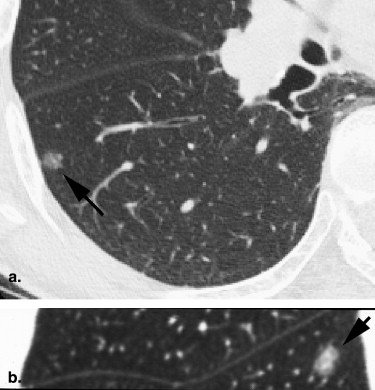
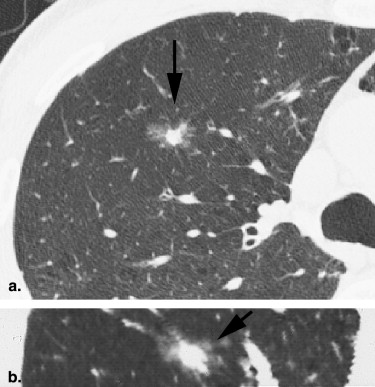
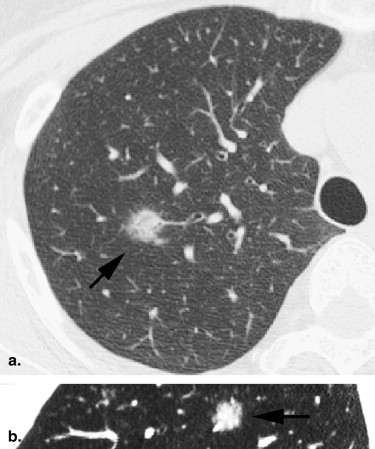
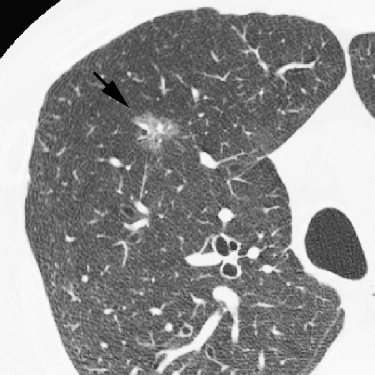
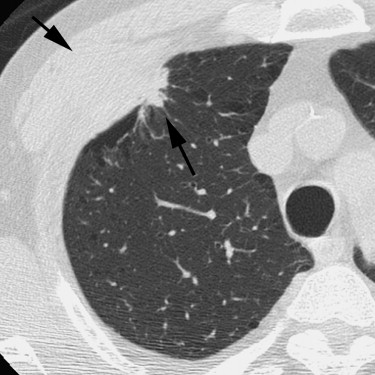
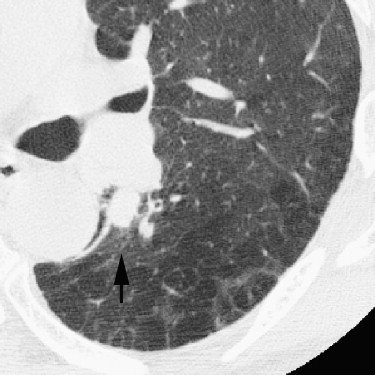
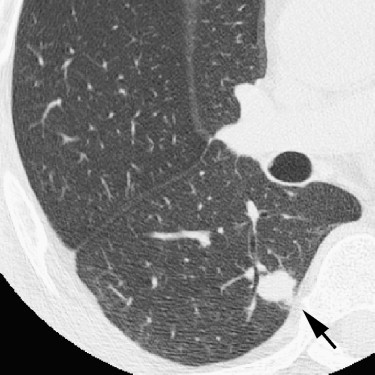
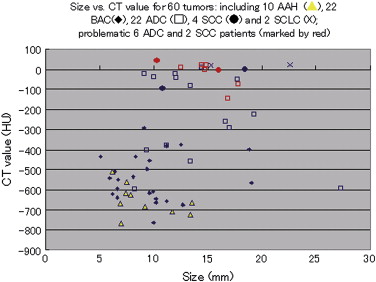
Rationale and Objectives
To define high-resolution computed tomography (HRCT) features of lung cancers detected by computed tomography (CT) screening according to histopathology and prognosis.
Methods and Materials
Tumor size, CT value, morphology, and tumor volume doubling time (TVDT) were determined for 10 atypical adenomatous hyperplasias (AAH) and 50 lung cancers followed between 1996 and 1998 to 2007.
