Rationale and Objectives
Targeted molecular probes offer the potential for greater specificity in cancer imaging with contrast-enhanced computed tomography (CT). We investigate a low-density lipoprotein (LDL) nanoparticle loaded with poly-iodinated triglyceride (ITG) in a proof of concept study of targeted x-ray imaging. LDLs are targeted to the LDL cell surface receptor (LDLR), which is overexpressed in several tumor types. The LDL-LDLR pathway presents a high-capacity and self-renewing transport system for molecular imaging in CT.
Materials and Methods
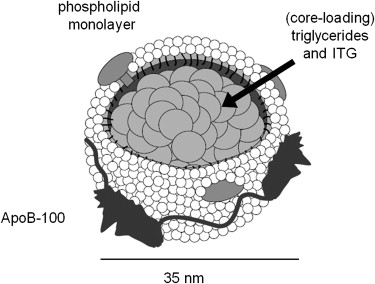
ITG was synthesized and loaded into the core of LDL particles to form a reconstituted nanoparticle, hereafter referred to as (rITG)LDL. Particle size was measured by dynamic light scattering. The x-ray attenuation of the (rITG)LDL solution was measured with CT imaging and signal enhancement was calibrated for equivalent iodine concentration. Cultured human hepatoblastoma G2 (HepG2) cells, which overexpress LDLR, were incubated with (rITG)LDL with or without native LDL. The cells were imaged with CT to characterize particle sequestration.
Results
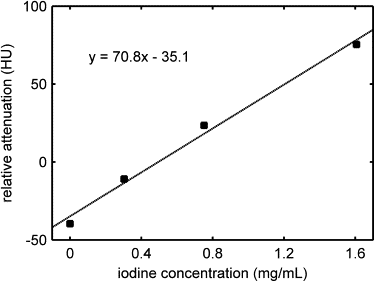
Reconstitution of LDL with ITG was successful and did not compromise the targeting function of the particle. Measurement of the x-ray attenuation properties of the (rITG)LDL solution revealed an effective iodine concentration of 0.78 mg/mL. In vitro studies of HepG2 cells demonstrated a significant increase in CT image intensity over control cells when incubated with (rITG)LDL.
Conclusion
The in vitro results of this study suggest that (rITG)LDL can provide adequate image enhancement for CT molecular imaging. Potential applications include breast imaging and small animal imaging at low x-ray energies. In vivo experiments will be required to verify that tumor uptake of (rITG)LDL is sufficient for enhanced detection. Use at higher x-ray energies, as used in conventional CT, will require a further increase in iodine loading.
Computed tomography (CT) plays an important role in diagnostic imaging of many types of cancer. High-quality imaging for the depiction of subtle anatomical and physiological changes associated with disease is achieved through the administration of exogenous x-ray contrast agents. These agents, typically water-soluble tri-iodinated derivatives of benzoic acid, impart imaging contrast in the body by absorbing x-rays more strongly than surrounding tissue structures, thus providing anatomical delineation of organs or tumors . The imaging contrast these agents provide is relatively short lived, typically on the order of minutes . To date, all of the approved x-ray contrast media are nonspecific extracellular agents; hence, once in the circulation, x-ray agents rapidly equilibrate with the extracellular space and subsequently undergo renal elimination.
Great efforts are being directed toward detecting cancer using targeted approaches in which conventional contrast agents are transported to malignant cells via nanoparticulate systems. Preferential accumulation of contrast media at tumor sites relative to surrounding normal tissues potentially allows lesions to be readily detected and characterized. This approach has been difficult to apply to CT imaging with the existing x-ray media because of the relatively low sensitivity of clinical CT and the commensurately high concentration of agent needed for contrast enhancement. It has been estimated that to provide a measurable signal in CT, a concentration of at least 0.5 mg/mL of iodine is required . As such, molecular imaging with an iodine-based x-ray agent would require large quantities of the contrast agent to be delivered to cancer cells. Few mechanisms exist in which such quantities of materials can be delivered and concentrated into cells. Here we describe the preliminary evaluation of the low-density lipoprotein (LDL) delivery pathway as a high-capacity, self-renewing system for molecular imaging of x-ray contrast agents.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Methods
