Rationale and Objectives
Knowledge of urinary stone composition can guide therapeutic intervention for patients with calcium oxalate (CaOx) or hydroxyapatite (HA) stones. In this study, we determined the accuracy of noninvasive differentiation of these two stone types using dual-energy CT (DECT) and urine supersaturation (SS) and pH values.
Materials and Methods
Patients who underwent clinically indicated DECT scanning for stone disease and subsequent surgical intervention were enrolled. Stone composition was determined using infrared spectroscopy. DECT images were processed using custom-developed software that evaluated the ratio of CT numbers between low- and high-energy images. Clinical information, including patient age, gender, and urine pH and supersaturation profile, was obtained from electronic medical records. Simple and multiple logistic regressions were used to determine if the ratio of CT numbers could discriminate CaOx from HA stones alone or in conjunction with urine supersaturation and pH.
Results
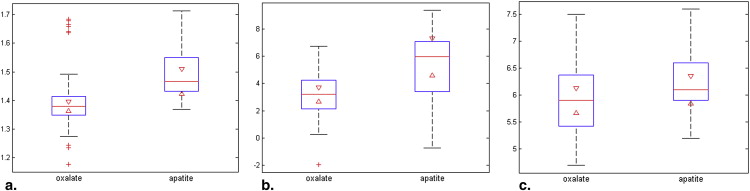
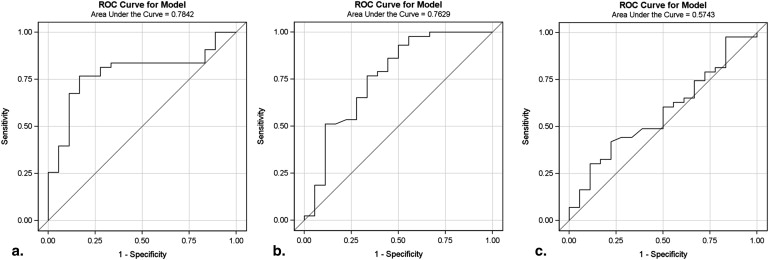
Urinary stones (CaOx n = 43, HA n = 18) from 61 patients were included in this study. In a univariate model, DECT data, urine SS-HA, and urine pH had an area under the receiver operating characteristic curve of 0.78 (95% confidence interval [CI] 0.66–0.91, P = .016), 0.76 (95% CI 0.61–0.91, P = .003), and 0.60 (95% CI 0.44–0.75, P = .20), respectively, for predicting stone composition. The combination of CT data and the urinary SS-HA had an area under the receiver operating characteristic curve of 0.79 (95% CI 0.66–0.92, P = .007) for correctly differentiating these two stone types.
Conclusions
DECT differentiated between CaOx and HA stones similarly to SS-HA, whereas pH was a poor discriminator. The combination of DECT and urine SS or pH data did not improve this performance.
Urinary tract stones (urolithiasis) are one of the most common causes of acute and chronic pain causing significant morbidity. The lifetime incidence in developed countries is about 10% to 15%, with a relapse rate of 50% in 10 years and 75% in 20 years . One of the key factors for optimizing the management of patients with urolithiasis is the chemical composition of the stone . Medical treatment, preoperative evaluation, and the strategy for recurrence prevention are all influenced by stone type. For example, because uric acid stones can dissolve in urine of higher pH, alkalization of the urine is often effective. Extracorporeal shock wave lithotripsy is commonly used for stone removal , but certain firm stones, such as brushite and some calcium oxalate (CaOx) stones, resist fragmentation. Overall, unnecessary procedures can potentially be avoided if the chemical composition is readily known in the initial stage of patient evaluation .
