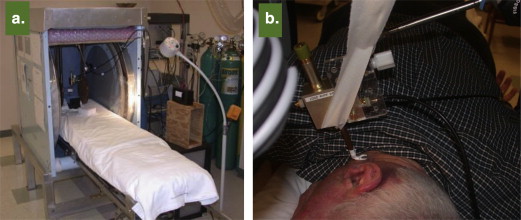
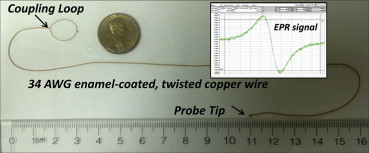
Electron paramagnetic resonance (EPR) spectroscopy has been well established as a viable technique for measurement of free radicals and oxygen in biological systems, from in vitro cellular systems to in vivo small animal models of disease. However, the use of EPR in human subjects in the clinical setting, although attractive for a variety of important applications such as oxygen measurement, is challenged with several factors including the need for instrumentation customized for human subjects, probe, and regulatory constraints. This article describes the rationale and development of the first clinical EPR systems for two important clinical applications, namely, measurement of tissue oxygen (oximetry) and radiation dose (dosimetry) in humans. The clinical spectrometers operate at 1.2 GHz frequency and use surface-loop resonators capable of providing topical measurements up to 1 cm depth in tissues. Tissue pO 2 measurements can be carried out noninvasively and repeatedly after placement of an oxygen-sensitive paramagnetic material (currently India ink) at the site of interest. Our EPR dosimetry system is capable of measuring radiation-induced free radicals in the tooth of irradiated human subjects to determine the exposure dose. These developments offer potential opportunities for clinical dosimetry and oximetry, which include guiding therapy for individual patients with tumors or vascular disease by monitoring of tissue oxygenation. Further work is in progress to translate this unique technology to routine clinical practice.
Electron paramagnetic resonance (EPR) (also called electron spin resonance) spectroscopy shares many of the features of nuclear magnetic resonance (NMR), spectroscopy and magnetic resonance imaging (MRI) including underlying principles, discovery period, and their evolution to become an indispensable tool for in vivo biological applications. Particularly, proton MRI has rapidly emerged to become a unique device for noninvasive measurement (imaging) of tissue pathophysiology in the clinic. On the other hand, EPR, which relies on paramagnetic species with unpaired electrons, despite its superiority to NMR in terms of detection sensitivity, has not advanced to use for pertinent clinical applications for a variety of reasons. The most important impediments are the lack of adequate levels of paramagnetic species in biological systems, shorter relaxation times of unpaired electrons when compared to protons, and the need to use microwave radiation as source of excitation. In spite of the limitations, the last three decades have seen some innovative and concerted successful progress that has developed this unique technology for making very useful measurements in living systems . The most pertinent advances include the development of low-frequency instrumentation including lumped-circuit resonators, time-domain detection, imaging capabilities, and molecular probes for extracting specific information of interest from tissues of both animal models and human . The technological advances have now reached a stage where useful clinical measurements such as tissue oxygenation (oximetry) or radiation exposure (dosimetry) in human subjects have become a reality ( Table 1 ). This article presents an overview of the unique opportunities of the EPR technology for measurements in humans and challenges that need to be addressed before it can achieve widespread acceptance as a useful clinical device. Two potential areas of clinical application, namely, oximetry and dosimetry, for which substantial progresses have been made are highlighted.
