Rationale and Objectives
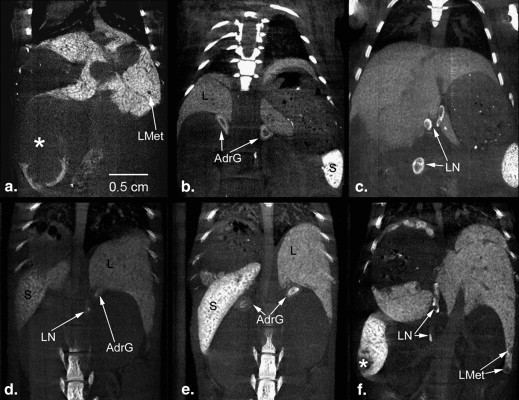
The purpose of this study was to compare different contrast agents for longitudinal liver and spleen imaging in a mouse model of liver metastasis.
Materials and Methods
Mice developing liver metastases underwent longitudinal micro-computed tomography imaging after injection of Fenestra LC, ExiTron nano 6000, or ExiTron nano 12000. Elimination times and contrast enhancement of liver and spleen were compared.
Results
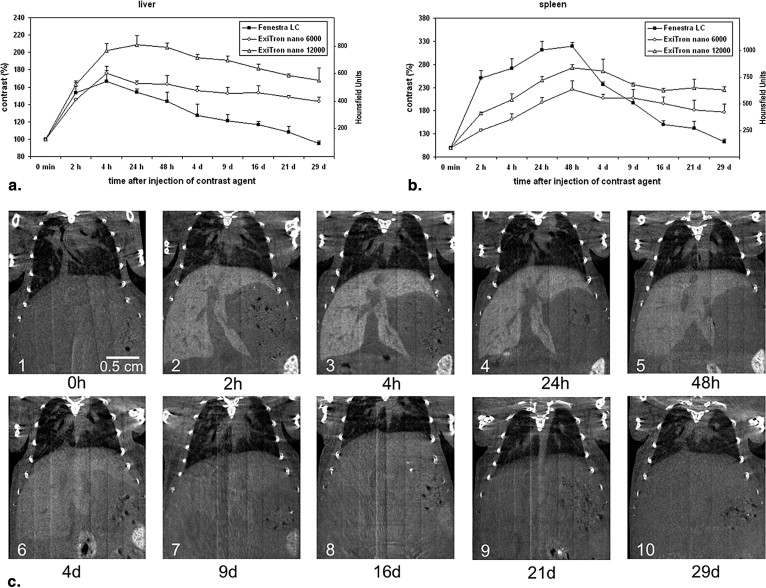
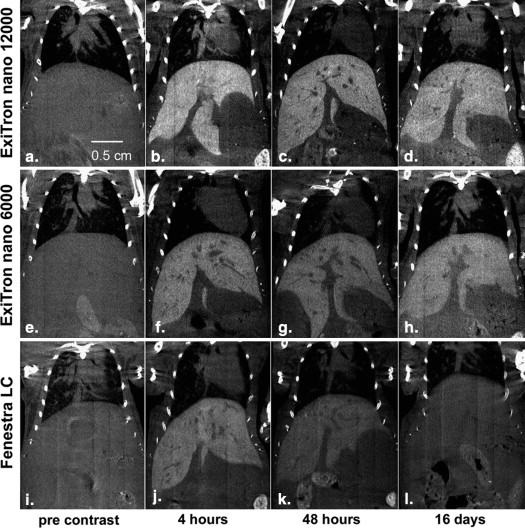
For all contrast agents, liver contrast peaked at approximately 4 hours and spleen contrast at 48 hours postinjection. A single dose of 100 μL of ExiTron nano 6000 or 12000 resulted in longstanding enhancement of liver and spleen tissue for longer than 3 weeks, whereas repeated injections of 400 μL of Fenestra LC were required to retain contrast at acceptable levels and allowed imaging of the liver/spleen for up to 2 and 9 days, respectively.
