Rationale and Objectives
The aim of this study was to quantify, using diffusion-weighted magnetic resonance imaging, the microscopic rate of water diffusion in pure germinoma and to determine whether or not the apparent diffusion coefficient (ADC) values correlated with the different histologic components.
Materials and Methods
A retrospective analysis of echoplanar diffusion-weighted magnetic resonance images was conducted on 10 patients with 11 germinoma lesions. All images were obtained using 1.5-T magnets with a b value of 1000 s/mm 2 . Regions of interest were drawn separately within the solid and the cystic or necrotic components of each germinoma, as well as within the normal gray and white matter of the respective cases, to calculate ADCs. The diffusion characteristics of the germinomas were assessed using mean and normalized ADC values. Histologic samples from all cases were blindly reviewed and then correlated with the ADC values.
Results
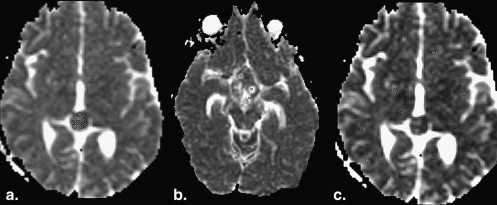
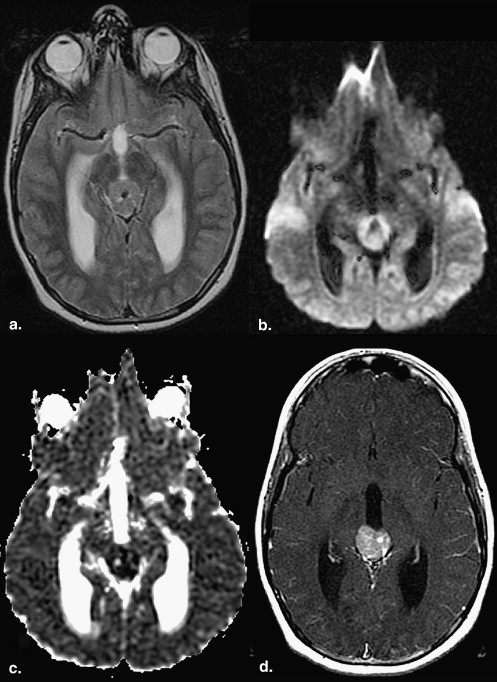
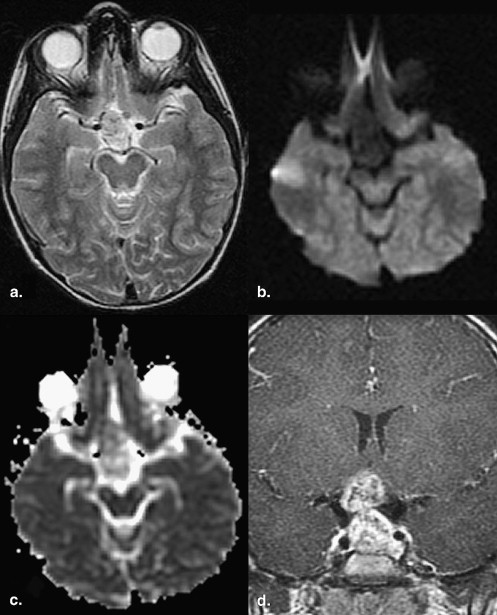
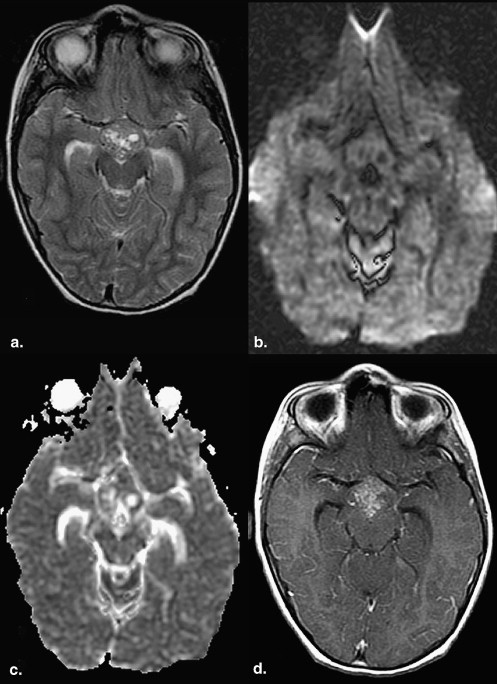
Data are expressed as mean ± standard error. Evaluation of the solid components revealed that 36% of germinomas (4 of 11) had predominantly restricted diffusion (ADC solid , 694.71 × 10 −6 ± 74.54 × 10 −6 s/mm 2 ; ADC ratio, 0.84 ± 0.07) compared to normal brain. The majority (55% [6 of 11]) had normal diffusion (ADC solid , 947.64 × 10 −6 ± 54.38 × 10 −6 s/mm 2 ; ADC ratio, 1.14 ± 0.10). Only 9% (1 of 11) had increased diffusion (ADC solid , 1172.30 × 10 −6 ± 48.52 × 10 −6 s/mm 2 ; ADC ratio, 1.67 ± 0.16). The cystic and necrotic components had a mean ADC ratio of 2.55 ± 0.25. There was no significant correlation between the histologic components and the ADC values of germinomas.