Table 1
Clinical EPR at Dartmouth
Clinical Problem Parameter to Be Measured Status of Measurements in Human Subjects Rationale for Using In Vivo EPR Measurements Dosimetry Radiation-induced free radicals Extensive measurements underway Fills unique niche for emergency dosimetry based on physical parameter Peripheral vascular disease Oxygen at sites of likely pathologies Measurements underway in normal volunteers; patients to be studied in near future The pO 2 in the tissues is the most significant pathophysiological variable; no other method available to make such direct measurements. The pO 2 measurements will facilitate evaluating progression of disease and success of therapeutic intervention Cancer pO 2 in tumors Measurements underway in patients with superficial tumors; IDEs submitted for permission to start studies with embedded materials The response of tumors to cytotoxic therapy, especially ionizing radiation, is critically dependent on pO 2 . Antitumor therapies are given repeatedly and often change pO 2 . Knowledge of the changes in individual patients would significantly optimize the timing of the therapy Wound healing pO 2 at various sites in wounds, perhaps reactive oxygen species Existing clinical protocols use an apparatus compatible with EPR measurements. The pO 2 is a critical variable for successful healing of wounds. Direct measurements would identify patients likely to have poor healing and follow responses to therapy Radiation-induced fibrosis pO 2 in irradiated tumor beds and peripheral normal tissue Initial studies have been started Radiation-induced hypoxia may play a critical role in the signaling of proinflammatory, profibrotic, and proangiogenic growth factors and cytokines that lead to tissue fibrosis.
EPR, electron paramagnetic resonance; IDE, investigational device exemption.
Measurement of tissue oxygenation (oximetry)
Although the discovery of oxygen was made in the 18th century, measurements of oxygen concentration (oximetry) in living systems (in vivo) are only recent phenomena. Some measurements were made in the 1960s, but it was in the late 1980s when the computerized polarographic needle electrode system became available that it was used extensively to assess the oxygenation in tumors clinically. The use of this technique helped to establish the role of hypoxia in the efficacy of radiation therapy . Now, there are several potentially clinically useful methods that are based on other principles, including fluorescence quenching, phosphorescence, optical detection, immunohistochemical, and magnetic resonance techniques. Some of the important criteria for improvements in the ability to make successful clinical measurements of oxygenation include minimal or no invasiveness, capability to make repeated measurements, accessibility to the region of interest, appropriate spatial resolution, adequate depth of measurement, accuracy and robustness of measurements, usefulness of the parameter reported for clinical purposes, measurement time consistent with use in patients, ease of use in the clinical setting, and potential for the instrumentation to be commercially available. It is especially important that the method should enable repeated measurements from the region of interest to follow the changes in oxygenation over a period of time, preferably for up to several weeks, months, or even years . The technique should also provide appropriate spatial and temporal resolution. The depth of measurement (penetration) and accessibility to the region of interest are some of the important factors for establishing the scope of applicability of the technique.
Existing methods for tissue oximetry in vivo
Although a few techniques are available that can provide direct measurements of tissue pO 2 (especially oxygen polarographic electrodes, the OxyLite fluorescence-quenching technique, and direct injection of oxygen-sensitive NMR probes based on fluorine ), these techniques have the disadvantage of not being able to be used repeatedly. The inability to make repeated measurements is a disadvantage because the most valuable clinical usefulness depends on obtaining repeated measurements so that changes due to the treatment and the disease process can be followed. There are also some concerns about the potential for artifacts because the invasiveness of these techniques can cause perturbations precisely at the time that the measurements are being made . To date, only the polarographic method has been systematically used in humans .
Some widely available noninvasive techniques to assess oxygenation provide data on parameters that, although related to tissue pO 2 , do not measure pO 2 directly. This especially includes techniques such as blood oxygen level–dependent (BOLD) MRI, NMR proton spectroscopy, diffusion-weighted imaging MRI, duplex Doppler ultrasound, 18 F-Miso positron emission tomography, and near-infrared (NIR) measurements of hemoglobin. These techniques can provide information on the saturation of hemoglobin within the vascular system or hemodynamics or redox-related metabolites or reactions, but although this can be valuable to know, it does not provide direct quantification of the pO 2 in the site of interest and surrounding tissues. The clinical importance of direct measurement of pO 2 is especially high in tumors, in which typically the microvasculature is complex and irregular and therefore is very difficult to extrapolate these indirect measurements to the pO 2 in the tumor . EPR oximetry could provide the data to facilitate relating these indirect methods to the actual tumor or tissue pO 2 , thereby clarifying the conditions under which these other methods can provide the desired clinically relevant information on pO 2 .