Results
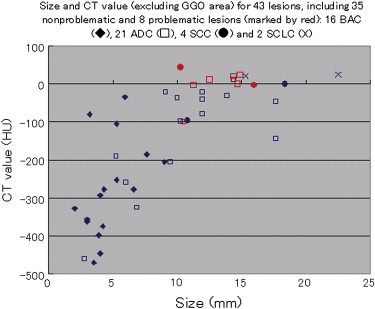
For the 10 AAHs, the density less than −500 HU in all lesions (mean, −654 HU) and the size was up to 14 mm (mean, 9 mm); all patients remain alive. Focal bronchioloalveolar cell carcinomas (BAC) were denser (mean, −537 HU) than AAH and mostly less dense than −350 HU; all patients remain alive. All 22 adenocarcinomas (ADC) were denser than −450 HU (mean, −186 HU); 6 were problematic and measured >−150HU and >10 mm or had >10 mm of central denser zone (CDZ) (partly solid tumors) or tumor size (solid tumor). Two of four squamous cell carcinomas (SCC) measuring 15 and 10 mm, respectively, were problematic. Two patients with small-cell lung carcinomas (SCLC) measuring 15 and 23 mm, respectively, remain alive. AAH, BAC, ADC, and SCC lesions were in general polygonal in shape. SCLC lesions appeared as round nodules with excrescence. The mean TVDT for AAH, BAC, ADC, SCC, and SCLC was 1278, 557, 466, 212, and 103 days, respectively.
Conclusion
The CT features reflected tumor aggressiveness. Non-SCLC lesions of >−150HU and >10 mm (or >−100HU and >10 mm for the solid portion of the tumor) are associated with unfavorable prognosis. Timely interventions should be undertaken before problematic increase in number of cases.
Lung cancer is currently the leading cause of cancer death in the United States; 215,020 cases of lung cancer are expected to be newly diagnosed and 161,840 patients will die from it in 2008 . This poor prognosis appears to reflect a small number of patients who are detected and treated at an early, curable stage . Low-dose computed tomography (CT) screening is effective in detecting peripheral small lung nodules . To translate early detection of lesions into true improvement of treatment outcome and prognosis, we need to establish an effective management strategy for small nodules by enhancing our understanding and interpretation of high-resolution CT (HRCT) findings. The aim of any such strategy should include the avoidance of both unnecessary invasive diagnostic procedures and treatment of indolent lesions and avoidance of underdiagnosis or delay in initiating treatment for genuine lung cancer .
We conducted a trial of low-dose CT screening for lung cancer between 1996 and 1998 . We report in this article the HRCT features of lung cancer found in the program, according to histopathology and prognosis of patients. This information would help radiologists in making meaningful workup examinations of HRCT scans.
Background of the study
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Subjects and study methods
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Methods
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Table 1
Demographic and Pathological Features of 10 Cases of AAH and 50 Lung Cancer Lesions (58 Patients)
n/lesion Sex (F/M) Smoking status (NS/S) Age (y), mean (min–max) Pathologic stage Outcome (Alive/survived with disease/dead) CT pattern AAH 10 6/4 7/3 64 (49–73) IA:10 10/0 none/part/solid: 6/4/0 BAC 21/22 12/9 15/6 65 (50–73) IA: 22 21/0 6/16/0 ADC 21/22 10/11 10/11 63 (46–74) IA:14, IB:1, IIA:1, IIIA:1, IIIB:1, IV:3 (including 2 cases with one each of small satellite lesion) 17/1/3 1/21/0 SCC 4 0/4 0/4 64 (47–73) IA:2, IIA:1, IIIB:1 3/0/1 0/0/4 SCLC 2 0/2 0/2 70 (67–73) IA:2 2/0/0 0/0/2 Total 58/60 28/30 32/28 64 (46–74) IA(52), IB(1), IIA(2), IIIA(1), IIIB(2), VI(1) 53/1/4 13/41/6
AAH, atypical adenomatous hyperplasia; BAC, bronchioloalveolar cell carcinoma; ADC, adenocarcinoma; SCC, squamous cell carcinoma; SCLC, small-cell lung carcinomas; NS, nonsmoker; S, smoker.