Conclusions
The vast majority of germinomas demonstrated predominantly restricted (36%) or normal (55%) diffusion. The histologic components were not correlated with the ADC values.
Primary intracranial germinomas are rare tumors of children and young adults . They make up the bulk of central nervous system (CNS) germ cell tumors and account for approximately 0.06% to 2.24% of all intracranial neoplasms . Like other germ cell tumors, germinomas are situated in the midline, with the majority of lesions occurring in the pineal and suprasellar regions. Histologically, they are characterized by variable proportions of cellular sheets or lobules of uniform germinoma cells with large round nuclei, prominent nucleoli, and clear cytoplasm interspersed by septal bands of connective tissue rich in capillaries, lymphocytes, and at times granulomas . Cystic degeneration and necrosis may be features of germinoma. Necrosis can be in the form of single cells, small microscopic foci, or in the most extreme cases, geographic zones.
Magnetic resonance (MR) imaging plays a vital role in the diagnosis of germ cell tumors . Diffusion-weighted (DW) imaging is a fairly recently developed MR sequence that has been used for the evaluation of neurologic diseases . With fast echoplanar technology, DW imaging has become widely clinically available and is part of the routine brain imaging protocol at our institution. It is highly resistant to motion, and imaging times are <30 seconds. DW imaging provides image contrast that is dependent on the molecular (Brownian) motion of water . The apparent diffusion coefficient (ADC) is a rotationally invariant measurement of the amount of total diffusion within a given tissue , and quantification of the degree of water motion requires ADC maps . The rate of molecular water diffusion as depicted on DW imaging may be altered in various diseases . Although DW imaging has had its major impact in the evaluation of hyperacute infarctions , it is assuming an increasingly important role in the evaluation of other intracranial pathologies, such as infections and neoplasms . DW imaging has been proposed as a mechanism for producing contrast in the demarcation of different brain neoplasms . Recent studies have demonstrated hyperintense DW imaging signal in certain primary CNS neoplasms, such as lymphoma, pineoblastoma, medulloblastoma, high-grade gliomas , and two germinoma cases . However, the single study that investigated germinoma included only two cases and did not report ADC signal or value. The purpose of this study was to quantify the diffusivity of germinoma as reflected by ADC values and to determine if the ADC values correlated with the various histologic components. We hypothesized that the ADC values would correlate with the different histologic components.