EPR oximetry for biological systems including viable tissues
Get Radiology Tree app to read full this article<
Paramagnetic probes for possible use in clinical EPR oximetry
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Multisite oximetry
Get Radiology Tree app to read full this article<
Clinical potential of EPR oximetry
Get Radiology Tree app to read full this article<
Constraints for clinical EPR development
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Clinical oximetry measurements
Get Radiology Tree app to read full this article<
Table 2
pO 2 Measurements in Human Tumors Measured In Vivo with Challenge by Breathing Enriched Oxygen
Number Cancer Site Response to Oxygen With Air (mmHg) With Oxygen (mmHg) 1 Melanoma Knee Yes <10 >50 2 Lymphoma Posterior thigh Yes <5 >100 3 Melanoma Foot No Anoxic Anoxic 4 Melanoma Scalp No Anoxic Anoxic 5 Sarcoma Nose No ∼10 ∼10 6 Melanoma Scalp and Neck Yes ∼6 ∼12 7 Melanoma Scapula Yes <1 <5 8 Melanoma Chest No ∼10 ∼10 9 Melanoma Calf Yes Anoxic ∼3 10 Sarcoma Posterior thigh Yes <3 >10 11 Sarcoma Posterior thigh Yes ∼1 >5
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Dosimetry
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusion
Get Radiology Tree app to read full this article<
Acknowledgment
Get Radiology Tree app to read full this article<
References
1. Salikhov I., Walczak T., Lesniewski P., et. al.: EPR spectrometer for clinical applications. Magn Reson Med 2005; 54: pp. 1317-1320.
2. Swartz H.M., Walczak T.: Developing in vivo EPR oximetry for clinical use. Adv Exp Med Biol 1998; 454: pp. 243-252.
3. Williams B.B., Khan N., Zaki B., et. al.: Clinical electron paramagnetic resonance (EPR) oximetry using India ink. Adv Exp Med Biol 2010; 662: pp. 149-156.
4. Swartz H.M., Berliner L.J.: Biological magnetic resonance.2003.Plenum PressNew York
5. Rockwell S., Moulder J.E.: Hypoxic fractions of human tumors xenografted into mice: a review. Int J Radiat Oncol Biol Phys 1990; 19: pp. 197-202.
6. Vikram D.S., Zweier J.L., Kuppusamy P.: Methods for noninvasive imaging of tissue hypoxia. Antioxid Redox Signal 2007; 9: pp. 1745-1756.
7. Swartz H.M.: Using EPR to measure a critical but often unmeasured component of oxidative damage: oxygen. Antioxid Redox Signal 2004; 6: pp. 677-686.
8. Swartz H.M., Khan N., Buckey J., et. al.: Clinical applications of EPR: overview and perspectives. NMR Biomed 2004; 17: pp. 335-351.
9. Springett R., Swartz H.M.: Measurements of oxygen in vivo: overview and perspectives on methods to measure oxygen within cells and tissues. Antioxid Redox Signal 2007; 9: pp. 1295-1301.
10. Mason R.P., Hunjan S., Le D., et. al.: Regional tumor oxygen tension: fluorine echo planar imaging of hexafluorobenzene reveals heterogeneity of dynamics. Int J Radiat Oncol Biol Phys 1998; 42: pp. 747-750.
11. Griffiths J.R., Robinson S.P.: The OxyLite: a fibre-optic oxygen sensor. Br J Radiol 1999; 72: pp. 627-630.
12. Braun R.D., Lanzen J.L., Snyder S.A., et. al.: Comparison of tumor and normal tissue oxygen tension measurements using OxyLite or microelectrodes in rodents. Am J Physiol Heart Circ Physiol 2001; 280: pp. H2533-H2544.
13. Nozue M., Lee I., Yuan F., et. al.: Interlaboratory variation in oxygen tension measurement by Eppendorf “Histograph” and comparison with hypoxic marker. J Surg Oncol 1997; 66: pp. 30-38.
14. O’Hara J.A., Hou H., Demidenko E., et. al.: Simultaneous measurement of rat brain cortex PtO2 using EPR oximetry and a fluorescence fiber-optic sensor during normoxia and hyperoxia. Physiol Meas 2005; 26: pp. 203-213.
15. O’Hara J.A., Khan N., Hou H., et. al.: Comparison of EPR oximetry and Eppendorf polarographic electrode assessments of rat brain PtO2. Physiol Meas 2004; 25: pp. 1413-1423.
