High-resolution computed tomography is a necessary tool used in the diagnosis of interstitial lung disease. The interpretation of high-resolution computed tomography can be difficult given the wide spectrum of imaging appearances within the same disease and among different diseases. The authors provide a new educational method to learn about the spectrum of idiopathic interstitial lung disease through the use of a free online digital atlas and review article. This atlas can be downloaded at http://www.seattlechildrens.org/radiologyeducation/ILD .
High-resolution computed tomographic (HRCT) imaging is an important diagnostic tool used in the evaluation of patients presenting with dyspnea and suspected interstitial lung disease (ILD) . The various idiopathic ILDs often present with similar clinical symptomatology, physical examination findings, and restrictive lung physiology. Knowledge and understanding of the various imaging presentations of disease is imperative to arrive at the correct diagnosis and guide patient management and eventual therapy. A printed HRCT atlas for lung diseases has been published but may be limited by the inconvenience of referring to multiple pages, the exclusion of certain levels of the lung, and the inability to compare images of the spectrum of disease and different ILDs side by side. This article and digital atlas focus on idiopathic ILDs and do not include other causes of diffuse parenchymal lung disease, such as sarcoidosis, Langerhans cell histiocytosis, and lymphangioleiomyomatosis, as discussed in the consensus statement of the American Thoracic Society .
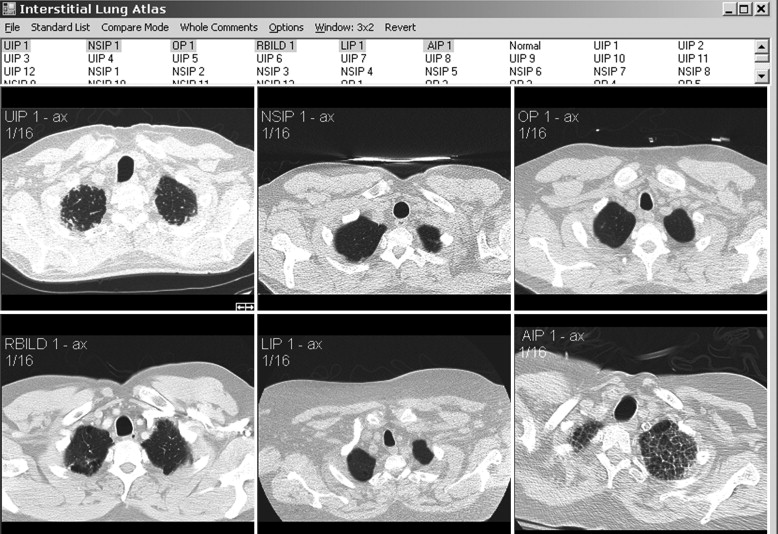
We present an interactive HRCT atlas of idiopathic ILD acquired in lung windows, with a brief review of the six core ILDs. Particular attention is paid to disease distribution and specific radiologic findings. The atlas was created to allow a more rapid assessment and comparison of the spectrum of imaging appearances within and across specific subtypes of ILD. This product will be especially useful for educating residents, practicing radiologists, medical students, and pulmonologists on the HRCT signs and patterns unique to different ILDs. Instructions for installation appear in Appendix A . A tutorial for use appears in Appendix B . Both of these appendices are available for download at http://www.academicradiology.org .
Materials and methods
Image Acquisition and Disease Selection
All HRCT examinations were performed from 2000 through 2010 on a 16-slice or 64-slice scanner. Most studies were performed using a standard HRCT protocol. Axial sections were acquired at 0.625-mm collimation in deep suspended inspiration and reconstructed at 1.5 mm. Images were displayed at a window width of 1000 to 1600 and a mean of −600 to −700. The field of view was approximately 35 cm. Images for the digital atlas were taken at 16 proximate predetermined levels from the inspiratory examination. Prone and expiratory images were not used for the atlas but are referred to in the comments when appropriate.
