Rationale and Objectives
Endometriosis is a common women’s health problem. Animal models provide an invaluable tool to study the natural history of endometriosis. We previously have reported that 99m Tc-labeled glutamate peptide-estradiol ( 99m Tc-GAP-EDL) is a useful agent for imaging functional estrogen receptor (ER) via an ER-mediated process. This study was to evaluate the feasibility of using radiolabeled GAP-EDL to image ER-positive (ER +) endometriosis in nonprimate animal models.
Materials and Methods
3-Aminoethyl estradiol (EDL) was conjugated to glutamate peptide (GAP) to yield GAP-EDL. In vitro cellular uptake studies of 99m Tc and 68 Ga-GAP-EDL inhibition with cold estrone were conducted in 13,762 rat mammary tumor cells. To create a rabbit model with endometriosis, part of uterine tissue was dissected and grafted in the peritoneal wall. Eight weeks after surgery, scintigraphic images were obtained after intravenous injection of 99m Tc-GAP-EDL (1 mCi/rabbit, intravenous) at 0.5–2.0 hours, and 68 Ga-GAP-EDL at 45 minutes. We also performed 68 Ga-GAP-EDL blocking study in rabbit model by using tamoxifen. The rabbits were sacrificed and the grafts were excised for histologic examination.
Results
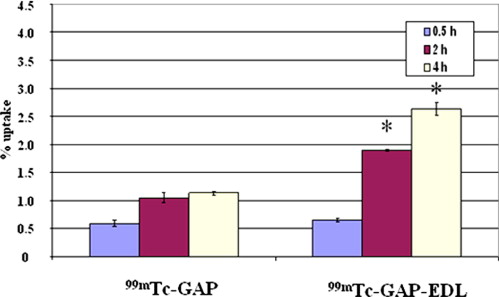
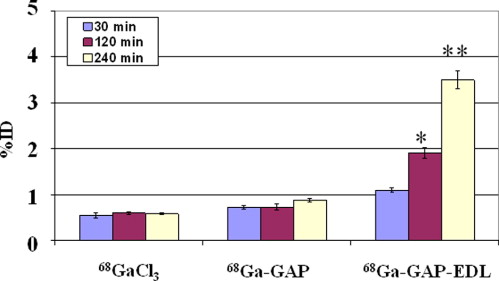
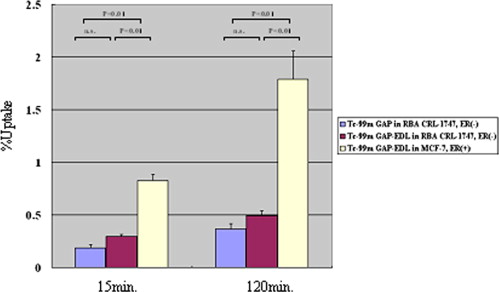
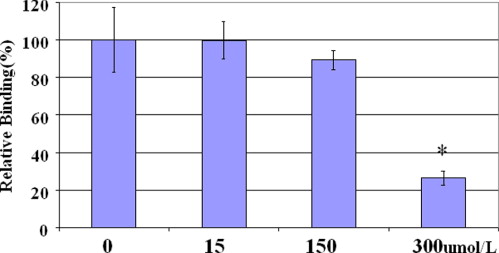
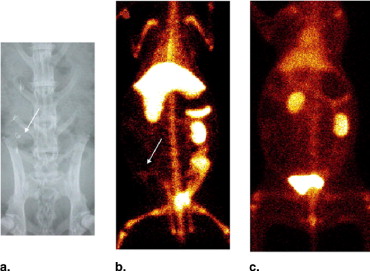
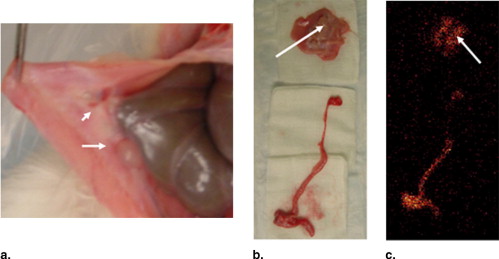
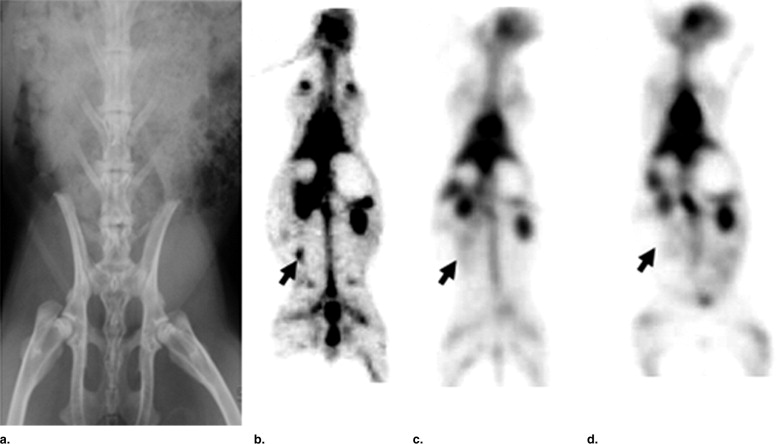
In vitro uptake study of 99m Tc- and 68 Ga-GAP-EDL in 13,762 rat breast cancer cells showed gradually increasing uptake of both tracers. Accumulation of 68 Ga-GAP-EDL in 13,762 cells was inhibited with cold estrone in a dose-dependent manner. In the endometriosis model, the grafted uterine tissue could be visualized by 99m Tc-GAP-EDL. Necropsy was performed at 2.5 hours after injection time. Four follicular endometrial lesions in eight implanted endometrial tissues were detected, and all lesions could be detected by 99m Tc-GAP-EDL. Planar scintigraphy of uterus, ovary and implants of necropsy specimen revealed an increased uptake of 99m Tc-GAP-EDL in comparison with surrounding abdominal wall tissue. Microscopic examinations support that 99m Tc-GAP-EDL was accumulated in the microinvasive endometrial tissue. After blocking with tamoxifen, 68 Ga-GAP-EDL accumulation in the endometrial grafts could not be visualized, and endometrial tissue-to-normal tissue count ratios were statistically higher in a nonblocked image than that in the blocked image.
Conclusions
Endometriosis uptake of radiolabeled GAP-EDL was via an estrogen receptor-mediated process. Radiolabeled-GAP-EDLs are useful agents for imaging endometriosis.