16. Vikram D.S., Bratasz A., Ahmad R., et. al.: A comparative evaluation of EPR and OxyLite oximetry using a random sampling of pO(2) in a murine tumor. Radiat Res 2007; 168: pp. 308-315.
17. Vaupel P., Schlenger K., Knoop C., et. al.: Oxygenation of human tumors: evaluation of tissue oxygen distribution in breast cancers by computerized O2 tension measurements. Cancer Res 1991; 51: pp. 3316-3322.
18. Baudelet C., Gallez B.: How does blood oxygen level-dependent (BOLD) contrast correlate with oxygen partial pressure (pO2) inside tumors?. Magn Reson Med 2002; 48: pp. 980-986.
19. Jordan B.F., Crokart N., Baudelet C., et. al.: Complex relationship between changes in oxygenation status and changes in R*2: the case of insulin and NS-398, two inhibitors of oxygen consumption. Magn Reson Med 2006; 56: pp. 637-643.
20. Ahmad R., Kuppusamy P.: Theory, instrumentation, and applications of electron paramagnetic resonance oximetry. Chem Rev 2010; 110: pp. 3212-3236.
21. Smirnov A.I., Norby S.W., Walczak T., et. al.: Physical and instrumental considerations in the use of lithium phthalocyanine for measurements of the concentration of the oxygen. J Magn Reson B 1994; 103: pp. 95-102.
22. Elas M., Ahn K.H., Parasca A., et. al.: Electron paramagnetic resonance oxygen images correlate spatially and quantitatively with OxyLite oxygen measurements. Clin Cancer Res 2006; 12: pp. 4209-4217.
23. Froncisz W., Lai C.S., Hyde J.S.: Spin-label oximetry: kinetic study of cell respiration using a rapid-passage T1-sensitive electron spin resonance display. Proc Natl Acad Sci U S A 1985; 82: pp. 411-415.
24. Gallez B., Bacic G., Goda F., et. al.: Use of nitroxides for assessing perfusion, oxygenation, and viability of tissues: in vivo EPR and MRI studies. Magn Reson Med 1996; 35: pp. 97-106.
25. Grinberg O.Y., Friedman B.J., Swartz H.M.: Intramyocardial pO2 measured by EPR. Adv Exp Med Biol 1997; 428: pp. 261-268.
26. Halpern H.J., Yu C., Peric M., et. al.: Measurement of differences in pO2 in response to perfluorocarbon/carbogen in FSa and NFSa murine fibrosarcomas with low-frequency electron paramagnetic resonance oximetry. Radiat Res 1996; 145: pp. 610-618.
27. Hou H., Dong R., Li H., et. al.: Dynamic changes in oxygenation of intracranial tumor and contralateral brain during tumor growth and carbogen breathing: a multisite EPR oximetry with implantable resonators. J Magn Reson 2012; 214: pp. 22-28.
28. Ilangovan G., Li H., Zweier J.L., et. al.: Effect of carbogen-breathing on redox status of the RIF-1 tumor. Adv Exp Med Biol 2003; 510: pp. 13-17.
29. Ilangovan G., Liebgott T., Kutala V.K., et. al.: EPR oximetry in the beating heart: myocardial oxygen consumption rate as an index of postischemic recovery. Magn Reson Med 2004; 51: pp. 835-842.
30. Khan M., Kutala V.K., Wisel S., et. al.: Measurement of oxygenation at the site of stem cell therapy in a murine model of myocardial infarction. Adv Exp Med Biol 2008; 614: pp. 45-52.
31. Khan M., Mohan I.K., Kutala V.K., et. al.: Sulfaphenazole protects heart against ischemia-reperfusion injury and cardiac dysfunction by overexpression of iNOS, leading to enhancement of nitric oxide bioavailability and tissue oxygenation. Antioxid Redox Signal 2009; 11: pp. 725-738.
32. Kuppusamy P., Afeworki M., Shankar R.A., et. al.: In vivo electron paramagnetic resonance imaging of tumor heterogeneity and oxygenation in a murine model. Cancer Res 1998; 58: pp. 1562-1568.