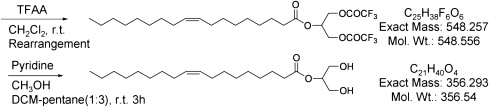
Preparation of ITG
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
LDL
Get Radiology Tree app to read full this article<
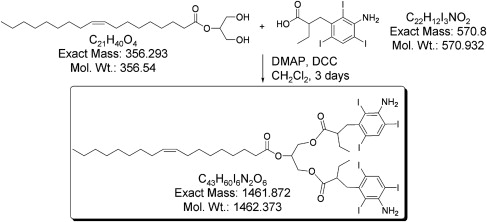
Preparation of (rITG)-LDL
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
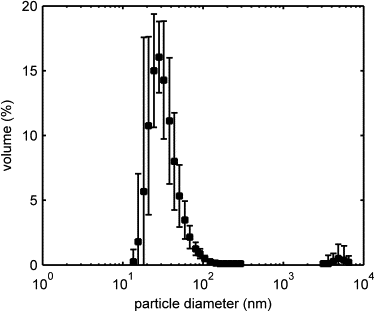
Size and Morphology
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
X-ray Solution Characterization
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
μx−μH2oμH2o−μair×1000 μ
x
−
μ
H
2
o
μ
H
2
o
−
μ
a
i
r
×
1000
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Cell Study
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Evaluation
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Table 1
(rITG)LDL Solution CT Number and Equivalent Iodine Concentration as a Function of Particle Concentration
Solution Sample (%) (rITG)LDL Concentration in Buffer (mg/mL) Relative Attenuation Coefficient (HU) Equivalent Iodine Concentration (mg/mL) 0 (buffer) 0 11 ± 0.4 N/A 25 0.465 32 ± 0.4 0.24 ± 0.01 50 0.93 49 ± 0.4 0.50 ± 0.01 100 1.86 70 ± 0.4 0.78 ± 0.01
(rITG), reconstituted poly-iodinated triglyceride; LDL, low-density lipoprotein; HU, Hounsfield units.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
HepG2 Cell Pellet Imaging Results
Cell Pellet Sample Relative Attenuation Coefficient (HU)P Value Equivalent Iodine Concentration (mg/mL) HepG2 (control) 45 ± 1 N/A 0 1. HepG2 + LDL + (rITG)LDL 36 ± 1 <1 × 1 −6 N/A 2. HepG2 + (rITG)LDL 76 ± 2 <1 × 1 −8 0.45 ± 0.03
(rITG), reconstituted poly-iodinated triglyceride; LDL, low-density lipoprotein; HU, Hounsfield units.
A one-tailed t -test was performed to determine statistical significance of the incubated pellet CT number compared to the control. The hypotheses were as follows, H 0 : μ - μ control = 0 and H a : μ - μ control < 0 for Experiment 1 and μ - μ control > 0 for Experiment 2.
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Krause W.: Delivery of diagnostic agents in computed tomography. Adv Drug Delivery Rev 1999; 37: pp. 159-173.
2. Rubin G.D.Rofsky N.M.CT and MR angiography: comprehensive vascular assessment.2009.Lippincott Williams & WilkinsPhiladelphia, PA:
3. Speck U.: Contrast agents: X-ray contrast agents and molecular imaging—a contradiction?.Semmler W.Schwaiger M.Molecular imaging I.2008.SpringerNew York: 185:167–175
4. Brown M.S., Goldstein J.L.: Receptor-mediated endocytosis: insights from the lipoprotein receptor system. Proc Natl Acad Sci 1979; 76: pp. 3330-3337.
5. Vitols S., Peterson C., Larsson O., et. al.: Elevated uptake of low density lipoproteins by human lung cancer tissue in vivo. Cancer Res 1992; 52: pp. 6244-6247.
6. Caruso M.G., Notarnicola M., Cavallini A., et. al.: Low density lipoprotein receptor and mRNA expression in human colorectal cancer. Anticancer Res 2001; 21: pp. 429-433.
7. Chen Y., Hughes-Fulford M.: Human prostate cancer cells lack feedback regulation of low-density lipoprotein receptor and its regulator, SREBP2. Int J Cancer 2001; 91: pp. 41-45.
8. Tatidis L., Masquelier M., Vitols S.: Elevated uptake of low density lipoprotein by drug resistant human leukemic cell lines. Biochem Pharmacol 2002; 63: pp. 2169-2180.
9. Niendorf A., Nagele H., Gerding D., et. al.: Increased LDL receptor mRNA expression in colon cancer is correlated with a rise in plasma cholesterol levels after curative surgery. Int J Cancer 1995; 61: pp. 461-464.
10. Graziani S.R., Igreja F.A., Hegg R., et. al.: Uptake of a cholesterol-rich emulsion by breast cancer. Gynecol Oncol 2002; 85: pp. 493-497.
11. Stranzl A., Schmidt H., Winkler R., et. al.: Low-density lipoprotein receptor mRNA in human breast cancer cells: influence by PKC modulators. Breast Cancer Res Treat 1997; 42: pp. 195-205.
12. Favre G.: Targeting of tumor cells by low density lipoproteins: principle and use of ellipticin derivatives. C R Seances Soc Biol Fil 1992; 186: pp. 73-87.
13. Lees A.M., Lees R.S.: 99mTechnetium-labeled low density lipoprotein: receptor recognition and intracellular sequestration of radiolabel. J Lipid Res 1991; 32: pp. 1-8.