Endometriosis is a common women’s health problem that is characterized by the presence of tissue resembling endometrium outside the uterus. The exact prevalence of endometriosis in the population cannot be ascertained because of the need to perform an invasive procedure to determine who is affected. Nevertheless, estimate range from 2% to 22% in asymptomatic women, 40%–60% in women with dysmenorrhea, and 20%–30% in women being investigated for subfertility ( ). The condition causes painful periods, chronic pelvic pain, and subfertility, which are potentially debilitating; and it affects millions of women worldwide. The diagnosis is made on visual inspection of the pelvis, usually at laparoscopy. The natural history is unknown, and well-controlled experiments are difficult to perform because of the need for repeated surgical procedures to assess endometriotic lesions over time. Thus despite more than 50 years’ research, the cause of endometriosis remains unclear, and treatment options are limited ( ).
Animal models provide an invaluable tool to study risk factors, prevalence, and the natural history of endometriosis. It has been reported that estrogen receptors (ERs) are overexpressed in uterine endometrium and endometriotic lesions of patients with endometriosis. Laparoscopy observation is generally performed to detect endometriosis. The decision to perform laparoscopy on patients with infertility is very complex owing to a number of factors, such as maternal age, semen parameters, tubal patency, pelvic symptoms, insurance coverage, surgical risks, and availability of surgical expertise. Moreover, the procedure is invasive and the information obtained is morphologic and anatomic changes. Thus it would be desirable to develop radiolabeled estradiol for noninvasive diagnosis and monitoring of the therapy for endometriosis.
Get Radiology Tree app to read full this article<
Material and methods
Chemical Analysis
Get Radiology Tree app to read full this article<
Synthesis of 99m Tc and 68 Ga-Glutamate Peptide-Estradiol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
In Vitro Cellular Uptake Studies
Get Radiology Tree app to read full this article<
Gamma Scintigraphic Studies in Rabbits with Endometriosis
Get Radiology Tree app to read full this article<
Positron Emission Tomography Studies in Rabbits with Endometriosis
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Chemistry
Get Radiology Tree app to read full this article<
In Vitro Cellular Uptake Studies
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Gamma Scintigraphic Studies in Rabbits with Endometriosis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Farquhar C.M.: Extracts from the “clinical evidence.”. BMJ 2000; 27: pp. 1449-1452.
2. Murphy A.A.: Clinical aspects of endometriosis. Ann N Y Acad Sci 2002; 955: pp. 1-10.
3. Takahashi N., Yang D.J., Kohanim S., et. al.: Targeted functional imaging of estrogen receptors with (99m)Tc-GAP-EDL. Eur J Nucl Med Mol Imaging 2007; 34: pp. 354-362.
4. Dunselman G.A., Willebrand D., Land J.A., et. al.: A rabbit model of endometriosis. Gynecol Obstet Invest 1989; 27: pp. 29-33.
5. Vercellini P., Fedele L., Aimi G., et. al.: Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Hum Reprod 2007; 22: pp. 266-271.