33. Liu K.J., Gast P., Moussavi M., et. al.: Lithium phthalocyanine: a probe for electron paramagnetic resonance oximetry in viable biological systems. Proc Natl Acad Sci U S A 1993; 90: pp. 5438-5442.
34. Krishna M.C., English S., Yamada K., et. al.: Overhauser enhanced magnetic resonance imaging for tumor oximetry: coregistration of tumor anatomy and tissue oxygen concentration. Proc Natl Acad Sci U S A 2002; 99: pp. 2216-2221.
35. Matsumoto A., Matsumoto K., Matsumoto S., et. al.: Intracellular hypoxia of tumor tissue estimated by noninvasive electron paramagnetic resonance oximetry technique using paramagnetic probes. Biol Pharm Bull 2011; 34: pp. 142-145.
36. Rosen G.M., Halpern H.J., Brunsting L.A., et. al.: Direct measurement of nitroxide pharmacokinetics in isolated hearts situated in a low-frequency electron spin resonance spectrometer: implications for spin trapping and in vivo oxymetry. Proc Natl Acad Sci U S A 1988; 85: pp. 7772-7776.
37. Zweier J.L., Kuppusamy P.: Electron paramagnetic resonance measurements of free radicals in the intact beating heart: a technique for detection and characterization of free radicals in whole biological tissues. Proc Natl Acad Sci U S A 1988; 85: pp. 5703-5707.
38. Li H., Hou H., Sucheta A., et. al.: Implantable resonators—a technique for repeated measurement of oxygen at multiple deep sites with in vivo EPR. Adv Exp Med Biol 2010; 662: pp. 265-272.
39. Vahidi N., Clarkson R.B., Liu K.J., et. al.: In vivo and in vitro EPR oximetry with fusinite: a new coal-derived, particulate EPR probe. Magn Reson Med 1994; 31: pp. 139-146.
40. Swartz H.M., Liu K.J., Goda F., et. al.: India ink: a potential clinically applicable EPR oximetry probe. Magn Reson Med 1994; 31: pp. 229-232.
41. Nakashima T., Goda F., Jiang J., et. al.: Use of EPR oximetry with India ink to measure the pO2 in the liver in vivo in mice. Magn Reson Med 1995; 34: pp. 888-892.
42. James P.E., Goda F., Grinberg O.Y., et. al.: Intrarenal pO2 measured by EPR oximetry and the effects of bacterial endotoxin. Adv Exp Med Biol 1997; 411: pp. 557-568.
43. Goda F., Liu K.J., Walczak T., et. al.: In vivo oximetry using EPR and India ink. Magn Reson Med 1995; 33: pp. 237-245.
44. Pandian R.P., Dolgos M., Dang V., et. al.: Structure and oxygen-sensing paramagnetic properties of a new lithium 1,8,15,22-tetraphenoxyphthalocyanine radical probe for biological oximetry. Chem Mater 2007; 19: pp. 3545-3552.
45. Pandian R.P., Dolgos M., Marginean C., et. al.: Molecular packing and magnetic properties of lithium naphthalocyanine crystals: hollow channels enabling permeability and paramagnetic sensitivity to molecular oxygen. J Mater Chem 2009; 19: pp. 4138-4147.
46. Pandian R.P., Kim Y.I., Woodward P.M., et. al.: Open molecular framework in lithium octabutoxy-naphthalocyanine paramagnetic crystal: implications for the detection of oxygen and nitric oxide by EPR spectroscopy. J Mater Chem 2006; 16: pp. 3609-3618.
47. Pandian R.P., Parinandi N.L., Ilangovan G., et. al.: Novel particulate spin probe for targeted determination of oxygen in cells and tissues. Free Radic Biol Med 2003; 35: pp. 1138-1148.
48. Khan N., Hou H., Hein P., et. al.: Black magic and EPR oximetry: from lab to initial clinical trials. Adv Exp Med Biol 2005; 566: pp. 119-125.
