Rationale and Objectives
Prediction of growth, in particular knowing the possibility of aggressive cancer in small renal masses on active surveillance, remains poorly understood. The study was designed to determine whether serial nephrometry score measurements could predict possibility of aggressive malignancy (grade of cancer) in patients with small renal masses opting for active surveillance initially.
Materials and Methods
One hundred sixteen patients between January 2000 and December 2016 undergoing partial nephrectomy were recruited. Out of these, 97 were analyzed using different nephrometry scoring systems. Measurement of nephrometry scores (Radius of tumors, Exo/Endophytic; Nearness of tumors to the collecting system or sinus; Anterior/posterior; Location in relation to polar lines, Preoperative Aspects and Dimensions Used for Anatomical, Centrality Index) was performed by two researchers. Among the patients opting for partial nephrectomy, 40 were on active surveillance for at least 12 months (mean 32; 12–60 months) before partial nephrectomy. Computed tomography scan images of these patients were retrieved and analyzed including comparison to histopathology.
Results
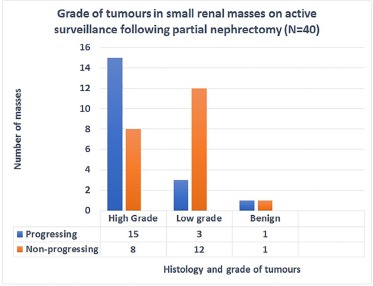
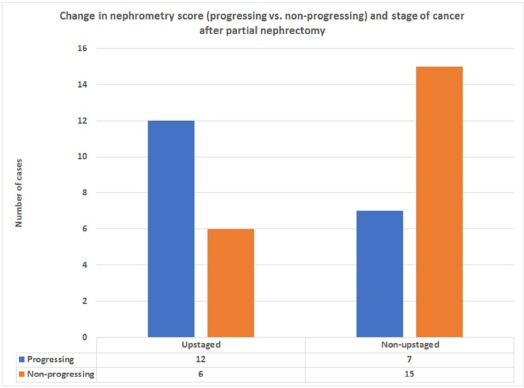
Nephrometry scores measured on serial computed tomography scan images showed a significant correlation between change in score and grade of cancer on multivariate analysis ( P value .001). Addition of multivariate analysis to nomogram based on change in size alone did not improve predictive value of area under the curve significantly.
Conclusions
Change in nephrometry scoring measurements correlates with grade of cancer in small renal masses but falls short of significantly predicting presence of malignancy or grade of cancer on nomogram in patients opting for active surveillance for small renal masses. At present, this approach may be inadequate for decision-making.
Introduction
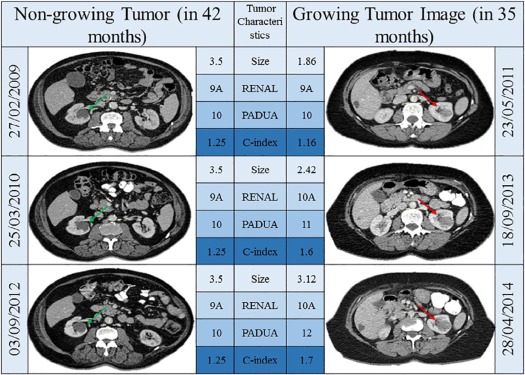
The size of small renal masses (SRMs) correlates with the rate of malignancy on histology following excision . Whether changes in nephrometry scoring, a system that measures a range of parameters including the position of tumors in relation to hilar structures, can predict the rate of malignancy or aggressiveness of cancer is not known. The information related to growth patterns in relation to hilar structures may inform physicians to adopt the best possible management plans, including the prediction of postoperative complications .