6. Cornillie F.J., Oosterlynck D., Lauweryns J., et. al.: Deeply infiltrating pelvic endometriosis: histology and clinical significance. Fertil Steril 1990; 53: pp. 978-983.
7. Koninckx P.R., Meuleman C., Demeyere S., et. al.: Suggestive evidence that pelvic endometriosis is a progressive disease, whereas deeply infiltrating endometriosis is associated with pelvic pain. Fertil Steril 1991; 55: pp. 759-765.
8. Ripps B.A., Martin D.C.: Focal pelvic tenderness, pelvic pain and dysmenorrhea in endometriosis. J Reprod Med 1991; 36: pp. 470-472.
9. Vercellini P., Bocciolone L., Vendola N., et. al.: Peritoneal endometriosis: morphologic appearance in women with chronic pelvic pain. J Reprod Med 1991; 36: pp. 533-536.
10. Perper M.M., Nezhat F., Goldstein H., et. al.: Dysmenorrhea is related to the number of implants in endometriosis patients. Fertil Steril 1995; 63: pp. 500-503.
11. Muzii L., Marana R., Pedullà S., et. al.: Correlation between endometriosis-associated dysmenorrhea and the presence of typical or atypical lesions. Fertil Steril 1997; 68: pp. 19-22.
12. Porpora M.G., Koninckx P.R., Piazze J., et. al.: Correlation between endometriosis and pelvic pain. J Am Assoc Gynecol Laparosc 1999; 6: pp. 429-434.
13. Parazzini F., Mais V., Cipriani S., et. al.: Adhesions and pain in women with first diagnosis of endometriosis: results from a cross-sectional study. J Minim Invasive Gynecol 2006; 13: pp. 49-54.
14. Fauconnier A., Chapron C., Dubuisson J.B., et. al.: Relation between pain symptoms and the anatomic location of deep infiltrating endometriosis. Fertil Steril 2002; 78: pp. 719-726.
15. Milingos S., Protopapas A., Kallipolitis G., et. al.: Endometriosis in patients with chronic pelvic pain: is staging predictive of the efficacy of laparoscopic surgery in pain relief?. Gynecol Obstet Invest 2006; 62: pp. 48-54.
16. Story L., Kennedy S.: Animal studies in endometriosis: a review. ILAR J 2004; 45: pp. 132-138.
17. Fedele L., Parazzini F., Bianchi S., et. al.: Stage and localization of pelvic endometriosis and pain. Fertil Steril 1990; 53: pp. 155-158.
18. Mahmood T.A., Templeton A.A., Thomson L., et. al.: Menstrual symptoms in women with pelvic endometriosis. Br J Obstet Gynaecol 1991; 98: pp. 558-563.
19. Marana R., Muzii L., Caruana P., et. al.: Evaluation of the correlation between endometriosis extent, age of the patients and associated symptomatology. Acta Eur Fertil 1991; 22: pp. 209-212.
20. Stout A.L., Steege J.F., Dodson W.C., et. al.: Relationship of laparoscopic findings to self-report of pelvic pain. Am J Obstet Gynecol 1991; 164: pp. 73-79.
21. Fedele L., Bianchi S., Bocciolone L., et. al.: Pain symptoms associated with endometriosis. Obstet Gynecol 1992; 79: pp. 767-769.
22. Matorras R., Rodriguez F., Pijoan J.I., et. al.: Women who are not exposed to spermatozoa and infertile women have similar rates of stage I endometriosis. Fertil Steril 2001; 76: pp. 923-928.
23. Vercellini P., Trespidi L., De Giorgi O., et. al.: Endometriosis and pelvic pain: relation to disease stage and localization. Fertil Steril 1996; 65: pp. 299-304.
24. Stovall D.W., Bowser L.M., Archer D.F., et. al.: Endometriosis associated pelvic pain: evidence for an association between the stage of disease and a history of chronic pelvic pain. Fertil Steril 1997; 68: pp. 13-18.
25. Szendei G., Hernadi Z., Devenyi N., et. al.: Is there any correlation between stages of endometriosis and severity of chronic pelvic pain?. Gynecol Endocrinol 2005; 21: pp. 93-100.
26. Schenken R.S., Asch R.H.: Surgical induction of endometriosis in the rabbit: effects on fertility and concentrations of peritoneal fluid prostaglandins. Fertil Steril 1980; 34: pp. 581-587.
27. Vernon M.W., Wilson E.A.: Studies on the surgical induction of endometriosis in the rat. Fertil Steril 1985; 44: pp. 684-694.
28. Steinleitner A., Lambert H., Suarez M., et. al.: Immunomodulation in the treatment of endometriosis-associated subfertility: use of pentoxifylline to reverse the inhibition of fertilization by surgically induced endometriosis in a rodent model. Fertil Steril 1991; 56: pp. 975-979.