49. Gallez B., Debuyst R., Dejehet F., et. al.: Small particles of fusinite and carbohydrate chars coated with aqueous soluble polymers: preparation and applications for in vivo EPR oximetry. Magn Reson Med 1998; 40: pp. 152-159.
50. Jordan B.F., Baudelet C., Gallez B.: Carbon-centered radicals as oxygen sensors for in vivo electron paramagnetic resonance: screening for an optimal probe among commercially available charcoals. MAGMA 1998; 7: pp. 121-129.
51. Mahy P., De Bast M., Gallez B., et. al.: In vivo colocalization of 2-nitroimidazole EF5 fluorescence intensity and electron paramagnetic resonance oximetry in mouse tumors. Radiother Oncol 2003; 67: pp. 53-61.
52. Ilangovan G., Manivannan A., Li H., et. al.: A naphthalocyanine-based EPR probe for localized measurements of tissue oxygenation. Free Radic Biol Med 2002; 32: pp. 139-147.
53. He J., Beghein N., Ceroke P., et. al.: Development of biocompatible oxygen-permeable films holding paramagnetic carbon particles: evaluation of their performance and stability in EPR oximetry. Magn Reson Med 2001; 46: pp. 610-614.
54. Meenakshisundaram G., Eteshola E., Pandian R.P., et. al.: Fabrication and physical evaluation of a polymer-encapsulated paramagnetic probe for biomedical oximetry. Biomed Microdevices 2009; 11: pp. 773-782.
55. Meenakshisundaram G., Pandian R.P., Eteshola E., et. al.: A paramagnetic implant containing lithium naphthalocyanine microcrystals for high-resolution biological oximetry. J Magn Reson 2010; 203: pp. 185-189.
56. Hou H., Li H., Dong R., et. al.: Cerebral oxygenation of the cortex and striatum following normobaric hyperoxia and mild hypoxia in rats by EPR oximetry using multi-probe implantable resonators. Adv Exp Med Biol 2011; 701: pp. 61-67.
57. Smirnov A.I., Norby S.W., Clarkson R.B., et. al.: Simultaneous multi-site EPR spectroscopy in vivo. Magn Reson Med 1993; 30: pp. 213-220.
58. Grinberg O.Y., Smirnov A.I., Swartz H.M.: High spatial resolution multi-site EPR oximetry. The use of convolution-based fitting method. J Magn Reson 2001; 152: pp. 247-258.
59. Som S., Potter L.C., Ahmad R., et. al.: EPR oximetry in three spatial dimensions using sparse spin distribution. J Magn Reson 2008; 193: pp. 210-217.
60. Aboagye E.O., Maxwell R.J., Horsman M.R., et. al.: The relationship between tumour oxygenation determined by oxygen electrode measurements and magnetic resonance spectroscopy of the fluorinated 2-nitroimidazole SR-4554. Br J Cancer 1998; 77: pp. 65-70.
61. Kim J.G., Zhao D., Song Y., et. al.: Interplay of tumor vascular oxygenation and tumor pO2 observed using near-infrared spectroscopy, an oxygen needle electrode, and 19F MR pO2 mapping. J Biomed Opt 2003; 8: pp. 53-62.
62. Laukemper-Ostendorf S., Scholz A., Burger K., et. al.: 19F-MRI of perflubron for measurement of oxygen partial pressure in porcine lungs during partial liquid ventilation. Magn Reson Med 2002; 47: pp. 82-89.
63. Mason R.P., Rodbumrung W., Antich P.P.: Hexafluorobenzene: a sensitive 19F NMR indicator of tumor oxygenation. NMR Biomed 1996; 9: pp. 125-134.
64. Prasad P.V., Edelman R.R., Epstein F.H.: Noninvasive evaluation of intrarenal oxygenation with BOLD MRI. Circulation 1996; 94: pp. 3271-3275.
65. Berliner L.J.: In vivo EPR (ESR): theory and applications.Berliner Biological magnetic resonance.2002.PlenumNew York:
66. Pandian R.P., Chacko S.M., Kuppusamy M.L., et. al.: Evaluation of lithium naphthalocyanine (LiNc) microcrystals for biological EPR oximetry. Adv Exp Med Biol 2011; 701: pp. 29-36.