There are three main nephrometry scoring systems described for kidney tumors according to pre-intervention computed tomography images and these are RENAL (Radius of tumors, Exo/Endophytic; Nearness of tumors to the collecting system or sinus; Anterior/posterior; Location in relation to polar lines) nephrometry score , PADUA (Preoperative Aspects and Dimensions Used for Anatomical) nephrometry score , and Centrality Index (C-index) . Detailed description of these methods is described in their original reports ; however, these measurement methods are designed to separate complex renal masses from noncomplex ones by measuring their anatomic location within renal parenchyma and their relationship to hilar vessels and renal pelvis. Published literature mainly focuses on using these scoring systems to assess the possibility of complications or technical difficulties that surgeons may encounter in the surgical resection of SRMs. Reports in literature suggest a good interobserver agreement in experienced hands and a predictability value of these scoring systems for postoperative complications following surgical excision and cryotherapy . Little is known about the predictive ability of these measurements to distinguish between benign and malignant histology and the grade of cancers. Few studies exploring the relations between nephrometry scores and histology concentrate on the single measurement of nephrometry scores , and a detailed correlation between changes in nephrometry scores and histology has not been reported.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Patients and Methods
Study Cohort
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 1
Basic Demographics of the Cohort
Characteristics_N_ (SD/%) Numbers 97 Age (y) 59.7(±12.4) Sex Male 61 Female 36 Operative characteristics Kidney Right 43 Left 54 Tumor characteristics Tumor location Anterior 9 Posterior 88 Tumor diameter, mean (range) 3.8 (range 2–4) Tumor pathology Malignant 86 Clear cell carcinoma 67 Papillary cell carcinoma 14 Chromophobe 4 Tubulocystic 1 Benign 11 Grade of cancer Low (Furhman 1 and 2) 46 High (Furhman 3 and 4) 51 RENAL nephrometry (mean) 6.8 (±4.2) PADUA score (mean) 8.3 (±4.8) C-index (mean) 2.1(±1.9)
Get Radiology Tree app to read full this article<
Image Retrieval and Measurements
Get Radiology Tree app to read full this article<
Pathologic and Follow-up Data
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
TABLE 2
Shows Association Between Type of Pathology, Complications, and Nephrometry Scores
Type of Cancer Patients in Total Patients with Complication R.E.N.A.L Score PADUA Score C-Index Score Statistical Significance ( P Value) Clear cell renal carcinoma 67 22 6.82 8.32 2.18 0.12 Papillary renal cell carcinoma 14 1 6.59 7.89 2.29 0.23 Chromophobe 4 0 6.64 7.9 2.32 0.10 Tubulocystic 1 0 8.12 9 1.84Benign 11 1 6.89 8.36 2.17 0.36
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 3
Scoring System Measurements and Histology of Excised SRMs
Type of Nephrometry Scoring Histological Type Following Nephron-sparing Surgery in SMRs Clear Cell Carcinoma, Mean (SD) Papillary Cell Carcinoma, Mean (SD) Chromophobe, Mean (SD) Benign, Mean (SD) Statistical Significance ( P Value) RENAL (Mean and SD) 6.82 (2.15) 6.56 (2.02) 6.64 (2.58) 6.89 (1.68) 0.21 PADUA (Mean and SD) 8.32 (2.11) 7.89 (1.67) 7.9 (3.20) 8.36 (1.87) 0.26 C-Index (Mean and SD) 2.18 (0.99) 2.29 (0.66) 2.33 (1.38) 2.17 (0.57) 0.46
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 4
Logistic Regression Analysis of Presurgical Factors Predicting Grade of Cancer in Excised Tumor (pT1a)
Presurgical Factors Small Renal Masses Removed by Partial Nephrectomy Univariate Analysis Multivariate Analysis Odds Ratio (95% CI)P Value Odds Ratio (95% CI)P Value Age (y) 1.004 (1.000–1.34) .021 1.04 (1.20–2.45) .102 Sex (male) 3.004 (2.01–3.34) .08 2.90 (1.5–4.98) .45 Increase in tumor size 4.02 (2.03–6.00) .001 3.02 (2.75–4.00) .001 Change in nephrometry score 5.00 (3.002–8.04) .0001 4.40 (2.09–5.45) .001 Obesity (BMI >29) 1.34 (0.09–2.03) .09 1.45 (1.12–3.45) .14 Multiple co-morbid conditions (>2) 2.001 (1.05–3.03) .12 2.98 (1.78–3.89) .23
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Ambani S.N., Morgan T.M., Montgomery J.S., et. al.: Predictors of delayed intervention for patients on active surveillance for small renal masses: does renal mass biopsy influence our decision?. Urology 2016; 98: pp. 88-96.
2. Altunrende F., Laydner H., Hernandez A.V., et. al.: Correlation of the RENAL nephrometry score with warm ischemia time after robotic partial nephrectomy. World J Urol 2013; 31: pp. 1165-1169.
3. Bruner B., Breau R.H., Lohse C.M., et. al.: Renal nephrometry score is associated with urine leak after partial nephrectomy. BJU Int 2011; 108: pp. 67-72.
4. Castle S.M., Gorbatiy V., Leveillee R.J.: Robotic partial nephrectomy outcomes at a single institution and experience with R.E.N.A.L. nephrometry score. J Rob Surg 2011; 5: pp. 209-214.