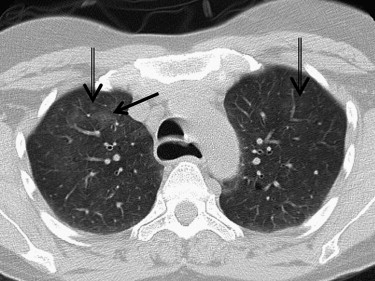
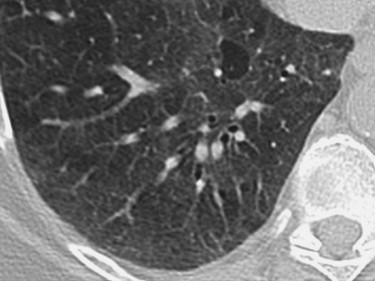
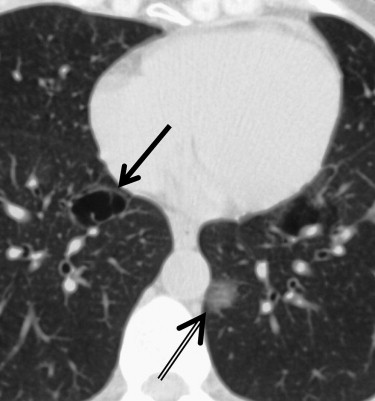
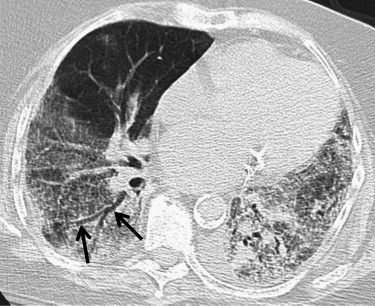
Cases included are those determined to have one of six idiopathic interstitial pneumonias: usual interstitial pneumonia (UIP), nonspecific interstitial pneumonia (NSIP), organizing pneumonia, the smoking-related ILDs (respiratory bronchiolitis [RB], RB-ILD, and desquamative interstitial pneumonia [DIP]), lymphocytic interstitial pneumonia (LIP), and acute interstitial pneumonia (AIP). Determination of disease was made by clinical, radiologic, and pathologic correlation. Pathologic confirmation occurred in approximately 70% of cases (33 of 47). Pathologic sampling was obtained from the most predictable area of disease as seen on computed tomography. Thoracic surgery performed the biopsies using video-assisted thoracoscopic surgery or wedge resection unless otherwise specified. When pathologic correlation was not available (approximately 30% of cases), radiologic and clinical determination were used. A total of 48 cases were included in the atlas, including one normal example. Original axial images were saved in Joint Photographic Experts Group image format and were cropped and reoriented, as needed, using Microsoft Office Picture Manager (Microsoft Corporation, Redmond, WA). Axial images were oriented in standard fashion.
Digital Atlas Program
The program used to run the atlas of images is written in C# (Microsoft Corporation) and was previously described .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion of idiopathic interstitial lung diseases
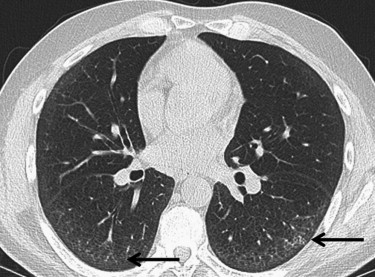
UIP
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
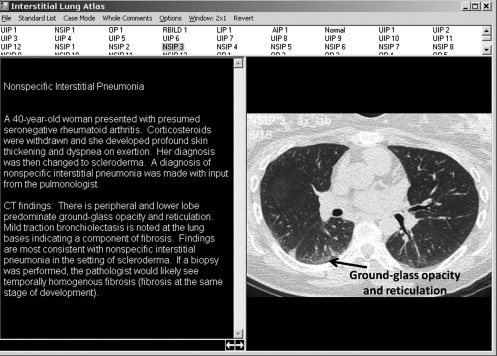
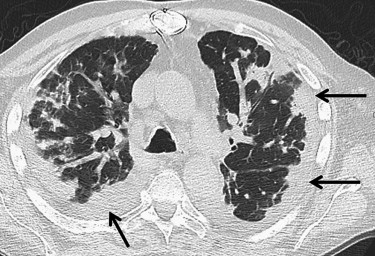
NSIP
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Organizing Pneumonia
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
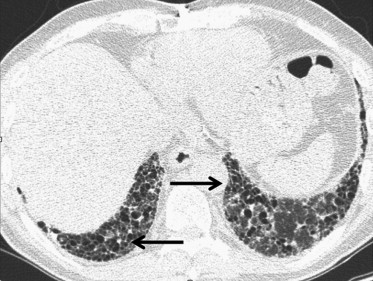
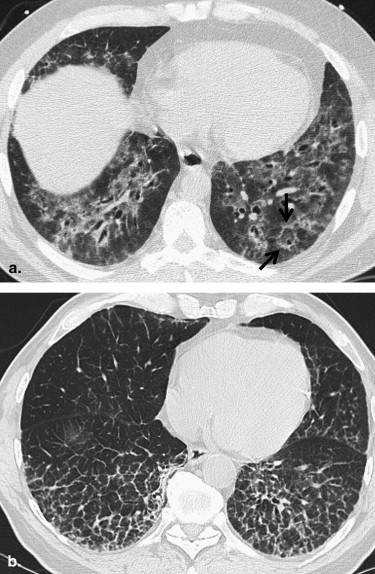
Smoking-related ILDs
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
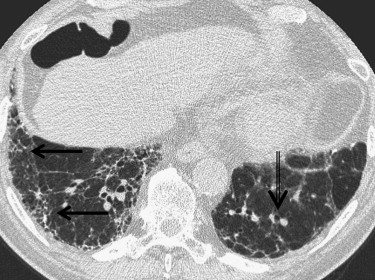
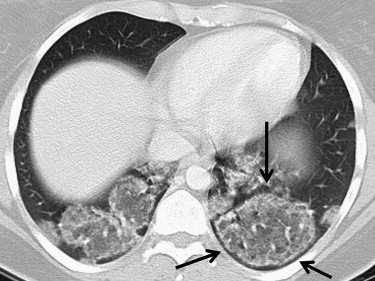
LIP
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
AIP
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Common HRCT Findings of Described ILDs
Disease Predilection of Disease HRCT Findings UIP Lower lung Subpleural and basal predominant lung fibrosis with honeycombing NSIP Lower lung Basal and peripheral ground-glass opacity with immediate subpleural sparing OP Slightly lower lung Nodular, masslike, or peribronchial consolidation, perilobular pattern, and reversed halo sign RB/RB-ILD Upper lung Patchy ground-glass opacity and ill-defined centrilobular nodules DIP Lower lung Diffuse ground-glass opacity and centrilobular emphysema LIP None Lung cysts, patchy ground-glass opacity, and poorly defined centrilobular nodules AIP Posterior and inferior lung Consolidation, ground-glass opacity, and traction bronchiectasis