Materials and methods
Patients
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Imaging Protocol
Get Radiology Tree app to read full this article<
Quantitative Analysis of the ADC Maps for Each Germinoma
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Analysis of Histopathology
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Solid Components of Germinoma
Get Radiology Tree app to read full this article<
Table 1
Summary of ADC Values (× 10 −6 s/mm 2 ) by Individual Cases
Case Germinoma Solid Component Uninvolved Brain Germinoma Cystic/Necrotic Component_P_ ∗ Diffusion Pattern † Solid/Uninvolved Brain Cystic/Necrotic/Uninvolved Brain 1 747.35 ± 26.00 809.72 ± 15.37 920.35 ± 0.66 .0473 Restricted 0.92 ± 0.04 1.14 ± 0.02 2 667.66 ± 30.56 820.18 ± 9.52 1296.88 ± 156.60 .0009 Restricted 0.81 ± 0.04 1.58 ± 0.19 3 656.89 ± 14.97 833.71 ± 18.28 1478.06 ± 46.03 <.0001 Restricted 0.79 ± 0.02 1.77 ± 0.07 4 714.34 ± 8.24 834.14 ± 16.25 NA <.0001 Restricted 0.86 ± 0.02 NA 5 984.96 ± 67.63 819.85 ± 9.83 2833.77 ± 169.80 .0705 Normal 1.20 ± 0.08 3.46 ± 0.21 6 944.69 ± 68.90 849.57 ± 22.15 2545.19 ± 264.61 .0934 Normal 1.11 ± 0.09 3.00 ± 0.32 7 849.43 ± 31.92 870.99 ± 29.27 NA .6518 Normal 0.98 ± 0.05 NA 8 843.03 ± 13.69 844.00 ± 13.33 2330.39 ± 138.98 .9599 Normal 1.00 ± 0.02 2.76 ± 0.17 9 794.16 ± 15.95 823.11 ± 12.07 2540.03 ± 32.63 .1724 Normal 0.96 ± 0.02 3.09 ± 0.06 10 850.90 ± 20.57 821.14 ± 13.34 2726.29 ± 215.22 .2289 Normal 1.04 ± 0.03 3.32 ± 0.27 11 1172.30 ± 48.52 845.66 ± 10.72 2452.06 ± 210.18 .0008 Increased 1.39 ± 0.06 2.90 ± 0.25
ADC, apparent diffusion coefficient.
Data are expressed as mean ± standard error. Standard errors in columns 7 and 8 were calculated from those in columns 2 to 4 using Tylor’s expansion method.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Cystic and Necrotic Components of Germinomas
Get Radiology Tree app to read full this article<
Table 2
Comparison of Apparent Diffusion Coefficient Values (× 10 −6 s/mm 2 )
Quantitative Diffusion Pattern ∗ Tissue Type Restricted Normal Increased Solid germinoma 694.71 ± 74.54 aA 947.64 ± 54.38 bD 1408.44 ± 131.64 cF Uninvolved brain 828.62 ± 8.56 dB 833.74 ± 6.46 dD 845.66 ± 16.68 dF Cystic/necrotic germinoma 2574.16 ± 125.42 eC 1875.84 ± 247.16 eE 2452.06 ± 210.18 eF Solid/uninvolved brain 0.84 ± 0.09 1.14 ± 0.07 1.67 ± 0.16 Cystic/necrotic/uninvolved brain 3.11 ± 0.15 2.25 ± 0.30 2.90 ± 0.26
Data are expressed as mean ± standard error. Standard errors in rows 5 and 6 were estimated from means and standard errors in rows 2 to 4 using Tylor’s expansion method.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Normal Gray and White Matter (Uninvolved Brain)
Get Radiology Tree app to read full this article<
Histologic Analysis of Germinoma
Get Radiology Tree app to read full this article<
Table 3
Comparison of Diffusion Pattern and Histopathologic Results
Quantitative Diffusion Pattern Component of Germinoma Classification Restricted Normal Increased_P_ Cystic/necrosis Absent 3 3 0 .55 Present 1 3 1 Connective tissue Low 0 4 0 .11 High 3 2 1 Granuloma Absent 0 4 1 .10 Present 3 2 0 Lymphocytic infiltrate Mild 0 1 1 .09 Intense 3 5 0 Cellularity <25% 0 0 0 .61 25%–50% 0 0 0 50%–75% 2 3 1 >75% 1 3 0
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Solid Components of Germinoma
Get Radiology Tree app to read full this article<
Germinomas with Predominantly Restricted Diffusion
Get Radiology Tree app to read full this article<
Germinomas with Predominantly Normal Diffusion
Get Radiology Tree app to read full this article<
Germinomas with Predominantly Increased Diffusion
Get Radiology Tree app to read full this article<
Cystic and Necrotic Components of Germinoma
Get Radiology Tree app to read full this article<
Analysis of Normal Gray and White Matter
Get Radiology Tree app to read full this article<
Study Limitations
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Kleihues P., Cavenee W.K.: International Agency for Research on Cancer. Pathology and genetics of tumours of the nervous system.2000.IARC PressLyon, France