Conclusion
Both ExiTron nano agents provide longer and stronger contrast enhancement of liver and spleen compared to Fenestra LC, and they do so at a 75% lower injection volume in mice.
Small rodents, especially mice, are frequently used to model and study human diseases. One of the methods used for noninvasive longitudinal monitoring of pathological processes in small animals is micro–computed tomography (micro-CT), which has gained increasing importance in preclinical research within the past decade . For micro-CT imaging of liver disease, high soft tissue contrast is required to detect small pathological lesions such as liver metastases. For this purpose, contrast agents providing a positive (ie, hyperdense) contrast of healthy liver tissue have been developed . One of the hitherto frequently used agents is Fenestra LC, which is selectively taken up by hepatocytes via an apolipoprotein E (apoE) receptor-mediated pathway. As tumor cells usually do not express apoE receptor and do not contain relevant numbers of macrophages, liver-specific contrast agents allow the delineation of the nonenhancing and thus relatively hypodense tumors within the hyperdense healthy liver tissue. Drawbacks of Fenestra LC are a relatively high injection volume of up to 400 μL/20-g mouse as recommended by the manufacturer and the required repeated injections for longitudinal studies due to the relatively short elimination time.
Recently, ExiTron nano, a novel preclinical liver-specific CT contrast agent, has been introduced. This alkaline earth metal–based nanoparticulate contrast agent creates a strong and specific contrast of the reticuloendothelial system (RES) of the liver and the spleen at injection volumes of as low as 100 μL. Furthermore, ExiTron nano provides a long-lasting contrast of the liver and spleen for several weeks after injection.
Get Radiology Tree app to read full this article<
Materials and methods
Mouse Model of Liver Metastasis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Micro-CT Contrast Agents
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Micro-CT Imaging Procedure
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Image Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Schambach S.J., Bag S., Schilling L., et. al.: Application of micro-CT in small animal imaging. Methods 2010; 50: pp. 2-13.
2. Weichert J.P., Longino M.A., Bakan D.A., et. al.: Polyiodinated triglyceride analogs as potential computed tomography imaging agents for the liver. J Med Chem 1995; 38: pp. 636-646.
3. Weber S.M., Peterson K.A., Durkee B., et. al.: Imaging of murine liver tumor using microCT with a hepatocyte-selective contrast agent: accuracy is dependent on adequate contrast enhancement. J Surg Res 2004; 119: pp. 41-45.
4. Montet X., Pastor C.M., Vallee J.P., et. al.: Improved visualization of vessels and hepatic tumors by micro-computed tomography (CT) using iodinated liposomes. Invest Radiol 2007; 42: pp. 652-658.
5. Longino M.A., Bakan D.A., Weichert J.P., et. al.: Formulation of polyiodinated triglyceride analogues in a chylomicron remnant-like liver-selective delivery vehicle. Pharm Res 1996; 13: pp. 875-879.
6. Bakan D.A., Weichert J.P., Longino M.A., et. al.: Polyiodinated triglyceride lipid emulsions for use as hepatoselective contrast agents in CT: effects of physicochemical properties on biodistribution and imaging profiles. Invest Radiol 2000; 35: pp. 158-169.
7. Bakan D.A., Longino M.A., Weichert J.P., et. al.: Physicochemical characterization of a synthetic lipid emulsion for hepatocyte-selective delivery of lipophilic compounds: application to polyiodinated triglycerides as contrast agents for computed tomography. J Pharm Sci 1996; 85: pp. 908-914.
8. Bakan D.A., Doerr-Stevens J.K., Weichert J.P., et. al.: Imaging efficacy of a hepatocyte-selective polyiodinated triglyceride for contrast-enhanced computed tomography. Am J Ther 2001; 8: pp. 359-365.
9. Willekens I., Lahoutte T., Buls N., et. al.: Time-course of contrast enhancement in spleen and liver with Exia 160, Fenestra LC, and VC. Mol Imaging Biol 2009; 11: pp. 128-135.
10. Suckow C.E., Stout D.B.: MicroCT liver contrast agent enhancement over time, dose, and mouse strain. Mol Imaging Biol 2008; 10: pp. 114-120.
