Rationale and Objectives
This study aims to describe the magnetic resonance imaging (MRI) features of fat necrosis on magnetic resonance mammography, which may downstage a suspicious lesion to a merely benign finding.
Materials and Methods
This prospective study included 82 female patients (mean age 50 years) who were diagnosed to have suspicious lesions by mammography, ultrasonography or both. All patients underwent MRI including diffusion-weighted imaging and spectroscopy. Image postprocessing and analysis included signal intensity, enhancement characteristics, diffusion restriction, and spectroscopic analysis. All patients underwent histopathological analysis for confirmation. Sensitivity, specificity, positive predictive value (PPV), and negative (NPV) predictive value were calculated.
Results
To label a lesion as fat necrosis on MRI analysis, presence of fat signal in a lesion revealed sensitivity of 98.04%, specificity of 100%, PPV of 100%, and NPP of 96.88%, whereas nonenhancement of the lesion itself revealed sensitivity of 96.08%, specificity of 100%, PPV of 100%, and NPP of 93.94%. However, adding both the nonrestriction on diffusion analysis and the lack of tCholine at 3.22 ppm increased the sensitivity and specificity to 100%, as well as PPV of 100% for fat necrosis and hence a NPV for malignancy of 100%.
Conclusions
MRI proved to be of value in differentiating fat necrosis from malignancy based on the molecular composition of fat necrosis, clearly depicted by MRI without the need for invasive confirmation by biopsy.
Introduction
Fat necrosis is a benign non–suppurative inflammatory process of adipose tissue , initially described in the breast in 1920s . It is described as “an innocent lesion” in medical literature labeled as BI-RADS 2, which stands for totally benign breast lesion if it met its classical oil cyst form on mammogram. Nonetheless, it gained its notorious reputation and clinicians thrive to diagnose it accurately, as it is the number one differential diagnosis of an early breast cancer . Before the era of imaging and tissue biopsy, the treatment of choice was a wide resection of the breast .
Even with the advent of the different breast imaging procedures, fat necrosis still displays a wide spectrum of morphologic criteria on the different imaging modalities. This depends on the pathological stage of fat necrosis process, which depends on the balance of fat content and the degree of inflammation and fibrosis of the lesions .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and Methods
Get Radiology Tree app to read full this article<
Patients
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
At the end of the year, we ended up with 82 female patients (mean age ± SD: 50.10 ± 10.55, age ranging between 34 and 76 years), with 58 being suspected for postmanagement recurrence, whereas the rest were newly diagnosed lesions.
Get Radiology Tree app to read full this article<
MRI
Get Radiology Tree app to read full this article<
MRI Protocols and Technique
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Image Post Processing
Get Radiology Tree app to read full this article<
Lesion Evaluation and Interpretation of Examination
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Histopathological Analysis
Get Radiology Tree app to read full this article<
Data and Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
MRI Findings
Morphology and Signal Intensity
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Enhancement Characteristics
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Diffusion-weighted Imaging (DWI) and Spectroscopic Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
BI-RADS Classification
Get Radiology Tree app to read full this article<
Radiologic-Pathologic Correlation of Cases with Fat Necrosis
Morphology and Signal Intensity
Get Radiology Tree app to read full this article<
Table 1
MRI Findings of Pathologically Proven Cases of Fat Necrosis
MRI Characteristics Number of Cases Total T1 intensity Hyperintense 50 51 Hypointense (signal void) 1 T2 fat-suppressed intensity Black hole effect (markedly hypointense) 50 51 Signal void 1 Lesion’s enhancement Enhancing rim with nonenhancing center 38 51 Nonenhancement of both center and surrounding fibrosis 11 Diffuse enhancement 2 Diffusion-weighted imaging Nonrestricted (≥1.5 × 10 −3 mm 2 /s) 42 51 Overlap zone (>1.3 × 10 −3 mm 2 /s) (<1.3 × 10 −3 mm 2 /s) 9 0 Restricted (≤1.0 × 10 −3 mm 2 /s) 0 Spectroscopic analysis Positive (at 3.22 ppm) 0 15 Positive (>3.28 ppm) 4 Negative (at 3.2 ppm and >3.28 ppm) 11
MRI, magnetic resonance imaging.