About 80% of stones are composed of CaOx and/or hydroxyapatite (HA) . These two stone types form under different physiochemical conditions, making the differentiation between the two stone types desirable for both research and treatment purposes . Twenty-four-hour urine collection used to measure key components in the urine is the most common method to assess kidney stone risk factors. For example, CaOx stones can form under a wide range of physiologic urinary pH conditions, while HA stones tend to form in alkaline urine . Urinary supersaturation (SS) calculated from the composition of a twenty-four-hour urine collection also correlates with the crystalline component of the stones passed . Therefore, physicians rely largely on urine pH and urine SS profile to predict stone composition when a previous stone analysis is not available.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Get Radiology Tree app to read full this article<
Patient Inclusion
Get Radiology Tree app to read full this article<
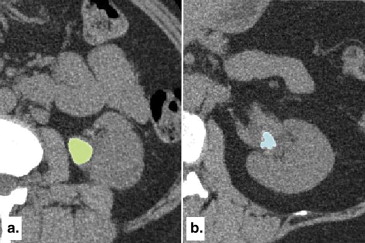
Clinical CT Data Acquisition and Image Processing
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Clinical Lab Urine Evaluations
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Dual-energy CTR, SS-HA, SS-CaOx, and Urine pH of CaOx and HA Stone Patients
CaOx HA_P_ Value Mean ± SD Range Mean ± SD Range CTR 1.40 ± 0.13 1.17 to 1.68 1.49 ± 0.11 1.35 to 1.71 .016 SS-HA, D.G. 3.29 ± 1.95 −1.94 to 6.72 5.43 ± 2.67 −0.74 to 9.36 <.01 SS-CaOx, D.G. 1.85 ± 0.57 0.29 to 2.86 1.68 ± 0.64 0.45 to 2.54 .32 Urine pH 6.0 ± 0.7 4.7 to 7.5 6.2 ± 0.6 5.2 to 7.6 .20
CaOx, calcium oxalate; CTR, computed tomography number ratio; D.G., delta gibbs; HA, hydroxyapatite; SD, standard deviation; SS, supersaturation.
Table 2
Summary of Statistical Analysis Result Using One or Combination of Variables as Predictor(s) of Stone Type
Variable(s) Area Under ROC Curve (C-statistic) 95% Confidence Interval_P_ Value CTR 0.78 0.66–0.91 .016 SS-HA 0.76 0.61–0.91 .003 SS-CaOx 0.57 0.42–0.73 .32 Urine pH 0.60 0.44–0.75 .20 CTR + SS-HA 0.79 0.66–0.92 .007 ∗ CTR + urine pH 0.77 0.65–0.88 .02 ∗ CTR + SS-HA + urine pH 0.80 0.67–0.92 .07 ∗
CaOx, calcium oxalate; CTR, computed tomography number ratio; HA, hydroxyapatite; SS, supersaturation.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion and conclusion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
References
1. Pak C.Y.: Kidney stones. Lancet 1998; 351: pp. 1797-1801.
2. Moe O.W.: Kidney stones: pathophysiology and medical management. Lancet 2006; 367: pp. 333-344.
3. Barcelo P., Wuhl O., Servitge E., et. al.: Randomized double-blind study of potassium citrate in idiopathic hypocitraturic calcium nephrolithiasis. J Urol 1993; 150: pp. 1761-1764.
4. Klee L.W., Brito C.G., Lingeman J.E.: The clinical implications of brushite calculi. J Urol 1991; 145: pp. 715-718.
5. Daudon M., Donsimoni R., Hennequin C., et. al.: Sex- and age-related composition of 10 617 calculi analyzed by infrared spectroscopy. Urol Res 1995; 23: pp. 319-326.
6. Pak C.Y., Poindexter J.R., Adams-Huet B., et. al.: Predictive value of kidney stone composition in the detection of metabolic abnormalities. Am J Med 2003; 115: pp. 26-32.
7. Coe F.L., Evan A., Worcester E.: Kidney stone disease. J Clin Invest 2005; 115: pp. 2598-2608.
8. Parks J.H., Worcester E.M., Coe F.L., et. al.: Clinical implications of abundant calcium phosphate in routinely analyzed kidney stones. Kidney Int 2004; 66: pp. 777-785.
9. Parks J.H., Coward M., Coe F.L.: Correspondence between stone composition and urine supersaturation in nephrolithiasis. Kidney Int 1997; 51: pp. 894-900.
10. Asplin J., Parks J., Lingeman J., et. al.: Supersaturation and stone composition in a network of dispersed treatment sites. J Urol 1998; 159: pp. 1821-1825.