AIP, acute interstitial pneumonia; DIP, desquamative interstitial pneumonia; HRCT, high-resolution computed tomographic; ILD, interstitial lung disease; LIP, lymphocytic interstitial pneumonia; NSIP, nonspecific interstitial pneumonia; OP, organizing pneumonia; RB, respiratory bronchiolitis; UIP, usual interstitial pneumonia.
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Supplementary data
Get Radiology Tree app to read full this article<
Appendix
Get Radiology Tree app to read full this article<
References
1. Grenier P., Valeyre D., Cluzel P., et. al.: Chronic diffuse interstitial lung disease: diagnostic value of chest radiography and high-resolution CT. Radiology 1991; 179: pp. 123-132.
2. Stern E.J., Swensen S.J., Kanne J.P.: High-resolution CT of the Chest: Comprehensive Atlas.3rd ed.2009.Lippincott Williams & WilkinsPhiladelphia 368
3. Society A.T., Society E.R.: American Thoracic Society/European Respiratory Society international multidisciplinary consensus classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 2002; 165: pp. 277-304.
4. Jelacic S., de Regt D., Weinberger E.: Interactive digital MR atlas of the pediatric brain. Radiographics 2006; 26: pp. 497-501.
5. Carrington C., Gaensler E., Coutu R., et. al.: Natural history and treated course of usual and desquamative interstitial pneumonia. N Engl J Med 1978; 298: pp. 801-809.
6. American Thoracic Society : Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS), and the European Respiratory Society (ERS). Am J Respir Crit Care Med 2000; 161: pp. 646-664.
7. Müller N., Guerry-Force M., Staples C., et. al.: Differential diagnosis of bronchiolitis obliterans with organizing pneumonia and usual interstitial pneumonia: clinical, functional, and radiologic findings. Radiology 1987; 162: pp. 151-156.
8. Bjoraker J., Ryu J., Edwin M., et. al.: Prognostic significance of histopathologic subsets in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 1998; 157: pp. 199-203.
9. King T.J., Schwarz M., Brown K., et. al.: Idiopathic pulmonary fibrosis: relationship between histopathologic features and mortality. Am J Respir Crit Care Med 2001; 164: pp. 1025-1032.
10. Katzenstein A., Myers J.: Idiopathic pulmonary fibrosis: clinical relevance of pathologic classification. Am J Respir Crit Care Med 1998; 157: pp. 1301-1315.
11. Nishimura K., Kitaichi M., Izumi T., et. al.: Usual interstitial pneumonia: histologic correlation with high-resolution CT. Radiology 1992; 182: pp. 337-342.
12. Johkoh T., Müller N., Cartier Y., et. al.: Idiopathic interstitial pneumonias: diagnostic accuracy of thin-section CT in 129 patients. Radiology 1999; 211: pp. 555-560.
13. Flaherty K., Thwaite E., Kazerooni E., et. al.: Radiological versus histological diagnosis in UIP and NSIP: survival implications. Thorax 2003; 58: pp. 143-148.
14. Travis W., Matsui K., Moss J., et. al.: Idiopathic nonspecific interstitial pneumonia: prognostic significance of cellular and fibrosing patterns: survival comparison with usual interstitial pneumonia and desquamative interstitial pneumonia. Am J Surg Pathol 2000; 24: pp. 19-33.
15. Nishiyama O., Kondoh Y., Taniguchi H., et. al.: Serial high resolution CT findings in nonspecific interstitial pneumonia/fibrosis. J Comput Assist Tomogr 2000; 24: pp. 41-46.
