Rationale and Objectives
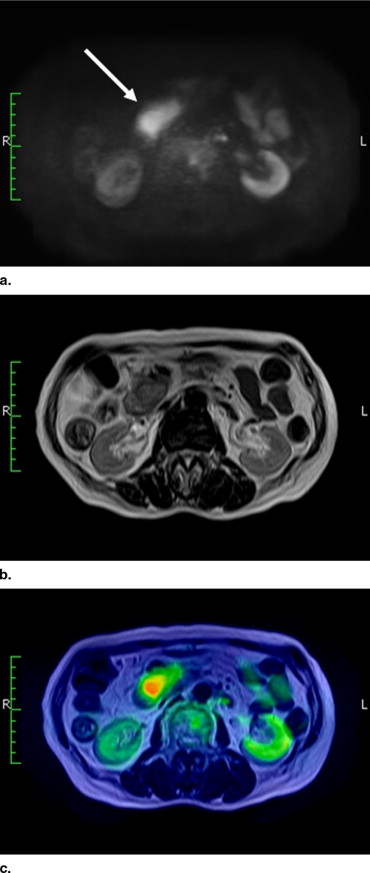
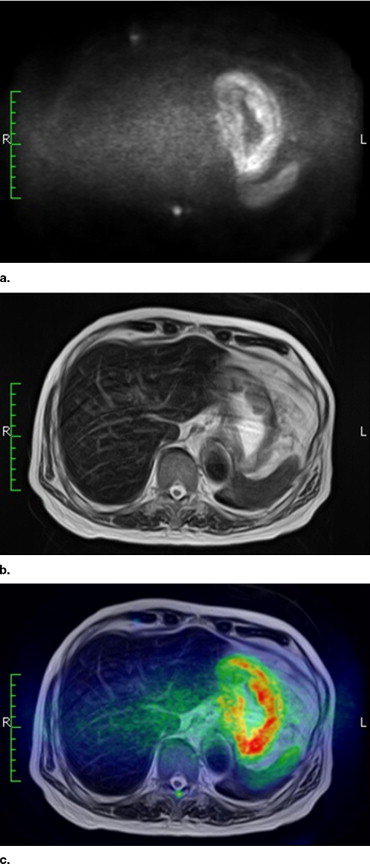
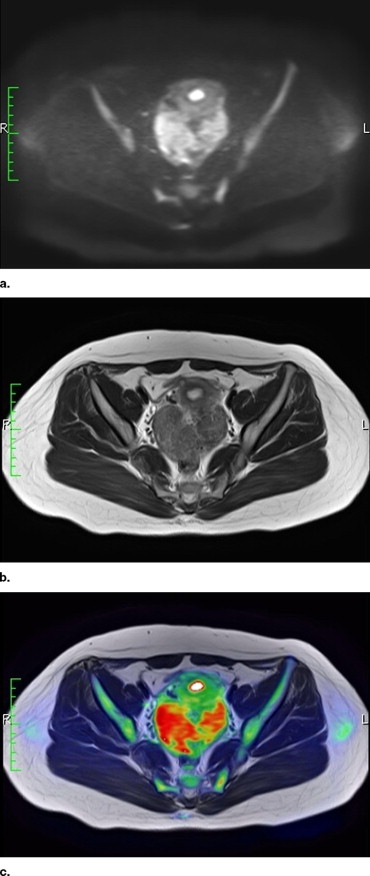
To evaluate the potential usefulness of high b -value body diffusion-weighted images (DWIs) as a screening tool in the depiction of abdominal malignant tumors.
Materials and Methods
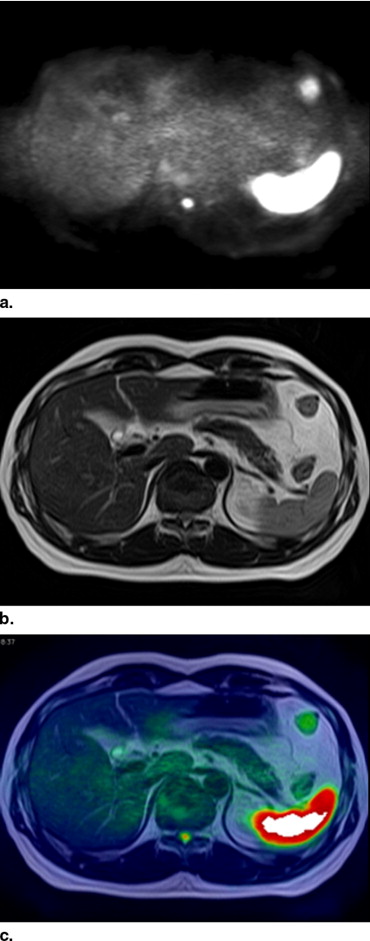
We selected 110 abdominal magnetic resonance examinations (1.5 T; 60 men; age range, 25–90 years) with and without malignant tumors ( n = 37 and n = 73, respectively). Axial DWIs were obtained by single-shot spin-echo (SE) type echo planar imaging (EPI) sequence with inversion pulse (repetition time, 6,800 msec; echo time, 100 msec; T1, 150 msec; b value, 1,000 sec/mm 2 ) without breath-holding. Two radiologists independently interpreted the DWIs, T2-weighted images (T2-WI), all three types of images including DWIs, T2-WIs, and fusion images at the same time (DWIs + T2-WIs + fusion) with 7–14 days’ interval, and the diagnostic confidence for each patient was scored.