5. Canter D., Kutikov A., Manley B., et. al.: Utility of the R.E.N.A.L. nephrometry scoring system in objectifying treatment decision-making of the enhancing renal mass. Urology 2011; 78: pp. 1089-1094.
6. Kutikov A., Smaldone M.C., Egleston B.L., et. al.: Anatomic features of enhancing renal masses predict malignant and high-grade pathology: a preoperative nomogram using the RENAL Nephrometry score. Eur Urol 2011; 60: pp. 241-248.
7. Ficarra V., Novara G., Secco S., et. al.: Preoperative aspects and dimensions used for an anatomical (PADUA) classification of renal tumours in patients who are candidates for nephron-sparing surgery. Eur Urol 2009; 56: pp. 786-793.
8. Simmons M.N., Ching C.B., Samplaski M.K., et. al.: Kidney tumor location measurement using the C index method. J Urol 2010; 183: pp. 1708-1713.
9. Spaliviero M., Poon B.Y., Aras O., et. al.: Interobserver variability of R.E.N.A.L., PADUA, and centrality index nephrometry score systems. World J Urol 2015; 33: pp. 853-858.
10. Okhunov Z., Rais-Bahrami S., George A.K., et. al.: The comparison of three renal tumor scoring systems: C-Index, P.A.D.U.A., and R.E.N.A.L. nephrometry scores. J Endourol 2011; 25: pp. 1921-1924.
11. Husain F.Z., Rosen D.C., Paulucci D.J., et. al.: R.E.N.A.L. nephrometry score predicts non-neoplastic parenchymal volume removed during robotic partial nephrectomy. J Endourol 2016; 30: pp. 1099-1104.
12. Camacho J.C., Kokabi N., Xing M., et. al.: R.E.N.A.L. (Radius, exophytic/endophytic, nearness to collecting system or sinus, anterior/posterior, and location relative to polar lines) nephrometry score predicts early tumor recurrence and complications after percutaneous ablative therapies for renal cell carcinoma: a 5-year experience. J Vasc Interv Radiol 2015; 26: pp. 686-693.
13. Zhang G.M., Zhu Y., Gan H.L., et. al.: Use of RENAL nephrometry scores for predicting tumor upgrading between core biopsies and surgical specimens: a prospective ex vivo study. Medicine (Baltimore) 2015; 94: pp. e581.
14. Paterson C., Yew-Fung C., Sweeney C., et. al.: Predictors of growth kinetics and outcomes in small renal masses (SRM </=4 cm in size): Tayside Active Surveillance Cohort (TASC) Study. Eur J Surg Oncol 2017; 43: pp. 1589-1597.
15. Leonard M., Tait C.D., Gillan A.S., et. al.: Impact of multiple deprivations on detection, progression and interventions in small renal masses (less than 4 cm) in a population based study. Eur J Surg Oncol 2013; 39: pp. 1157-1163.
16. El-Mokadem I., Budak M., Pillai S., et. al.: Progression, interobserver agreement, and malignancy rate in complex renal cysts (>/= Bosniak category IIF). Urol Oncol 2014; 32: pp. 24. e21-7
17. Ganeswaran D., Sweeney C., Yousif F., et. al.: Population-based linkage of health records to detect urological complications and hospitalisation following transrectal ultrasound-guided biopsies in men suspected of prostate cancer. World J Urol 2014; 32: pp. 309-315.
18. Simhan J., Canter D.J., Sterious S.N., et. al.: Pathological concordance and surgical outcomes of sporadic synchronous unilateral multifocal renal masses treated with partial nephrectomy. J Urol 2013; 189: pp. 43-47.
19. Bagrodia A., Harrow B., Liu Z.W., et. al.: Evaluation of anatomic and morphologic nomogram to predict malignant and high-grade disease in a cohort of patients with small renal masses. Urol Oncol 2014; 32: pp. 37.e17-37.e23.
20. Mullins J.K., Kaouk J.H., Bhayani S., et. al.: Tumor complexity predicts malignant disease for small renal masses. J Urol 2012; 188: pp. 2072-2076.
21. Antonelli A., Furlan M., Sandri M., et. al.: The R.E.N.A.L. nephrometric nomogram cannot accurately predict malignancy or aggressiveness of small renal masses amenable to partial nephrectomy. Clin Genitourin Cancer 2014; 12: pp. 366-372.