Get Radiology Tree app to read full this article<
Enhancement Characteristics
Get Radiology Tree app to read full this article<
DWI and Spectroscopic Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Diagnostic Performance
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Tan P.H., Lai L.M., Carrington E.V., et. al.: Fat necrosis of the breast—a review. The Breast 2006; 15: pp. 313-318.
2. Chan L.P., Gee R., Keogh C., et. al.: Imaging features of fat necrosis. AJR Am J Roentgenol 2003; 181: pp. 955-959.
3. Daly C.P., Jaeger B., Sill D.S.: Variable appearances of fat necrosis on breast MRI. AJR Am J Roentgenol 2008; 191: pp. 1374-1380.
4. Taboada J.L., Stephens T.W., Krishnamurthy S., et. al.: The many faces of fat necrosis in the breast. AJR Am J Roentgenol 2009; 192: pp. 815-825.
5. Kerridge W.D., Kryvenko O.N., Thompson A., et. al.: Fat necrosis of the breast: a pictorial review of the mammographic, ultrasound, CT, and MRI findings with histopathologic correlation. Radiol Res Pract 2015; 2015: pp. 613139.
6. Chala L.F., de Barros N., Moraes P., et. al.: Fat necrosis of the breast: mammographic, sonographic, computed tomography, and magnetic resonance imaging findings. Curr Probl Diagn Radiol 2004; 33: pp. 106-126.
7. Sergi Ganau S., Tortajada L., Escribano F., et. al.: The great mimicker: fat necrosis of the breast—magnetic resonance mammography approach. Curr Probl Diagn Radiol 2009; 38: pp. 189-197.
8. Stanwell P., Gluch L., Clark D., et. al.: Specificity of choline metabolites for in vivo diagnosis of breast cancer using 1H MRS at 1.5 T. Eur Radiol 2005; 15: pp. 1037-1043.
9. Sardanelli F., Fausto A., DiLeo G., et. al.: In vivo proton MR spectroscopy of the breast using the total choline peak integral as a marker of malignancy. AJR Am J Roentgenol 2009; 192: pp. 1608-1617.
10. Partridge S.C., DeMartini W.B., Kurland B.F., et. al.: Quantitative diffusion-weighted imaging as an adjunct to conventional breast MRI for improved positive predictive value. AJR Am J Roentgenol 2009; 193: pp. 1716-1722.
11. Kul S., Cansu A., Alhan E., et. al.: Contribution of diffusion weighted imaging to dynamic contrast-enhanced MRI in the characterization of breast tumors. AJR Am J Roentgenol 2011; 196: pp. 210-217.
12. Gonzales M.A., Castañon A.I., Baltar N., et. al.: Diffusion weighted MR imaging assessment after breast conservative treatment. Poster session in ECR 2012 Educational Exhibit. March 1–52012. Vienna, Austria
13. Şahin C., Arıbal E.: The role of apparent diffusion coefficient values in the differential diagnosis of breast lesions in diffusion-weighted MRI. Diagn Interv Radiol 2013; 19: pp. 457-462.
14. Hassan H.H.M., Zahran M.H.M., Hassan H.E., et. al.: Diffusion magnetic resonance imaging of breast lesions: initial experience at Alexandria University. Alexandria J Med 2013; 49: pp. 2652-2672.
15. Hetta W.: Role of diffusion weighted images combined with breast MRI in improving the detection and differentiation of breast lesions. Egypt J Radio Nuc Med 2015; 46: pp. 259-270.
16. Tsushima Y., Takahashi-Taketomi A., Endo K.: Magnetic resonance (MR) differential diagnosis of breast tumors using apparent diffusion coefficient (ADC) on 1.5-T. J Magn Reson Imaging 2009; 30: pp. 249-255.