2. Horowitz M.B., Hall W.A.: Central nervous system germinomas. A review. Arch Neurol 1991; 48: pp. 652-657.
3. Salzman K.L., Rojiani A.M., Buatti J., et. al.: Primary intracranial germ cell tumors: clinicopathologic review of 32 cases. Pediatr Pathol Lab Med 1997; 17: pp. 713-727.
4. Chang T., Teng M.M., Guo W.Y., Sheng W.C.: CT of pineal tumors and intracranial germ-cell tumors. AJNR Am J Neuroradiol 1989; 10: pp. 1039-1044.
5. Higano S., Takahashi S., Ishii K., Matsumoto K., Ikeda H., Sakamoto K.: Germinoma originating in the basal ganglia and thalamus: MR and CT evaluation. AJNR Am J Neuroradiol 1994; 15: pp. 1435-1441.
6. Kim D.I., Yoon P.H., Ryu Y.H., Jeon P., Hwang G.J.: MRI of germinomas arising from the basal ganglia and thalamus. Neuroradiology 1998; 40: pp. 507-511.
7. Konno S., Oka H., Utsuki S., et. al.: Germinoma with a granulomatous reaction. Problems of differential diagnosis. Clin Neuropathol 2002; 21: pp. 248-251.
8. Koren A.: Bifocal primary intracranial germinoma in a child. Case report. Radiol Oncol 2001; 35: pp. 179-183.
9. Ballesteros M.D., Duran A., Arrazola J., et. al.: Primary intrasellar germinoma with synchronous pineal tumor. Neuroradiology 1997; 39: pp. 860-862.
10. Schaefer P.W., Grant P.E., Gonzalez R.G.: Diffusion-weighted MR imaging of the brain. Radiology 2000; 217: pp. 331-345.
11. Le Bihan D.: Molecular diffusion nuclear magnetic resonance imaging. Magn Reson Q 1991; 7: pp. 1-30.
12. Castillo M., Mukherji S.K.: Diffusion-weighted imaging in the evaluation of intracranial lesions. Semin Ultrasound CT MR 2000; 21: pp. 405-416.
13. Le Bihan D., Breton E., Lallemand D., Aubin M.L., Vignaud J., Laval-Jeantet M.: Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 1988; 168: pp. 497-505.
14. DeLano M.C., Cooper T.G., Siebert J.E., Potchen M.J., Kuppusamy K.: High-b-value diffusion-weighted MR imaging of adult brain: image contrast and apparent diffusion coefficient map features. AJNR Am J Neuroradiol 2000; 21: pp. 1830-1836.
15. Cartes-Zumelzu F.W., Stavrou I., Castillo M., Eisenhuber E., Knosp E., Thurnher M.M.: Diffusion-weighted imaging in the assessment of brain abscesses therapy. AJNR Am J Neuroradiol 2004; 25: pp. 1310-1317.
16. Guzman R., Barth A., Lovblad K.O., et. al.: Use of diffusion-weighted magnetic resonance imaging in differentiating purulent brain processes from cystic brain tumors. J Neurosurg 2002; 97: pp. 1101-1107.
17. Hartmann M., Jansen O., Heiland S., Sommer C., Munkel K., Sartor K.: Restricted diffusion within ring enhancement is not pathognomonic for brain abscess. AJNR Am J Neuroradiol 2001; 22: pp. 1738-1742.
18. Desprechins B., Stadnik T., Koerts G., Shabana W., Breucq C., Osteaux M.: Use of diffusion-weighted MR imaging in differential diagnosis between intracerebral necrotic tumors and cerebral abscesses. AJNR Am J Neuroradiol 1999; 20: pp. 1252-1257.
19. Tung G.A., Evangelista P., Rogg J.M., Duncan J.A.: Diffusion-weighted MR imaging of rim-enhancing brain masses: is markedly decreased water diffusion specific for brain abscess?. AJR Am J Roentgenol 2001; 177: pp. 709-712.