Results
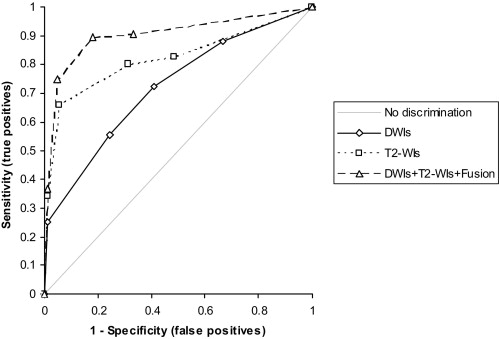
The area under the curve (AUC) of the composite receiver operating characteristic (ROC) curve of DWIs + T2-WIs + fusion (0.904) was significantly higher than those of DWIs (0.720; P < .001) and T2-WIs (0.822; P < .05). Both sensitivity and specificity were higher in DWIs + T2-WIs + fusion (89.5% and 81.9%, respectively) compared with those of DWIs (72.4% and 59.0%; P < .01 and P < .001, respectively).
Conclusions
Abdominal high b -value DWIs have a high sensitivity and specificity for malignant tumors when T2-WIs are referred and image fusion technique is employed, suggesting that it may potentially be a new screening tool.
Recent advances in magnetic resonance (MR) technology allow us to obtain diffusion-weighted images (DWIs) with high b -factor not only in the brain but in the body, and the image quality of body DWIs has improved even under a free breathing using a short TI inversion recovery echo planar imaging sequence that allows potent fat suppression ( ).
There have been several reports that showed the high ability of body DWIs to detect various malignant tumors ( ). For instance, Ichikawa et al ( ) reported that the high b -value DWIs allow detection of colorectal adenocarcinoma with a high sensitivity and specificity, and Nasu et al ( ) reported that combined reading of DWIs, and T1- and T2-weighted images had the higher accuracy in the detection of hepatic metastases than did reading of superparamagnetic iron oxide particles (SPIO)-enhanced MR images. These reports let us suggest the potential capability of body DWIs as a screening tool for malignant tumors.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Patients
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
MR Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
CT Imaging
Get Radiology Tree app to read full this article<
Image Fusion Between DWIs and T2-WIs
Get Radiology Tree app to read full this article<
Image Analyses
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Table 1
Results of Receiver Operating Characteristic Analyses of Two Readers
Reader 1 Reader 2 Composite AUC Sens. Spec. AUC Sens. Spec. AUC Sens. Spec. DWIs 0.667 73.7% 45.8% 0.763 71.1% 72.2% 0.720 72.4% 59.0% *2 *1 *1 T2-WIs 0.856 *2 89.5% *1 51.4% *3 0.810 71.1% 86.1% *1 0.822 *3 80.3% *2 68.8% *3 *2 *1 *2 DWIs+T2-WIs+Fusion 0.931 94.7% 76.4% 0.883 84.2% 87.5% 0.904 89.5% 81.9%
AUC = area under curve; Sens. = sensitivity; Spec. = specificity.
*1 P < 0.05; *2 P < 0.01; *3 P < 0.001; Bonferroni’s method was applied.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Takahara T., Imai Y., Yasumasa T., et. al.: Diffusion weighted whole body imaging with background body signal suppression (DWIBS): technical improvement using free breathing, STIR and high resolution 3D display. Radiat Med 2004; 22: pp. 275-282.
2. Ichikawa T., Erturk S.M., Motosugi U., et. al.: High-b-value diffusion-weighted MRI in colorectal cancer. AJR Am J Roentgenol 2006; 187: pp. 181-184.
3. Nasu K., Kuroki Y., Nawano S., et. al.: Hepatic metastases: diffusion-weighted sensitivity-encoding versus SPIO-enhanced MR imaging. Radiology 2006; 239: pp. 122-130.
4. Thoeny H.C., De Keyzer F., Chen F., et. al.: Diffusion-weighted MR imaging in monitoring the effect of a vascular targeting agent on rhabdomyosarcoma in rats. Radiology 2005; 234: pp. 756-764.
5. Dzik-Jurasz A., Domenig C., George M., et. al.: Diffusion MRI for prediction of response of rectal cancer to chemoradiation. Lancet 2002; 360: pp. 307-308.
6. Roth Y., Tichler T., Kostenich G., et. al.: High-b-value diffusion-weighted MR imaging for pretreatment prediction and early monitoring of tumor response to therapy in mice. Radiology 2004; 232: pp. 685-692.
7. Ustunomiya D., Shiraishi S., Imuta M., et. al.: Added value of SPECT/CT fusion in assessing suspected bone metastasis: comparison with scintigraphy alone and nonfused scintigraphy and CT. Radiology 2006; 238: pp. 264-271.
8. Furtado C.D., Aguirre D.A., Sirlin C.B., et. al.: Whole-body CT screening: spectrum of findings and recommendations in 1192 patients. Radiology 2005; 237: pp. 385-394.
9. Hillman B.J.: CT screening: who benefits and who pays. Radiology 1993; 228: pp. 26-28.
10. Brant-Zawadzki M.: CT screening: why I do it. AJR Am J Roentgenol 2002; 179: pp. 319-326.
11. Hall F.M.: Callback and follow-up guidelines for whole-body CT screening. Radiology 2006; 241: pp. 627-628.