 36 43.91 Durie and Salmon I 23 28.0 II 11 13.4 III 48 58.6 ISS I 42 51.2 II 13 15.8 III 27 33.0
ISS, International Staging System; SD, standard deviation.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Maintenance Therapy Regimens at Time Point of Follow-up Examination
n % Overall_82_100 Primary therapy 34 41.4 Lenalidomide and dexamethasone_10_12.1 VCD_7_8.9 VPM_4_4.8 Pomalidomide and dexamethasone_4_4.8 Bortezomib and dexamethasone_3_3.6 Revlimid and dexamethasone_2_2.4 Melphalan_2_2.4 Cyclophosphamide mono_1_1.2 Daratumumab + bortezomib + dexamethasone_1_1.2 Remission control/maintenance therapy 18 19.3 Lenalidomide 8 9.7 Pomalidomide/Dexamethasone 4 4.8 Thalidomide 2 2.4 Bortezomib 1 1.2 Carfilzomib/Dexamethasone 1 1.2 No therapy 30 39.3
VCD, bortezomib + cyclophosphamide + dexamethasone; VPM, bortezomib + melphalan + dexamethasone.
Get Radiology Tree app to read full this article<
CT Image Protocol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
CT Image Post-processing
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Image Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Standard of Reference
Get Radiology Tree app to read full this article<
Classification of the Disease Course at Follow-up Based on CT Imaging
Get Radiology Tree app to read full this article<
Hematologic Classification of the Disease Course at Follow-up
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Hematologic Classification of the Disease Course
Get Radiology Tree app to read full this article<
Table 3
Overview of Response Categories
Response Categories Hematologic-based Classification Bone Subtraction CT-based Classification (5-mm axial + 2-mm MPR)n (%)n (%)n (%) Progressive disease (PD) 16 (22.3%) 21 (25.6%) 15 (18.5%) Stable disease (SD) 66 (77.7%) 61 (74.3%) 67 (81.5%)
CT, computed tomography; MPR, multiplanar reformat reconstructions.
Get Radiology Tree app to read full this article<
Classification of the Disease Course Based on CT Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 4
Overview of Longitudinal Changes in Bone Attenuation in Patients Developing Bone Sclerosis
Progress (Patients, n = 21) Stable (Patients, n = 61) Baseline Examination Follow-up Examination_P_ Baseline Examination Follow-up Examination_P_ Sclerotic lesions Mean HU 187.85 ± 229.58 432.76 ± 173.93 0.001 \* 250.42 ± 242.87 497.98 ± 211.39 0.001 \* Min HU −106.23 ± 240.93 13.33 ± 226.89 0.001 \* −62.58 ± 221.13 79.73 ± 239.42 0.001 \* Max HU 508.23 ± 284.33 845.54 ± 254.90 0.001 \* 648.86 ± 346.49 926.52 ± 318.31 0.001 \*
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 5
Lesion Based Sensitivity, Specificity, Accuracy, Positive Predictive Value, and Negative Predictive Value
Axial 5-mm + 2-mm MPR (%) Axial 1 mm (%) Bone Subtraction (%) Sensitivity 97.6 97.7 97.8 Specificity 92.3 95.2 96.7 Accuracy 97.2 97.4 97.7 Positive predictive value 99.4 99.4 99.4 Negative predictive value 75.0 83.3 88.2
MPR, multiplanar reformat reconstructions.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Reading Time
Get Radiology Tree app to read full this article<
Interobserver Agreement
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Roodman G.D.: Osteoblast function in myeloma. Bone 2011; 48: pp. 135-140.
2. Roodman G.D.: Skeletal imaging and management of bone disease. Hematology Am Soc Hematol Educ Program 2008; 2008: pp. 313-319.
3. Kyle R.A., Gertz M.A., Witzig T.E., et. al.: Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc 2003; 78: pp. 21-33.
4. Lecouvet F.E., De Nayer P., Garbar C., et. al.: Treated plasma cell lesions of bone with MRI signs of response to treatment: unexpected pathological findings. Skeletal Radiol 1998; 27: pp. 692-695.
