Rationale and Objectives
Chemotherapy has many side effects on breast cancer patients, including cognition and other brain functions impairment, which can be studied using functional magnetic resonance imaging (fMRI). Our study aimed at investigating the executive function alternations of breast cancer patients after chemotherapy using resting-state fMRI.
Materials and Methods
This study included 32 breast cancer patients (BC group) and 24 control subjects (HC group). The functional connectivity of the dorsolateral prefrontal cortex (DLPFC) of the two groups was calculated from the resting-state fMRI data, and the correlation between the strength of the right DLPFC’s connectivity and the behavior performance was analyzed with two-tailed Pearson correlative analysis.
Results
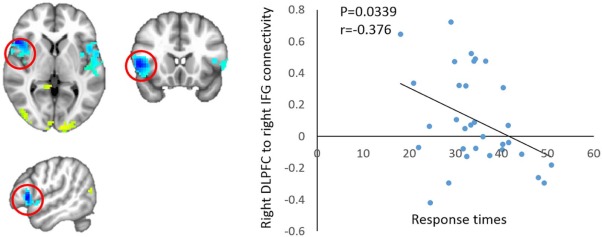
Evaluation of the capability of processing various complex cognition events showed that the executive function of the BC group was impaired after chemotherapy in comparison with the HC group. The functional connectivities of the right DLPFC with the right inferior frontal gyrus, right medial frontal gyrus, and left superior temporal gyrus in the BC group were significantly decreased in comparison with those in the HC group, respectively. The executive deficits were found correlated with the functional connectivity between the right DLPFC and the right inferior frontal gyrus. Meantime, the functional connectivity from the right DLPFC to the right middle temporal gyrus and the precuneus was compensatorily increased in the BC group, respectively.
Conclusions
These findings suggest that breast cancer patients after chemotherapy demonstrate executive control impairment, and provide evidence that the observed defects are correlated with alternations in the executive network of the brain.
Introduction
Breast cancer is the most common cancer in women . Most of the patients diagnosed of breast cancer are treated with surgery and combined with chemotherapy or endocrine therapy. However, with the increase of survival probability, the side effects of chemotherapy and endocrine therapy are reported frequently . Chemotherapy is commonly applied in cancer treatment; nevertheless, its central nervous system toxicity affects long-term quality of life .
Chemotherapy influences the central nervous system possibly through triggering cell death, increasing oxidative stress and microglia activity, suppressing hippocampal neurogenesis, and decreasing the neurotrophic factors and hippocampal catecholamines . Previous studies indicate that breast cancer patients show cognitive impairments after chemotherapy, particularly in the capability of memory, attention, executive controlling, and information processing . The executive function mainly includes information processing, problem solving, planning, and execution . Central executive network, which is responsible for the executive function, mainly involves the frontal lobe and part of the parietal and temporal lobes . The prefrontal cortex (PFC) comprises the frontal part of the frontal lobe and connects several neocortical areas that send and receive projections from almost all of the cortical sensory systems, motor systems, and numerous subcortical structures . Clinical neuropsychologic trials indicate that the PFC plays a central role in the cognitive control behavior. Correspondingly, patients with PFC damage show impaired judgment, organization, and decision-making capability, as well as neuropsychologic disinhibition and intellectual difficulties .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and Methods
Participants
Get Radiology Tree app to read full this article<
Table 1
Summary of Characteristics and Neuropsychologic Tests of Patients and Healthy Controls Recruited for This Study
Variable BC Group ( n = 32) HC Group ( n = 24)t Value_P_ value Mean (SD) Mean (SD) Age (y) 44.19(6.61) 43.25(8.45) 0.450 0.655 Education (y) 10.56(2.63) 10.63(2.26) −0.095 0.924 Fatigue 23.03(3.80) 21.71(4.13) 1.227 0.226 HAMA 4.47(1.24) 4.42(1.25) 0.155 0.878 HAMD 4.91(1.20) 4.54(1.38) 0.329 0.744 MoCA 25.28(1.42) 25.50(1.29) −0.603 0.549 Stroop Word Test (s) 16.38(3.69) 14.07(2.65) 2.731 0.009 Stroop Color Test (s) 21.73(3.90) 17.99(2.69) 4.238 <0.001 Stroop Interference Test (s) 34.28(7.90) 29.72(5.29) 2.586 0.012
BC, breast cancer; HAMA, Hamilton Anxiety Rating Scale; HAMD, Hamilton Depression Rating Scale; HC, healthy controls; MoCA, Montreal Cognitive Assessment test; SD, standard deviation.