17. Chansakul T., Lai K.C., Slanetz P.J.: The post-conservation breast: part 2, imaging findings of tumor recurrence and other long-term sequelae. AJR Am J Roentgenol 2012; 98: pp. 331-333.
18. Trimboli R.M., Carbonaro L.A., Cartia F., et. al.: MRI of fat necrosis of the breast: the “black hole” sign at short tau inversion recovery. Eur J Radiol 2012; 81: pp. e573-e579.
19. Sardanelli F., Carbonaro L.A., Montemezzi S., et. al.: Clinical breast MR using MRS or DWI: who is the winner?. Front Oncol 2016; 6: pp. e217.
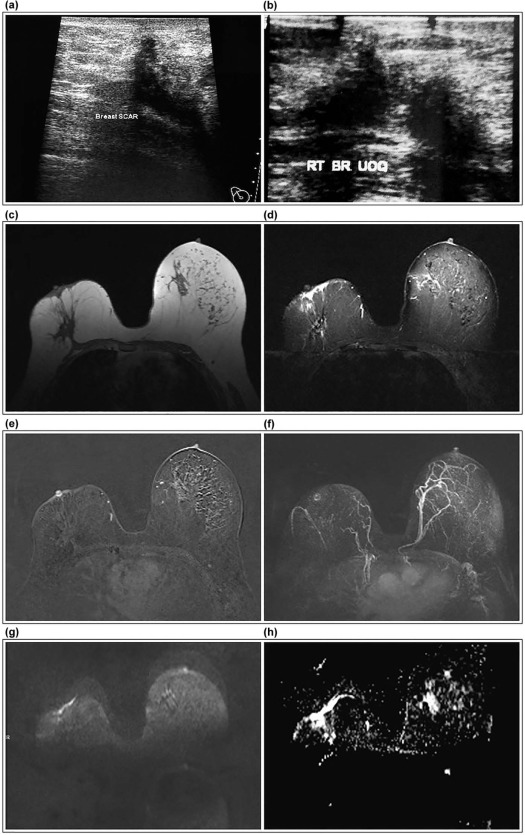
![Figure 2, A 54-year-old female patient with a history of right breast cancer managed surgically followed by chemo- and radiotherapy on follow-up. Mammographic MLO view of the right breast ( a ) revealed upper outer quadrant focal asymmetry, and ultrasound ( b ) identified a suspicious focus within the operative bed. Axial T1 non–fat-suppressed ( c ) and T2 fat-suppressed ( d ) images revealed a markedly hypointense focus on both sequences along the operative scar associated with surrounding edema. No fat signal is identified, still marked signal void of hemosiderin deposition is the most likely cause. Postcontrast subtracted image for mean curve analysis ( e ) revealed focal enhancement of type I b progressive enhancement with delayed washout. No central nonenhancement identified, making the diagnosis equivocal for recurrence; still no choline trace was identified on spectroscopic analysis (f) , with the value of tCholine being zero at the 3.22 ppm point along the scale, endorsing the BI-RADS 3 class assigned to the lesion based on morphology and kinetics analysis and excluding the suspicion of recurrence. Diffusion-weighted image ( g ) with b-value of 800 and ADC map ( h ) showed no evidence of diffusion restriction, confirming the spectroscopic analysis and excluding recurrence. Low power microscopic examination of the lesion ( i ) shows multiple communicating fat cysts, hemorrhage, and a diffuse mixed inflammatory infiltrate (hematoxylin and eosin [H&E] ×100), higher power examination ( j ) shows large fat cyst surrounded by necrosis entangling karyorrhectic debris and a mixed inflammatory infiltrate (H&E × 200). High power examination ( k ) confirms the inflammatory infiltrate formed by large foamy histiocytes (arrows), neutrophils, lymphocytes, and hemosiderin-laden macrophages (H&E × 400).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/FatNecrosisoftheBreast/1_1s20S1076633218300011.jpg)