Rationale and Objectives
The authors present their initial experience using a 3-T whole-body scanner equipped with a 128-channel coil applied to lung motion assessment. Recent improvements in fast magnetic resonance imaging (MRI) technology have enabled several trials of free-breathing three-dimensional (3D) imaging of the lung. A large number of image frames necessarily increases the difficulty of image analysis and therefore warrants automatic image processing. However, the intensity homogeneities of images of prior dynamic 3D lung MRI studies have been insufficient to use such methods. In this study, initial data were obtained at 3 T with a 128-channel coil that demonstrate the feasibility of acquiring multiple sets of 3D pulmonary scans during free breathing and that have sufficient quality to be amenable to automatic segmentation.
Materials and Methods
Dynamic 3D images of the lungs of two volunteers were acquired with acquisition times of 0.62 to 0.76 frames/s and an image matrix of 128 × 128, with 24 to 30 slice encodings. The volunteers were instructed to take shallow and deep breaths during the scans. The variation of lung volume was measured from the segmented images.
Results
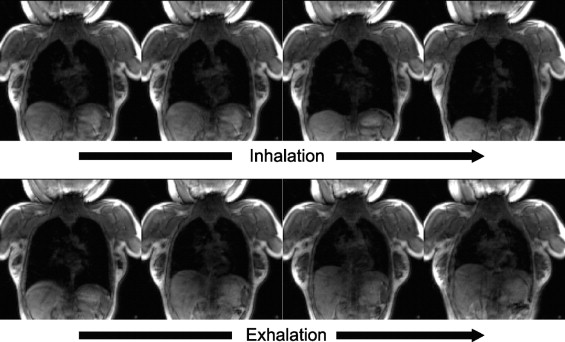
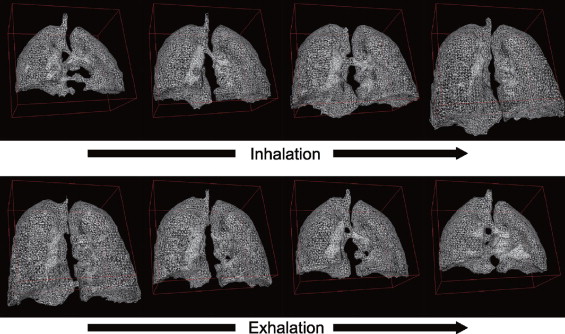
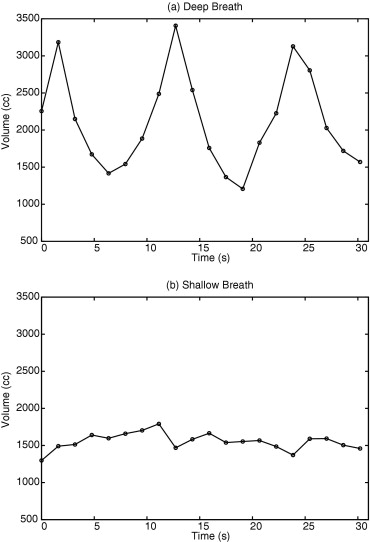
Dynamic 3D images were successfully acquired for both respiratory conditions for each subject. The images showed whole-lung motion, including lifting of the chest wall and the displacement of the diaphragm, with sufficient contrast to distinguish these structures from adjacent tissues. The average time to complete segmentation for one 3D image was 4.8 seconds. The tidal volume measured was consistent with known tidal volumes for healthy subjects performing deep-breathing maneuvers. The temporal resolution was insufficient to measure tidal volumes for shallow breathing.
Conclusion
This initial experience with a 3-T whole-body scanner and a 128-channel coil showed that the scanner and imaging protocol provided dynamic 3D images with spatial and temporal resolution sufficient to delineate the diaphragmatic domes and chest wall during active breathing. In addition, the intensity homogeneities and signal-to-noise ratio were adequate to perform automatic segmentation.