5. Gleeson T.G., Moriarty J., Shortt C.P., et. al.: Accuracy of whole-body low-dose multidetector CT (WBLDCT) versus skeletal survey in the detection of myelomatous lesions, and correlation of disease distribution with whole-body MRI (WBMRI). Skeletal Radiol 2009; 38: pp. 225-236.
6. Raje N., Anderson K.C.: Multiple myeloma. Curr Treat Options Oncol 2000; 1: pp. 73-82.
7. O’Connor S.D., Yao J., Summers R.M.: Lytic metastases in thoracolumbar spine: computer-aided detection at CT–preliminary study. Radiology 2007; 242: pp. 811-816.
8. Herman G.T., Coin C.G.: The use of three-dimensional computer display in the study of disk disease. J Comput Assist Tomogr 1980; 4: pp. 564-567.
9. Hohne K.H., Bernstein R.: Correction to “shading 3D-images from CT using gray-level gradients”. IEEE Trans Med Imaging 1986; 5: pp. 165.
10. Evans H.R., Karmakharm T., Lawson M.A., et. al.: Osteolytica: an automated image analysis software package that rapidly measures cancer-induced osteolytic lesions in in vivo models with greater reproducibility compared to other commonly used methods. Bone 2016; 83: pp. 9-16.
11. Durie B.G., Kyle R.A., Belch A., et. al.: Myeloma management guidelines: a consensus report from the Scientific Advisors of the International Myeloma Foundation. Hematol J 2003; 4: pp. 379-398.
12. Palumbo A., Avet-Loiseau H., Oliva S., et. al.: Revised International Staging System for multiple myeloma: a report from International Myeloma Working Group. J Clin Oncol 2015; 33: pp. 2863-2869.
13. Horger M., Pereira P., Claussen C.D., et. al.: Hyperattenuating bone marrow abnormalities in myeloma patients using whole-body non-enhanced low-dose MDCT: correlation with haematological parameters. Br J Radiol 2008; 81: pp. 386-396.
14. Homann G., Mustafa D.F., Ditt H., et. al.: Improved detection of bone metastases from lung cancer in the thoracic cage using 5- and 1-mm axial images versus a new CT software generating rib unfolding images: comparison with standard (1)(8)F-FDG-PET/CT. Acad Radiol 2015; 22: pp. 505-512.
15. Viola P., Jones M.: Robust real-time face detection. Int J Comput Vis 2004; 57: pp. 137-154.
16. Viola P.W.W.: Alignment by maximization of mutual information. Int J Comput Vis 1997; 24: pp. 137-154.
17. International Myeloma Working Group: Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 2003; 121: pp. 749-757.
18. Lecouvet F.E., Vande Berg B.C., Michaux L., et. al.: Stage III multiple myeloma: clinical and prognostic value of spinal bone marrow MR imaging. Radiology 1998; 209: pp. 653-660.
19. Song I.C., Kim J.N., Choi Y.S., et. al.: Diagnostic and prognostic implications of spine magnetic resonance imaging at diagnosis in patients with multiple myeloma. Cancer Res Treat 2015; 47: pp. 465-472.
20. Horger M., Kanz L., Denecke B., et. al.: The benefit of using whole-body, low-dose, nonenhanced, multidetector computed tomography for follow-up and therapy response monitoring in patients with multiple myeloma. Cancer 2007; 109: pp. 1617-1626.
21. Schulze M., Weisel K., Grandjean C., et. al.: Increasing bone sclerosis during bortezomib therapy in multiple myeloma patients: results of a reduced-dose whole-body MDCT study. AJR Am J Roentgenol 2014; 202: pp. 170-179.
22. Mohty M., Malard F., Mohty B., et. al.: The effects of bortezomib on bone disease in patients with multiple myeloma. Cancer 2014; 120: pp. 618-623.
23. Letourneau D., Kaus M., Wong R., et. al.: Semiautomatic vertebrae visualization, detection, and identification for online palliative radiotherapy of bone metastases of the spine. Med Phys 2008; 35: pp. 367-376.
24. Homann G., Weisel K., Mustafa D.F., et. al.: Improvement of diagnostic confidence for detection of multiple myeloma involvement of the ribs by a new CT software generating rib unfolded images: comparison with 5- and 1-mm axial images. Skeletal Radiol 2015; 44: pp. 971-979.