11. Gault M.H., Parfrey P.S., Robertson W.G.: Idiopathic calcium phosphate nephrolithiasis. Nephron 1988; 48: pp. 265-273.
12. Smith R.C., Verga M., McCarthy S., et. al.: Diagnosis of acute flank pain: value of unenhanced helical CT. AJR Am J Roentgenol 1996; 166: pp. 97-101.
13. Primak A.N., Fletcher J.G., Vrtiska T.J., et. al.: Noninvasive differentiation of uric acid versus non-uric acid kidney stones using dual-energy CT. Acad Radiol 2007; 14: pp. 1441-1447.
14. Graser A., Johnson T.R., Bader M., et. al.: Dual energy CT characterization of urinary calculi: initial in vitro and clinical experience. Invest Radiol 2008; 43: pp. 112-119.
15. Matlaga B.R., Kawamoto S., Fishman E.: Dual source computed tomography: a novel technique to determine stone composition. Urology 2008; 72: pp. 1164-1168.
16. Boll D.T., Patil N.A., Paulson E.K., et. al.: Renal stone assessment with dual-energy multidetector CT and advanced postprocessing techniques: improved characterization of renal stone composition–pilot study. Radiology 2009; 250: pp. 813-820.
17. Qu M., Ramirez-Giraldo J.C., Leng S., et. al.: Dual-Energy Dual-Source CT With Additional Spectral Filtration Can Improve the Differentiation of Non-Uric Acid Renal Stones: An Ex Vivo Phantom Study. Am J Roentgenol 2011; 196: pp. 1279-1287.
18. Qu M., Jaramillo-Alvarez G., Ramirez-Giraldo J.C., et. al.: Urinary stone differentiation in patients with large body size using dual-energy dual-source computed tomography. Eur Radiol 2012;
19. Krambeck A.E., Khan N.F., Jackson M.E., et. al.: Inaccurate reporting of mineral composition by commercial stone analysis laboratories: implications for infection and metabolic stones. J Urol 2010; 184: pp. 1543-1549.
20. Werness P.G., Brown C.M., Smith L.H., et. al.: EQUIL2: a BASIC computer program for the calculation of urinary saturation. J Urol 1985; 134: pp. 1242-1244.
21. Schubert G.: Stone analysis. Urol Res 2006; 34: pp. 146-150.
22. Levy F.L., Adams-Huet B., Pak C.Y.: Ambulatory evaluation of nephrolithiasis: an update of a 1980 protocol. Am J Med 1995; 98: pp. 50-59.
23. Lingeman J., Kahnoski R., Mardis H., et. al.: Divergence between stone composition and urine supersaturation: clinical and laboratory implications. J Urol 1999; 161: pp. 1077-1081.
24. Coe F.L., Evan A.P., Lingeman J.E., et. al.: Plaque and deposits in nine human stone diseases. Urol Res 2010; 38: pp. 239-247.
25. Evan A.P., Lingeman J.E., Coe F.L., et. al.: Randall’s plaque of patients with nephrolithiasis begins in basement membranes of thin loops of Henle. J Clin Investig 2003; 111: pp. 607-616.
26. Evan A.P., Lingeman J., Coe F., et. al.: Renal histopathology of stone-forming patients with distal renal tubular acidosis. Kidney Int 2007; 71: pp. 795-801.
27. Evan A.P.: Physiopathology and etiology of stone formation in the kidney and the urinary tract. Pediatr Nephrol 2010; 25: pp. 831-841.
28. Mandel N., Mandel I., Fryjoff K., et. al.: Conversion of calcium oxalate to calcium phosphate with recurrent stone episodes. J Urol 2003; 169: pp. 2026-2029.
29. Krambeck A.E., Handa S.E., Evan A.P., et. al.: Brushite stone disease as a consequence of lithotripsy?. Urol Res 2010; 38: pp. 293-299.
30. Yu L., Primak A.N., Liu X., et. al.: Image quality optimization and evaluation of linearly mixed images in dual-source, dual-energy CT. Med Phys 2009; 36: pp. 1019-1024.