14. Moerlein S.M., Daugherty A., Sobel B.E., et. al.: Metabolic imaging with gallium-68- and indium-111-labeled low-density lipoprotein. J Nucl Med 1991; 32: pp. 300-307.
15. Zheng G., Li H., Yang K., et. al.: Tricarbocyanine cholesteryl laurates labeled LDL: new near infrared fluorescent probes (NIRFs) for monitoring tumors and gene therapy of familial hypercholesterolemia. Bioorg Med Chem Lett 2002; 12: pp. 1485-1488.
16. Li H., Zhang Z., Blessington D., et. al.: Carbocyanine labeled LDL for optical imaging of tumors. Acad Radiol 2004; 11: pp. 669-677.
17. Corbin I.R., Lui H., Chen J., et. al.: Low-density lipoprotein nanoparticles as magnetic resonance imaging contrast agents. Neoplasia 2006; 8: pp. 488-498.
18. Crich S.G., Lanzardoy S., Alberti D., et. al.: Magnetic resonance imaging detection of tumor cells by targeting low-density lipoprotein receptors with Gd-loaded low-density lipoprotein particles. Neoplasia 2007; 9: pp. 1046-1056.
19. Weichert J.P., Longino M.A., Bakan D.A., et. al.: Polyiodinated triglyceride analogs as potential computed tomography imaging agents for the liver. J Med Chem 1995; 38: pp. 636-646.
20. Weichert J.P., Lee F.T., Longino M.A., et. al.: Computed tomography scanning of hepatic tumors with polyiodinated triglycerides. Acad Radiol 1996; 3: pp. 229-231.
21. Bakan D.A., Longino M.A., Weichert J.P., et. al.: Physicochemical characterization of a synthetic lipid emulsion for hepatocyte-selective delivery of lipophilic compounds: application to polyiodinated triglycerides as contrast agents for computed tomography. J Pharm Sci 1996; 85: pp. 908-914.
22. Stamatov S.D., Stawinski J.: Regioselective opening of an oxirane system with trifluoroacetic anhydride. A general method for the synthesis of 2-monoacyl- and 1,3 symmetrical triacylglycerols. Tetrahedron 2005; 61: pp. 3659-3669.
23. Lund-Katz S., Laplaud P.M., Phillips M.C., et. al.: Apolipoprotein B-100 conformation and particle surface charge in human LDL subspecies: implication for LDL receptor interaction. Biochemistry 1998; 37: pp. 12867-12874.
24. Krieger M.: Reconstitution of the hydrophobic core of low-density lipoprotein. Methods Enzymol 1986; 128: pp. 608-613.
25. Hill mL, Mainprize J.G., Mawdsley G.E., et. al.: A solid iodinated phantom material for use in tomographic xray imaging. Med Phys 2009; 36: pp. 4409-4420.
26. Johns H.E., Cunningham J.R.: The physics of radiology.1983.Charles C ThomasSpringfield, IL
27. Ford N.L., Thorton M.M., Holdsworth D.W.: Fundamental image quality limits for microcomputed tomography in small animals. Med Phys 2003; 30: pp. 2869-2877.
28. Jong R.A., Yaffe M.J., Skarpathiotakis M., et. al.: Contrast-enhanced digital mammography: initial clinical experience. Radiology 2003; 228: pp. 842-850.
29. Niklason L.T., Christian B.T., Niklason L.E., et. al.: Digital tomosynthesis in breast imaging. Radiology 1997; 205: pp. 399-406.
30. Paulus M.J., Gleason S.S., Kennel S.J., et. al.: High resolution X-ray computed tomography: an emerging tool for small animal cancer research. Neoplasia 2000; 2: pp. 62-70.

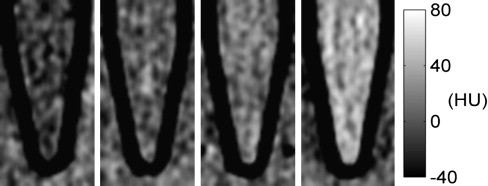
![Figure 8, Computed tomography slice images of cell pellets (4X magnification) (a) HepG2, (b) HepG2 incubated with low-density lipoprotein and reconstituted poly-iodinated triglyceride low-density lipoprotein ([rITG]LDL) in a 10:1 ratio, and (c) HepG2 incubated with (rITG)LDL. The cell pellets are visible as roughly round masses at the bottom of the tubes (indicated by arrows), suspended in a small amount of phosphate-buffered saline (PBS). The Eppendorf tube walls and the vegetable oil (above PBS) appear black in the images.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/InVitroAssessmentofPolyiodinatedTriglycerideReconstitutedLowDensityLipoprotein/7_1s20S1076633210003119.jpg)