Table 2
CT Features of 10 Cases of AAH and 50 Lung Cancer Lesions (58 Patients)
Measurements on last HRCT, mean (min–max) CDZ TVDT Shape Size (mm) CT value Size CT value Screen CT HRCT Polygonal Miscellaneous AAH 9 (6– 14) -654 (-765, -512) 3 (3, 5, n = 4) -305 (-463, -130) 1254 (457, 3730, n = 7) 1336 (649, 2607, n = 3) 9 1 (epicycloid) BAC 10 (5–19) -537 (-763, -291) 5 (2, 9, n = 16) -278 (-470, -34) 571 (73, 2166, n = 9) 536 (113, 1799, n = 6) 21 1 (hypocycloid) ADC 14 (7–27) -186 (-640, 22) 11 (3, 18, n = 20) -96 (-461, 22) 503 (59, 2865, n = 15) 227 (185, 258, n = 4) 20 1 (epicycloid), 1 (irregularly infiltrative) SCC 14 (10–18) -15 (-97, 43) 212 (73, 350, n = 2) 4 (including 2 with round margins) SCLC 19 (15–23) 14 (10, 16) 103 (64, 142, n = 2) 2 (lobulated mass) Total 12 (5–27) -377 (-765, 43) 631 (59, 3730, n = 35) 527 (113, 2607, n = 12) 54 6
Abbreviations as in Table 1 . TVDT, tumor volume doubling time; CDZ, central denser zone.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Acknowledgment
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Jemal A., Siegel R., Ward E., et. al.: Cancer statistics, 2008. CA Cancer J Clin 2008; 58: pp. 71-96.
2. Martini N., Bains M.S., Burt M.E., et. al.: Incidence of local recurrence and second primary tumors in resected stage I lung cancer. J Thorac Cardiovasc Surg 1995; 109: pp. 120-129.
3. Asamura H., Goya T., Koshiishi Y., et. al.: A Japanese lung cancer registry study. Prognosis of 13,010 resected lung cancers. J Thorac Oncol 2008; 3: pp. 46-52.
4. Shepherd F.A., Crowley J., Van Houtte P., et. al.: The International Association for the Study of Lung Cancer Lung Cancer Staging Project: proposals regarding the clinical staging of small cell lung cancer in the forthcoming (seventh) edition of the tumor, node, metastasis classification for lung cancer. J Thorac Oncol 2007; 2: pp. 1067-1077.
5. Henschke C.I., Yankelevitz D.F., Libby D.M., et. al., The International Early Lung Cancer Action Program (I-ELCAP) Investigators: Survival of patients with Stage I lung cancer detected on CT screening. N Engl J Med 2006; 355: pp. 1763-1771.
6. Sone S., Nakayama T., Honda T., et. al.: Long-term follow-up study of a population-based 1996–1998 mass screening programme for lung cancer using mobile low-dose spiral computed tomography. Lung Cancer 2007; 58: pp. 329-341.
7. Noguchi M., Morikawa A., Kawasaki M., et. al.: Small adenocarcinoma of the lung. Histologic characteristics and prognosis. Cancer 1995; 75: pp. 2844-2852.
8. Yokose T., Suzuki K., Nagai K., et. al.: Favorable and unfavorable morphological prognostic factors in peripheral adenocarcinoma of the lung 3 cm or less in diameter. Lung Cancer 2000; 29: pp. 179-188.
9. Suzuki K., Yokose T., Yoshida J., et. al.: Prognostic significance of the size of central fibrosis in peripheral adenocarcinoma of the lung. Ann Thorac Surg 2000; 69: pp. 893-897.
10. Watanabe S.-i., Oda M., Go T., et. al.: Should mediastinal nodal dissection be routinely be undertaken in patients with peripheral small-sized (2cm or less) lung cancer? Retrospective analysis of 225 patients. Eur J Cardiothorac Surg 2001; 20: pp. 1007-1011.
11. Gajra A., Newman N., Gamble G.P., et. al.: Impact of tumor size on survival in stage IA non-small cell lung cancer: a case for subdividing stage IA disease. Lung Cancer 2003; 42: pp. 51-57.
12. Terasaki H., Niki T., Matsuno Y., et. al.: Lung adenocarcinoma with mixed bronchioloalveolar and invasive components. Am J Surg Pathol 2003; 27: pp. 937-951.