Magnetic resonance imaging (MRI) is a suitable option to analyze lung motion ( ). MRI has an advantage over other imaging modalities in motion analysis because it does not expose subjects to ionizing radiation, allowing baseline studies to be obtained in healthy subjects and compared to disease studies. As reported elsewhere, three-dimensional (3D) MRI is especially useful in studying the movement of the diaphragm and the rib cage ( ). One limitation of current 3D MRI of the lung is that it requires subjects to hold their breath at multiple respiratory phases ( ). It is well known from lung motion analysis using biplanar fluoroscopy that the motion of the lung during normal breathing has hysteresis that does not appear in breath-hold imaging ( ). Furthermore, subjects with chronic obstructive pulmonary disease typically have difficulty holding their breath, limiting the feasibility of MRI motion analysis in these subjects. Other investigators have tried to overcome these problems by performing dynamic 3D image studies by recording two-dimensional multislice imaging taken in free-breathing subjects ( ).
Recent improvements in fast imaging have enabled an advanced form of dynamic 3D imaging in free-breathing subjects ( ). Blackall et al ( ) proposed the use of a fast field echo and echo-planar imaging (EPI) sequence and achieved an imaging speed of 330 ms/frame, with acquisition matrix size of 128 × 256 for 25 to 27 slices. This imaging method was sufficiently fast to investigate the intracycle and intercycle reproducibility of respiratory motion, including hysteresis analysis. Parallel imaging is another possible solution to enable lung MRI in free-breathing subjects, but without the serious image distortion likely found in EPI-based sequences. Plathow et al ( ) reported motion analysis of the lung using a 3D fast low-angle shot (FLASH) sequence combined with a parallel imaging technique and view sharing on a 1.5-T whole-body magnetic resonance scanner with a 6-channel coil. This imaging speed was also sufficiently fast to monitor lung motion and to correlate pulmonary function and intrathoracic tumor mobility.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Subjects
Get Radiology Tree app to read full this article<
MRI Protocol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Postprocessing and Visualization of Magnetic Resonance Images
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Gierada D.S., Curtin J.J., Erickson S.J., Prost R.W., Strandt J.A., Goodman L.R.: Diaphragmatic motion: fast gradient-recalled-echo MR imaging in healthy subjects. Radiology 1995; 194: pp. 879-884.
2. Gierada D.S., Curtin J.J., Erickson S.J., Prost R.W., Strandt J.A., Goodman L.R.: Fast gradient echo magnetic resonance imaging of the normal diaphragm. J Thorac Imaging 1997; 12: pp. 70-74.
3. Gierada D.S., Hakimian S., Slone R.M., Yusen R.D.: MR analysis of lung volume and thoracic dimensions in patients with emphysema before and after lung volume reduction surgery. AJR Am J Roentgenol 1998; 170: pp. 707-714.
4. Suga K., Tsukuda T., Awaya H., et. al.: Impaired respiratory mechanics in pulmonary emphysema: evaluation with dynamic breathing MRI. J Magn Reson Imaging 1999; 10: pp. 510-520.
5. Iwasawa T., Yoshiike Y., Saito K., Kagei S., Gotoh T., Matsubara S.: Paradoxical motion of the hemidiaphragm in patients with emphysema. J Thorac Imaging 2000; 15: pp. 191-195.
6. Shimizu S., Shirato H., Aoyama H., et. al.: High-speed magnetic resonance imaging for four-dimensional treatment planning of conformal radiotherapy of moving body tumors. Int J Radiat Oncol Biol Phys 2000; 48: pp. 471-474.
7. Gee J., Sundaram T., Hasegawa I., Uematsu H., Hatabu H.: Characterization of regional pulmonary mechanics from serial magnetic resonance imaging data. Acad Radiol 2003; 10: pp. 1147-1152.
8. Kiryu S., Loring S.H., Mori Y., Rofsky N.M., Hatabu H., Takahashi M.: Quantitative analysis of the velocity and synchronicity of diaphragmatic motion: dynamic MRI in different postures. Magn Reson Imaging 2006; 24: pp. 1325-1332.