Get Radiology Tree app to read full this article<
Neuropsychologic Background Tests
Get Radiology Tree app to read full this article<
Stroop Color-word Test
Get Radiology Tree app to read full this article<
Image Acquisition
Get Radiology Tree app to read full this article<
Image Preprocessing
Get Radiology Tree app to read full this article<
Functional Connectivity Analysis
Get Radiology Tree app to read full this article<
Group and Correlative Analysis
Get Radiology Tree app to read full this article<
Results
Neuropsychologic Evaluation
Get Radiology Tree app to read full this article<
Alternation of the Central Executive Network Connectivity
Get Radiology Tree app to read full this article<
Table 2
Group Analysis Results of Functional Connectivity of the Right DLPFC
Region (BA) MNI coordinates Side X (mm) Y (mm) Z (mm) Peak (Z value) Superior temporal gyrus (22) L −56 0 0 −2.94 Inferior frontal gyrus (47) R 54 18 2 −3.71 Medial frontal gyrus (6) R 10 2 56 −2.98 Middle temporal gyrus (39) R 52 −70 14 3.08 Precuneus (31) R 6 −52 30 2.51 Declive R 4 −82 −18 2.81
BA, Brodmann area; DLPFC, dorsolateral prefrontal cortex; L, left; MNI, Montreal Neurological Institute; R, right.
The threshold was set to P ≤ 0.05, α ≤ 0.05 (corrected using the Monte Carlo method).
Get Radiology Tree app to read full this article<
Correlation Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Functional Connectivity Reduction of Areas Related to the Executive Network
The Decreased Functional Connectivity Was Correlated to the Worse Neuropsychologic Performance
Get Radiology Tree app to read full this article<
The Decreased Functional Connectivity Was Related to the Executive Network
Get Radiology Tree app to read full this article<
Compensatory Increase of Connectivity Between the Right Middle Temporal Gyrus, Right Precuneus, and Right DLPFC
Get Radiology Tree app to read full this article<
Limitations of This Study
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Ethical Approval
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Fan L., Strasser-Weippl K., Li J.-J., et. al.: Breast cancer in China. Lancet Oncol 2014; 15: pp. e279-e289.
2. Schilder C.M., Seynaeve C., Beex L.V., et. al.: Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: results from the neuropsychological side study of the tamoxifen and exemestane adjuvant multinational trial. J Clin Oncol 2010; 28: pp. 1294-1300.
3. Hershman D.L., Lacchetti C., Dworkin R.H., et. al.: Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2014; 32: pp. 1941-1967.
4. Fardell J.E., Vardy J., Johnston I.N., et. al.: Chemotherapy and cognitive impairment: treatment options. Clin Pharmacol Ther 2011; 90: pp. 366-376.
5. Chen X., Zhu C., Li J., et. al.: Dissociation of decision making under ambiguity and decision making under risk in breast cancer patients receiving adjuvant chemotherapy: a neuropsychological study. Brain Res 2013; 1533: pp. 63-72.
6. Elliott R.: Executive functions and their disorders. Imaging in clinical neuroscience. Br Med Bull 2003; 65: pp. 49-59.
7. Rusnakova S., Daniel P., Chladek J., et. al.: The executive functions in frontal and temporal lobes: a flanker task intracerebral recording study. J Clin Neurophysiol 2011; 28: pp. 30-35.
8. Stretton J., Thompson P.J.: Frontal lobe function in temporal lobe epilepsy. Epilepsy Res 2012; 98: pp. 1-13.
9. Miller E.K., Cohen J.D.: An integrative theory of prefrontal cortex function. Annu Rev Neurosci 2001; 24: pp. 167-202.
10. Stuss D.T., Benson D.F.: Neuropsychological studies of the frontal lobes. Psychol Bull 1984; 95: pp. 3-28.
11. Funahashi S.: Neuronal mechanisms of executive control by the prefrontal cortex. Neurosci Res 2001; 39: pp. 147-165.
12. Bruno J., Hosseini S.M., Kesler S.: Altered resting state functional brain network topology in chemotherapy-treated breast cancer survivors. Neurobiol Dis 2012; 48: pp. 329-338.
13. McDonald B.C., Conroy S.K., Smith D.J., et. al.: Frontal gray matter reduction after breast cancer chemotherapy and association with executive symptoms: a replication and extension study. Brain Behav Immun 2013; 30: pp. S117-S125.
14. de Ruiter M.B., Reneman L., Boogerd W., et. al.: Late effects of high-dose adjuvant chemotherapy on white and gray matter in breast cancer survivors: converging results from multimodal magnetic resonance imaging. Hum Brain Mapp 2012; 33: pp. 2971-2983.
15. Deprez S., Amant F., Yigit R., et. al.: Chemotherapy-induced structural changes in cerebral white matter and its correlation with impaired cognitive functioning in breast cancer patients. Hum Brain Mapp 2011; 32: pp. 480-493.
16. McDonald B.C., Conroy S.K., Ahles T.A., et. al.: Gray matter reduction associated with systemic chemotherapy for breast cancer: a prospective MRI study. Breast Cancer Res Treat 2010; 123: pp. 819-828.
17. Dietrich J., Prust M., Kaiser J.: Chemotherapy, cognitive impairment and hippocampal toxicity. Neuroscience 2015; 309: pp. 224-232.
18. Talati A., Hirsch J.: Functional specialization within the medial frontal gyrus for perceptual go/no-go decisions based on “what,” “when,” and “where” related information: an fMRI study. J Cogn Neurosci 2005; 17: pp. 981-993.