20. Guo A.C., Cummings T.J., Dash R.C., Provenzale J.M.: Lymphomas and high-grade astrocytomas: comparison of water diffusibility and histologic characteristics. Radiology 2002; 224: pp. 177-183.
21. Brunberg J.A., Chenevert T.L., McKeever P.E., et. al.: In vivo MR determination of water diffusion coefficients and diffusion anisotropy: correlation with structural alteration in gliomas of the cerebral hemispheres. AJNR Am J Neuroradiol 1995; 16: pp. 361-371.
22. Eis M., Els T., Hoehn-Berlage M.: High resolution quantitative relaxation and diffusion MRI of three different experimental brain tumors in rat. Magn Reson Med 1995; 34: pp. 835-844.
23. Krabbe K., Gideon P., Wagn P., Hansen U., Thomsen C., Madsen F.: MR diffusion imaging of human intracranial tumours. Neuroradiology 1997; 39: pp. 483-489.
24. Tien R.D., Felsberg G.J., Friedman H., Brown M., MacFall J.: MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echo-planar pulse sequences. AJR Am J Roentgenol 1994; 162: pp. 671-677.
25. Okamoto K., Ito J., Ishikawa K., Sakai K., Tokiguchi S.: Diffusion-weighted echo-planar MR imaging in differential diagnosis of brain tumors and tumor-like conditions. Eur Radiol 2000; 10: pp. 1342-1350.
26. Edwards M.S., Hudgins R.J., Wilson C.B., Levin V.A., Wara W.M.: Pineal region tumors in children. J Neurosurg 1988; 68: pp. 689-697.
27. Liang L., Korogi Y., Sugahara T., et. al.: MRI of intracranial germ-cell tumours. Neuroradiology 2002; 44: pp. 382-388.
28. Matsumura N., Kurimoto M., Endo S., Fukuda O., Takaku A.: Intracranial germinoma associated with Down’s syndrome. Report of 2 cases. Pediatr Neurosurg 1998; 29: pp. 199-202.
29. Moon W.K., Chang K.H., Han M.H., Kim I.O.: Intracranial germinomas: correlation of imaging findings with tumor response to radiation therapy. AJR Am J Roentgenol 1999; 172: pp. 713-716.
30. Osborn A.G.: Diagnostic imaging. Brain.2004.AmirsysSalt Lake City, UT
31. Gupta R.K., Sinha U., Cloughesy T.F., Alger J.R.: Inverse correlation between choline magnetic resonance spectroscopy signal intensity and the apparent diffusion coefficient in human glioma. Magn Reson Med 1999; 41: pp. 2-7.
32. Oppenheim C., Grandin C., Samson Y., et. al.: Is there an apparent diffusion coefficient threshold in predicting tissue viability in hyperacute stroke?. Stroke 2001; 32: pp. 2486-2491.
33. Sugahara T., Korogi Y., Kochi M., et. al.: Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 1999; 9: pp. 53-60.
34. Castillo M., Smith J.K., Kwock L., Wilber K.: Apparent diffusion coefficients in the evaluation of high-grade cerebral gliomas. AJNR Am J Neuroradiol 2001; 22: pp. 60-64.
35. Camacho D.L., Smith J.K., Castillo M.: Differentiation of toxoplasmosis and lymphoma in AIDS patients by using apparent diffusion coefficients. AJNR Am J Neuroradiol 2003; 24: pp. 633-637.
36. Helenius J., Soinne L., Perkio J., et. al.: Diffusion-weighted MR imaging in normal human brains in various age groups. AJNR Am J Neuroradiol 2002; 23: pp. 194-199.
37. Maier S.E., Bogner P., Bajzik G., et. al.: Normal brain and brain tumor: multicomponent apparent diffusion coefficient line scan imaging. Radiology 2001; 219: pp. 842-849.
38. Chenevert T.L., McKeever P.E., Ross B.D.: Monitoring early response of experimental brain tumors to therapy using diffusion magnetic resonance imaging. Clin Cancer Res 1997; 3: pp. 1457-1466.