67. Dinguizli M., Beghein N., Gallez B.: Retrievable micro-inserts containing oxygen sensors for monitoring tissue oxygenation using EPR oximetry. Physiol Meas 2008; 29: pp. 1247-1254.
68. Dinguizli M., Jeumont S., Beghein N., et. al.: Development and evaluation of biocompatible films of polytetrafluoroethylene polymers holding lithium phthalocyanine crystals for their use in EPR oximetry. Biosens Bioelectron 2006; 21: pp. 1015-1022.
69. Meenakshisundaram G., Eteshola E., Pandian R.P., et. al.: Oxygen sensitivity and biocompatibility of an implantable paramagnetic probe for repeated measurements of tissue oxygenation. Biomed Microdevices 2009; 11: pp. 817-826.
70. Mata A., Fleischman A.J., Roy S.: Characterization of polydimethylsiloxane (PDMS) properties for biomedical micro/nanosystems. Biomed Microdevices 2005; 7: pp. 281-293.
71. Abbasi F., Mirzadeh H., Katbab A.A.: Modification of polysiloxane polymers for biomedical applications: a review. Polym Int 2001; 50: pp. 1279-1287.
72. Belanger M.C., Marois Y.: Hemocompatibility, biocompatibility, inflammatory and in vivo studies of primary reference materials low-density polyethylene and polydimethylsiloxane: a review. J Biomed Mater Res 2001; 58: pp. 467-477.
73. Jiang J., Nakashima T., Liu K.J., et. al.: Measurement of PO2 in liver using EPR oximetry. J Appl Physiol 1996; 80: pp. 552-558.
74. Liu K.J., Miyake M., James P.E., et. al.: Separation and enrichment of the active component of carbon based paramagnetic materials for use in EPR oximetry. J Magn Reson 1998; 133: pp. 291-298.
75. O’Hara J.A., Goda F., Demidenko E., et. al.: Effect on regrowth delay in a murine tumor of scheduling split-dose irradiation based on direct pO2 measurements by electron paramagnetic resonance oximetry. Radiat Res 1998; 150: pp. 549-556.
76. Khan N., Williams B.B., Hou H., et. al.: Repetitive tissue pO2 measurements by electron paramagnetic resonance oximetry: current status and future potential for experimental and clinical studies. Antioxid Redox Signal 2007; 9: pp. 1169-1182.
77. Swartz H.M., Burke G., Coey M., et. al.: In vivo EPR for dosimetry. Radiat Meas 2007; 42: pp. 1075-1084.
78. Swartz H.M., Flood A.B., Williams B.B., et. al.: Electron paramagnetic resonance dosimetry for a large-scale radiation incident. Health Phys 2012; 103: pp. 255-267.
79. Black P.J., Swarts S.G.: Ex vivo analysis of irradiated fingernails: chemical yields and properties of radiation-induced and mechanically-induced radicals. Health Phys 2010; 98: pp. 301-308.
80. He X., Gui J., Matthews T.P., et. al.: Advances towards using finger/toenail dosimetry to triage a large population after potential exposure to ionizing radiation. Radiat Meas 2011; 46: pp. 882-887.
81. Romanyukha A., Reyes R.A., Trompier F., et. al.: Fingernail dosimetry: current status and perspectives. Health Phys 2010; 98: pp. 296-300.
82. Wilcox D.E., He X., Gui J., et. al.: Dosimetry based on EPR spectral analysis of fingernail clippings. Health Phys 2010; 98: pp. 309-317.
83. Williams B.B., Dong R., Flood A.B., et. al.: A Deployable in vivo EPR tooth dosimeter for triage after a radiation event involving large populations. Radiat Res 2011; 46: pp. 772-777.
84. Williams B.B., Dong R., Kmiec M., et. al.: Development of in vivo tooth EPR for individual radiation dose estimation and screening. Health Phys 2010; 98: pp. 327-338.
85. Williams B.B., Dong R., Nicolalde R.J., et. al.: Physically-based biodosimetry using in vivo EPR of teeth in patients undergoing total body irradiation. Int J Radiat Biol 2011; 87: pp. 766-775.