19. de Ruiter M.B., Reneman L., Boogerd W., et. al.: Cerebral hyporesponsiveness and cognitive impairment 10 years after chemotherapy for breast cancer. Hum Brain Mapp 2011; 32: pp. 1206-1219.
20. Clément F., Belleville S.: Effect of disease severity on neural compensation of item and associative recognition in mild cognitive impairment. J Alzheimers Dis 2011; 29: pp. 109-123.
21. Kim M.A., Tura E., Potkin S.G., et. al.: Working memory circuitry in schizophrenia shows widespread cortical inefficiency and compensation. Schizophr Res 2010; 117: pp. 42-51.
22. Ragland J.D., Moelter S.T., Bhati M.T., et. al.: Effect of retrieval effort and switching demand on fMRI activation during semantic word generation in schizophrenia. Schizophr Res 2008; 99: pp. 312-323.
23. Angel L., Bastin C., Genon S., et. al.: Differential effects of aging on the neural correlates of recollection and familiarity. Cortex 2013; 49: pp. 1585-1597.
24. Nasreddine Z.S., Phillips N.A., Bedirian V., et. al.: The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005; 53: pp. 695-699.
25. So W.K.W., Dodgson J., Tai J.W.M.: Fatigue and quality of life among Chinese patients with hematologic malignancy after bone marrow transplantation. Cancer Nurs 2003; 26: pp. 211-219.
26. Lezak M.D.: Neuropsychological assessment in behavioral-toxicology—developing techniques and interpretative issues. Scand J Work Environ Health 1984; 10: pp. 25-29.
27. Rottschy C., Langner R., Dogan I., et. al.: Modelling neural correlates of working memory: a coordinate-based meta-analysis. Neuroimage 2012; 60: pp. 830-846.
28. Simo M., Rifa-Ros X., Rodriguez-Fornells A., et. al.: Chemobrain: a systematic review of structural and functional neuroimaging studies. Neurosci Biobehav Rev 2013; 37: pp. 1311-1321.
29. Pochon J.-B., Levy R., Poline J.-B., et. al.: The role of dorsolateral prefrontal cortex in the preparation of forthcoming actions: an fMRI study. Cereb Cortex 2001; 11: pp. 260-266.
30. Bunge S.A., Dudukovic N.M., Thomason M.E., et. al.: Immature frontal lobe contributions to cognitive control in children: evidence from fMRI. Neuron 2002; 33: pp. 301-311.
31. Aron A.R., Robbins T.W., Poldrack R.A.: Inhibition and the right inferior frontal cortex. Trends Cogn Sci 2004; 8: pp. 170-177.
32. Langenecker S.A., Nielson K.A., Rao S.M.: fMRI of healthy older adults during Stroop interference. Neuroimage 2004; 21: pp. 192-200.
33. Johnstone S.J., Barry R.J., Markovska V., et. al.: Response inhibition and interference control in children with AD/HD: a visual ERP investigation. Int J Psychophysiol 2009; 72: pp. 145-153.
34. Zunini R.A.L., Scherling C., Wallis N., et. al.: Differences in verbal memory retrieval in breast cancer chemotherapy patients compared to healthy controls: a prospective fMRI study. Brain Imaging Behav 2013; 7: pp. 460-477.
35. Hackett T.A.: Anatomical organization of the auditory cortex. J Am Acad Audiol 2008; 19: pp. 774-779.
36. Bigler E.D., Mortensen S., Neeley E.S., et. al.: Superior temporal gyrus, language function, and autism. Dev Neuropsychol 2007; 31: pp. 217-238.
37. Friederici A.D., Rüschemeyer S.-A., Hahne A., et. al.: The role of left inferior frontal and superior temporal cortex in sentence comprehension: localizing syntactic and semantic processes. Cereb Cortex 2003; 13: pp. 170-177.
38. Hoshi E.: Functional specialization within the dorsolateral prefrontal cortex: a review of anatomical and physiological studies of non-human primates. Neurosci Res 2006; 54: pp. 73-84.
39. Friston K.J.: Functional and effective connectivity: a review. Brain Connect 2011; 1: pp. 13-36.
40. Strand F., Forssberg H., Klingberg T., et. al.: Phonological working memory with auditory presentation of pseudo-words—an event related fMRI Study. Brain Res 2008; 1212: pp. 48-54.
41. Dronkers N.F., Wilkins D.P., Van Valin R.D., et. al.: Lesion analysis of the brain areas involved in language comprehension. Cognition 2004; 92: pp. 145-177.
42. Cavanna A.E., Trimble M.R.: The precuneus: a review of its functional anatomy and behavioural correlates. Brain 2006; 129: pp. 564-583.
43. Li Q., Jiang Q., Guo M., et. al.: Grey and white matter changes in children with monocular amblyopia: voxel-based morphometry and diffusion tensor imaging study. Br J Ophthalmol 2013; 97: pp. 524-529.