13. Funai K., Yokose T., Ishii G.-i., et. al.: Clinicopathologic characteristics of peripheral squamous cell carcinoma of the lung. Am J Surg Pathol 2003; 27: pp. 978-984.
14. MacMahon H., Austin J.H.M., Gamsu G., et. al.: Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the Fleischner Society. Radiology 2005; 237: pp. 395-400.
15. Sone S., Takashima S., Li F., et. al.: Mass screening for lung cancer with mobile spiral computed tomography scanner. Lancet 1998; 351: pp. 1242-1245.
16. Sone S., Li F., Yang Z.G., et. al.: Characteristics of small lung cancers invisible on conventional chest radiography and detected by population based screening using spiral CT. Br J Radiol 2000; 73: pp. 137-145.
17. Sone S., Li F., Yang Z.G., et. al.: Results of three-year mass screening programme for lung cancer using mobile low-dose spiral computed tomography scanner. Br J Cancer 2001; 84: pp. 25-32.
18. Hasegawa M., Sone S., Takashima S., et. al.: Growth rate of small lung cancers detected on mass CT screening. Br J Radiol 2000; 73: pp. 1252-1259.
19. Kawakami S., Sone S., Takashima S., et. al.: Atypical adenomatous hyperplasia of the lung: correlation between high-resolution CT findings and histopathologic features. Eur Radiol 2001; 11: pp. 811-814.
20. Sone S., Nakayama T., Honda T., et. al.: CT findings of early-stage small cell lung cancer in a low-dose CT screening programme. Lung Cancer 2007; 56: pp. 207-215.
21. Aoki T., Nakata H., Watanabe H., et. al.: Evolution of peripheral lung adenocarcinomas: CT findings correlated with histology and tumor doubling time. Am J Roentgenol 2000; 174: pp. 763-768.
22. Reid L.: The secondary lobule in the adult human lung, with special reference to its appearance in bronchograms. Thorax 1958; 13: pp. 110-115.
23. Raskin S.P.: The pulmonary acinus. Radiology 1982; 144: pp. 31-34.
24. Gamsu G., Thurlbeck W.M., Macklem P.T., et. al.: Roentgenographic appearance of the human pulmonary acinus. Invest Radiol 1971; 6: pp. 171-175.
25. Pump K.K.: Morphology of the acinus of the human lung. Chest 1969; 56: pp. 126-134.
26. Itoh H., Murata K., Konishi J., et. al.: Diffuse lung disease: Pathologic basis for the high resolution computed tomography findings. J Thorac Imaging 1993; 8: pp. 176-188.
27. Giovagnorio F., Cavallo V.: HRCT evaluation of secondary lobules and acini of the lung. J Thorac Imaging 1995; 10: pp. 129-133.
28. Webb W.R., Müller N.L., Naidich D.P.: High-resolution CT of the lung.1992.Raven PressNew York
29. Travis W.D., Garg K., Franklin W.A., et. al.: Evolving concept in the pathology and computed tomography imaging of lung adenocarcinoma and bronchioloalveolar carcinoma. J Clin Oncol 2005; 23: pp. 3279-3287.
30. Nakahara R., Yokose T., Nagai K., et. al.: Atypical adenomatous hyperplasia of the lung: a clinicopathological study of 118 cases including cases with multiple atypical adenomatous hyperplasia. Thorax 2001; 56: pp. 302-305.
31. Kim H.Y., Shim Y.M., Lee K.S., et. al.: Persistent pulmonary nodular ground-glass opacity at thin-section CT: histopathologic comparisons. Radiology 2007; 245: pp. 267-275.
32. Oda S., Awai K., Liu D., et. al.: Ground-glass opacities on thin-section helical CT: differentiation between bronchioloalveolar carcinoma and atypical adenomatous hyperplasia. Am J Roentgenol 2008; 190: pp. 1363-1368.
33. Miller W.T., Husted J., Freiman D., et. al.: Bronchioloalveolar carcinoma: two clinical entities with one pathologic diagnosis. Am J Roentgenol 1978; 130: pp. 905-912.
34. Hill C.A.: Bronchioloalveolar carcinoma: a review. Radiology 1984; 150: pp. 15-20.
35. Sakurai H., Asamura H., Watanabe S.-i., et. al.: Clinicopathologic features of peripheral squamous cell carcinoma of the lung. Ann Thorac Surg 2004; 78: pp. 222-227.