9. Paiva M., Verbanck S., Estenne M., Poncelet B., Segebarth C., Macklem P.T.: Mechanical implications of in vivo human diaphragm shape. J Appl Physiol 1992; 72: pp. 1407-1412.
10. Gauthier A.P., Verbanck S., Estenne M., Segebarth C., Macklem P.T., Paiva M.: 3-dimensional reconstruction of the in-vivo human diaphragm shape at different lung-volumes. J Appl Physiol 1994; 76: pp. 495-506.
11. Cluzel P., Similowski T., Chartrand-Lefebvre C., Zelter M., Derenne J.P., Grenier P.A.: Diaphragm and chest wall: assessment of the inspiratory pump with MR imaging-preliminary observations. Radiology 2000; 215: pp. 574-583.
12. Craighero S., Promayon E., Baconnier P., Lebas J.F., Coulomb M.: Dynamic echo-planar MR imaging of the diaphragm for a 3D dynamic analysis. Eur Radiol 2005; 15: pp. 742-748.
13. Seppenwoolde Y., Shirato H., Kitamura K., et. al.: Precise and real-time measurement of 3D tumor motion in lung due to breathing and heartbeat, measured during radiotherapy. Int J Radiat Oncol Biol Phys 2002; 53: pp. 822-834.
14. von Siebenthal M., Szekely G., Gamper U., Boesiger P., Lomax A., Cattin P.: 4D MR imaging of respiratory organ motion and its variability. Phys Med Biol 2007; 52: pp. 1547-1564.
15. Plathow C., Fink C., Sandner A., et. al.: Comparison of relative forced expiratory volume of one second with dynamic magnetic resonance imaging parameters in healthy subjects and patients with lung cancer. J Magn Reson Imaging 2005; 21: pp. 212-218.
16. Plathow C., Schoebinger M., Fink C., et. al.: Evaluation of lung volumetry using dynamic three-dimensional magnetic resonance imaging. Invest Radiol 2005; 40: pp. 173-179.
17. Blackall J.M., Ahmad S., Miquel M.E., McClelland J.R., Landau D.B., Hawkes D.J.: MRI-based measurements of respiratory motion variability and assessment of imaging strategies for radiotherapy planning. Phys Med Biol 2006; 51: pp. 4147-4169.
18. Plathow C., Fink C., Ley S., et. al.: Measurement of tumor diameter-dependent mobility of lung tumors by dynamic MRI. Radiother Oncol 2004; 73: pp. 349-354.
19. Schmitt M., Potthast A., Sosnovik D.E., et. al.: A 128 channel receive-only cardiac coil for highly accelerated cardiac MRI at 3 tesla. Magn Reson Imaging 2008; 59: pp. 1431-1439.
20. Yoo T.: Insight into images principles and practice for segmentation, registration and image analysis.2004.A.K. PetersWellesley, MA
21. Lorensen W.E., Cline H.E.: Marching cubes: a high resolution 3D surface construction algorithm. Comput Graph 1987; 21: pp. 163-169.
22. Schroeder W., Martin K., Lorensen B.: The Visualization Toolkit: an object-oriented approach to 3-d graphics.4th ed.2006.Kitware, IncClifton Park, NY
23. Low D.A., Parikh P.J., Lu W., et. al.: Novel breathing motion model for radiotherapy. Int J Radiat Oncol Biol Phys 2005; 63: pp. 921-929.
24. Lu W., Parikh P.J., El Naqa I.M., et. al.: Quantitation of the reconstruction quality of a four-dimensional computed tomography process for lung cancer patients. Med Phys 2005; 32: pp. 890-901.
25. Simon L., Giraud P., Servois V., Rosenwald J.C.: Lung volume assessment for a cross-comparison of two breathing-adapted techniques in radiotherapy. Int J Radiat Oncol Biol Phys 2005; 63: pp. 602-609.