22. Osawa T., Hafez K.S., Miller D.C., et. al.: Comparison of percutaneous renal mass biopsy and R.E.N.A.L. nephrometry score nomograms for determining benign vs malignant disease and low-risk vs high-risk renal tumors. Urology 2016; 96: pp. 87-92.
23. Pierorazio P.M., Patel H.D., Johnson M.H., et. al.: Distinguishing malignant and benign renal masses with composite models and nomograms: A systematic review and meta-analysis of clinically localized renal masses suspicious for malignancy. Cancer 2016; 122: pp. 3267-3276.
24. Fujita T., Iwamura M., Wakatabe Y., et. al.: Predictors of benign histology in clinical T1a renal cell carcinoma tumors undergoing partial nephrectomy. Int J Urol 2014; 21: pp. 100-102.
25. Kava B.R., Eldefrawy A., Ayyathurai R., et. al.: Impact of body mass index and tumor location on the incidence of benign histology at the time of nephron-sparing surgery. Int Urol Nephrol 2012; 44: pp. 1319-1324.
26. Ball M.W., Gorin M.A., Bhayani S.B., et. al.: Preoperative predictors of malignancy and unfavorable pathology for clinical T1a tumors treated with partial nephrectomy: a multi-institutional analysis. Urol Oncol 2015; 33: pp. 112. e9-14
27. Jeon H.G., Lee S.R., Kim K.H., et. al.: Benign lesions after partial nephrectomy for presumed renal cell carcinoma in masses 4 cm or less: prevalence and predictors in Korean patients. Urology 2010; 76: pp. 574-579.
28. Murphy A.M., Buck A.M., Benson M.C., et. al.: Increasing detection rate of benign renal tumors: evaluation of factors predicting for benign tumor histologic features during past two decades. Urology 2009; 73: pp. 1293-1297.
29. Pahernik S., Ziegler S., Roos F., et. al.: Small renal tumors: correlation of clinical and pathological features with tumor size. J Urol 2007; 178: pp. 414-417. discussion 416-7
30. Gorin M.A., Ball M.W., Pierorazio P.M., et. al.: Outcomes and predictors of clinical T1 to pathological T3a tumor up-staging after robotic partial nephrectomy: a multi-institutional analysis. J Urol 2013; 190: pp. 1907-1911.
31. Zhao B., Tan Y., Bell D.J., et. al.: Exploring intra- and inter-reader variability in uni-dimensional, bi-dimensional, and volumetric measurements of solid tumors on CT scans reconstructed at different slice intervals. Eur J Radiol 2013; 82: pp. 959-968.
32. Punnen S., Haider M.A., Lockwood G., et. al.: Variability in size measurement of renal masses smaller than 4 cm on computerized tomography. J Urol 2006; 176: pp. 2386-2390. discussion 2390
33. Hawken S.R., Krishnan N.K., Ambani S.N., et. al.: Effect of delayed resection after initial surveillance and tumor growth rate on final surgical pathology in patients with small renal masses (SRMs). Urol Oncol 2016; 34: pp. 486.e9-486.e15.
34. Jewett M.A., Mattar K., Basiuk J., et. al.: Active surveillance of small renal masses: progression patterns of early stage kidney cancer. Eur Urol 2011; 60: pp. 39-44.
35. Pierorazio P.M., Johnson M.H., Ball M.W., et. al.: Five-year analysis of a multi-institutional prospective clinical trial of delayed intervention and surveillance for small renal masses: the DISSRM registry. Eur Urol 2015; 68: pp. 408-415.
36. Nayyar M., Cheng P., Desai B., et. al.: Active surveillance of small renal masses: a review on the role of imaging with a focus on growth rate. J Comput Assist Tomogr 2016; 40: pp. 517-523.
37. Gill I.S., Kavoussi L.R., Lane B.R., et. al.: Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol 2007; 178: pp. 41-46.
38. Simhan J., Smaldone M.C., Tsai K.J., et. al.: Objective measures of renal mass anatomic complexity predict rates of major complications following partial nephrectomy. Eur Urol 2011; 60: pp. 724-730.
39. Helenon O., Eiss D., Debrito P., et. al.: How to characterise a solid renal mass: a new classification proposal for a simplified approach. Diagn Interv Imaging 2012; 93: pp. 232-245.
40. Somani B.K., Nabi G., Thorpe P., et. al.: Clinical urological surgeons, image-guided biopsy-diagnosed renal cell carcinoma: critical appraisal of technique and long-term follow-up. Eur Urol 2007; 51: pp. 1289-1295. discussion 1296-7