11. Ohta S., Lai E.W., Morris J.C., et. al.: MicroCT for high-resolution imaging of ectopic pheochromocytoma tumors in the liver of nude mice. Int J Cancer 2006; 119: pp. 2236-2241.
12. Almajdub M., Nejjari M., Poncet G., et. al.: In-vivo high-resolution X-ray microtomography for liver and spleen tumor assessment in mice. Contrast Media Mol Imaging 2007; 2: pp. 88-93.
13. Martiniova L., Schimel D., Lai E.W., et. al.: In vivo micro-CT imaging of liver lesions in small animal models. Methods 2010; 50: pp. 20-25.
14. Boll H., Nittka S., Doyon F., et. al.: Micro-CT based experimental liver imaging using a nanoparticulate contrast agent: a longitudinal study in mice. PLoS One 2011; 6: pp. e25692.
15. Schwegler C., Dorn-Beineke A., Nittka S., et. al.: Monoclonal anti-idiotype antibody 6G6.C4 fused to GM-CSF is capable of breaking tolerance to carcinoembryonic antigen (CEA) in CEA-transgenic mice. Cancer Res 2005; 65: pp. 1925-1933.
16. Eades-Perner A.M., van der Putten H., Hirth A., et. al.: Mice transgenic for the human carcinoembryonic antigen gene maintain its spatiotemporal expression pattern. Cancer Res 1994; 54: pp. 4169-4176.
17. Boll H., Bag S., Schambach S.J., et. al.: High-speed single-breath-hold micro-computed tomography of thoracic and abdominal structures in mice using a simplified method for intubation. J Comput Assist Tomogr 2010; 34: pp. 783-790.
18. Boll H., Bag S., Nolte I.S., et. al.: Double-contrast micro-CT colonoscopy in live mice. Int J Colorectal Dis 2011; 26: pp. 721-727.
19. Kerl H.U., Isaza C.T., Boll H., et. al.: Evaluation of a continuous-rotation, high-speed scanning protocol for micro-computed tomography. J Comput Assist Tomogr 2011; 35: pp. 517-523.
20. Fiebig T., Boll H., Figueiredo G., et. al.: Three-dimensional in vivo imaging of the murine liver: a micro-computed tomography-based anatomical study. PLoS One 2012; 7: pp. e31179.
21. Martiniova L., Kotys M.S., Thomasson D., et. al.: Noninvasive monitoring of a murine model of metastatic pheochromocytoma: a comparison of contrast-enhanced microCT and nonenhanced MRI. J Magn Reson Imaging 2009; 29: pp. 685-691.
22. Graham K.C., Detombe S.A., MacKenzie L.T., et. al.: Contrast-enhanced microcomputed tomography using intraperitoneal contrast injection for the assessment of tumor-burden in liver metastasis models. Invest Radiol 2008; 43: pp. 488-495.
23. Advanced Research Technologies (ART) Inc. User guide: Vascular Imaging with Fenestra VC and Siemens’ MicroCAT II Scanner . Available at: http://www.art.ca/docs/products/fenestra/PMK-UG009-E02%20Vascular%20Imaging%20with%20Fenestra%20VC.pdf . Accessed July 17, 2013.
24. Ton GN, Chia NPH, Dow WC, Bakan DA, Weichert JP. Poster 243: MicroCT Evaluations of a Long-Lasting Blood Pool and Hepatobiliary Contrast Agents Following Multiple Intravenous Injections in Normal Mouse Models. Annual conference of the AMI, Orlando, Florida, March 25-29, 2006.
25. Advanced Research Technologies (ART) Inc. Imaging Hepatic CT-26 Tumors in Balb/c Mice as a Model of Metastatic Liver Cancer with Fenestra LC and Siemens’ MicroCAT II Scanner. Available at: http://www.art.ca/docs/products/fenestra/PMK-UG010-E02%20Imaging%20Hepatic%20CT-26%20Tumors%20with%20Fenestra%20LC.pdf . Accessed July 17, 2013.