16. Park J., Lee K., Kim J., et. al.: Nonspecific interstitial pneumonia with fibrosis: radiographic and CT findings in seven patients. Radiology 1995; 195: pp. 645-648.
17. Kim T., Lee K., Chung M., et. al.: Nonspecific interstitial pneumonia with fibrosis: high-resolution CT and pathologic findings. AJR Am J Roentgenol 1998; 171: pp. 1645-1650.
18. Nagai S., Kitaichi M., Itoh H., et. al.: Idiopathic nonspecific interstitial pneumonia/fibrosis: comparison with idiopathic pulmonary fibrosis and BOOP. Eur Respir J 1998; 12: pp. 1010-1019.
19. Hartman T., Swensen S., Hansell D., et. al.: Nonspecific interstitial pneumonia: variable appearance at high-resolution chest CT. Radiology 2000; 217: pp. 701-705.
20. Bouchardy L., Kuhlman J., Ball W.J., et. al.: CT findings in bronchiolitis obliterans organizing pneumonia (BOOP) with radiographic, clinical, and histologic correlation. J Comput Assist Tomogr 1993; 17: pp. 352-357.
21. Epler G.: Bronchiolitis obliterans organizing pneumonia: definition and clinical features. Chest 1992; 102: pp. 2S-6S.
22. Lee K., Kullnig P., Hartman T., et. al.: Cryptogenic organizing pneumonia: CT findings in 43 patients. AJR Am J Roentgenol 1994; 162: pp. 543-546.
23. Pipavath S, Chung J, Godwin JD, Chien JW. Cryptogenic organizing pneumonia (COP) in post hematopietic stem cell transplantation (HSCT): CT features and association with graft versus host disease (GVHD). Presented at: Society of Thoracic Radiology; Las Vegas, NV; March 25–27, 2007.
24. Ujita M., Renzoni E., Veeraraghavan S., et. al.: Organizing pneumonia: perilobular pattern at thin-section CT. Radiology 2004; 232: pp. 757-761.
25. Kim S., Lee K., Ryu Y., et. al.: Reversed halo sign on high-resolution CT of cryptogenic organizing pneumonia: diagnostic implications. AJR Am J Roentgenol 2003; 180: pp. 1251-1254.
26. Lee J., Lynch D., Sharma S., et. al.: Organizing pneumonia: prognostic implication of high-resolution computed tomography features. J Comput Assist Tomogr 2003; 27: pp. 260-265.
27. Heyneman L., Ward S., Lynch D., et. al.: Respiratory bronchiolitis, respiratory bronchiolitis-associated interstitial lung disease, and desquamative interstitial pneumonia: different entities or part of the spectrum of the same disease process?. AJR Am J Roentgenol 1999; 173: pp. 1617-1622.
28. Myers J., Veal C.J., Shin M., et. al.: Respiratory bronchiolitis causing interstitial lung disease. A clinicopathologic study of six cases. Am Rev Respir Dis 1987; 135: pp. 880-884.
29. Attili A., Kazerooni E., Gross B., et. al.: Smoking-related interstitial lung disease: radiologic-clinical-pathologic correlation. Radiographics 2008; 28: pp. 1383-1396.
30. Holt R., Schmidt R., Godwin J., et. al.: High resolution CT in respiratory bronchiolitis-associated interstitial lung disease. J Comput Assist Tomogr 1993; 17: pp. 46-50.
31. Chung J., Kanne J.: Smoking-related interstitial lung diseases. Semin Roentgenol 2010; 45: pp. 29-35.
32. Hartman T., Primack S., Swensen S., et. al.: Desquamative interstitial pneumonia: thin-section CT findings in 22 patients. Radiology 1993; 187: pp. 787-790.
33. Nicholson A., Wotherspoon A., Diss T., et. al.: Reactive pulmonary lymphoid disorders. Histopathology 1995; 26: pp. 405-412.
34. Betsuyaku T., Munakata M., Yamaguchi E., et. al.: Establishing diagnosis of pulmonary malignant lymphoma by gene rearrangement analysis of lymphocytes in bronchoalveolar lavage fluid. Am J Respir Crit Care Med 1994; 149: pp. 526-529.
35. Ichikawa Y., Kinoshita M., Koga T., et. al.: Lung cyst formation in lymphocytic interstitial pneumonia: CT features. J Comput Assist Tomogr 1994; 18: pp. 745-748.
36. Johkoh T., Müller N., Pickford H., et. al.: Lymphocytic interstitial pneumonia: thin-section CT findings in 22 patients. Radiology 1999; 212: pp. 567-572.
37. Johkoh T., Müller N., Taniguchi H., et. al.: Acute interstitial pneumonia: thin-section CT findings in 36 patients. Radiology 1999; 211: pp. 859-863.