Rationale and Objectives
In patients with severe internal carotid artery steno-occlusive lesions (ISOL), impaired cerebrovascular reactivity (CVR) is predictive of future ischemic stroke (IS) or transient ischemic attack (TIA). Therefore, the evaluation of CVR in ISOL patients may be a means to evaluate the risk for IS/TIA and decide on an intervention. Our aim was (1) to explore the feasibility of concurrent near-infrared spectroscopy (NIRS-DOS), diffuse correlation spectroscopy, and transcranial Doppler for CVR assessment in ISOL patients, and (2) to compare macrovascular and microvascular CVR in ISOL patients and explore its potential for IS/TIA risk stratification.
Materials and Methods
Twenty-seven ISOL patients were recruited. The changes in continuous microvascular and macrovascular hemodynamics upon acetazolamide injection were used to determine CVR.
Results
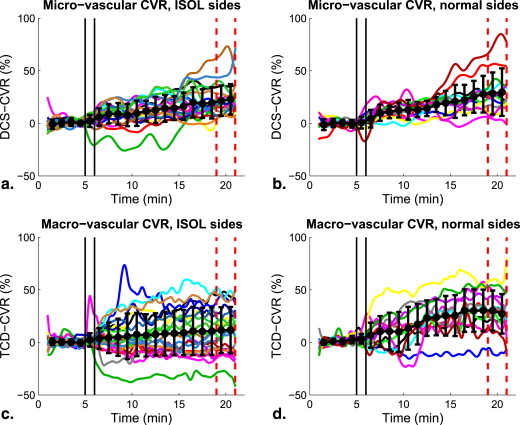
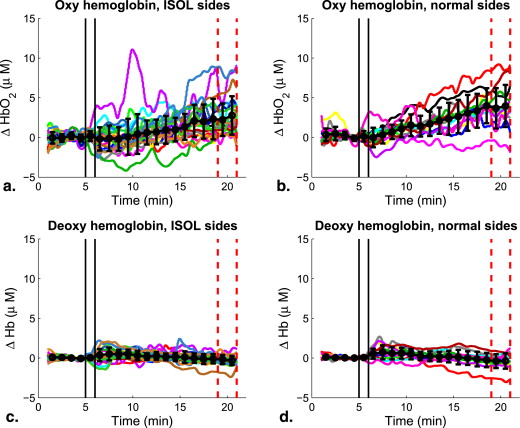
Oxyhemoglobin (HbO 2 , by near-infrared spectroscopy), microvascular cerebral blood flow (CBF, by diffuse correlation spectroscopy) and CBF velocity (by transcranial Doppler) showed significant increases upon acetazolamide injection in all subjects ( P < .03). Only macrovascular CVR ( P = .024) and none of the microvascular measures were significantly dependent on the presence of ISOL. In addition, while CBF was significantly correlated with HbO 2 , neither of these microvascular measures correlated with macrovascular CBF velocity.
Conclusions
We demonstrated the simultaneous, continuous, and noninvasive evaluation of CVR at both the microvasculature and macrovasculature. We found that macrovascular CVR response depends on the presence of ISOL, whereas the microvascular CVR did not significantly depend on the ISOL presence, possibly due to the role of collaterals other than those of the circle of Willis. The concurrent microvascular and macrovascular CVR measurement in the ISOL patients might improve future IS/TIA risk assessment.
Severe stenosis or occlusion of the internal carotid artery (ICA) increases the risk of subsequent ischemic stroke (IS) and transient ischemic attack (TIA) . Moreover, patients with severe internal carotid artery steno-occlusive lesions (ISOL) are found to be at a particularly higher risk of imminent stroke events when autoregulatory vasodilation capacity of the cerebral terminal arterioles in response to a reduced perfusion pressure, or cerebrovascular reactivity (CVR), is impaired . Thus, to aid in the decision of the suitable treatment, such as carotid revascularization versus drug therapy, the assessment of CVR has been proposed by several authors as a screening method to stratify ISOL patients based on their assumed risk of future IS or TIA .
CVR is normally evaluated by measuring the maximum vasodilation capacity of cerebral vasculature. The maximum vasodilation is induced by introducing a potent vasodilatory stimulus, most commonly CO 2 , inhalation or acetazolamide (ACZ) infusion. Consequent cerebrovascular changes are then followed by a method capable of measuring microvascular cerebral blood flow (CBF), blood oxygen saturation, blood volume, or macrovascular cerebral blood flow velocity (CBFV).
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Subjects
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Diffuse Optics and TCD Protocol
Get Radiology Tree app to read full this article<
Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Table 1
Demographic and Clinical Characteristics of the Population Included for the Analysis
Parameter Number Total no. of subjects 20 Total no. of hemispheres 33 Male 16 (80%) Age, (mean ± STD years) 64.6 ± 9.7 Degree of ICA stenosis 0%–49% 12 (36%) 70%–99% 14 (43%) Occluded 7 (21%) Laterality of stenosis Unilateral steno-occlusion 16 (80%) Bilateral steno-occlusion 4 (20%) Presenting events Symptomatic arteries 8 (24%) Transient ischemic attack 1 Ischemic stroke 7 Asymptomatic arteries 25 (76%) Other conditions Smoker 6 (30%) Diabetic 8 (40%) Hypercholesterol 12 (60%) Hypertension 16 (80%)
Data are given as number of subjects and percentage of the total included subjects [No. (%)].
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Median (Interquartile Range [IQR]), and P Values for Optical and Ultrasound Data
Parameter ISOL Side Normal Side Median IQR Median IQR ΔHb o 2 , μmol/L 3.7 ∗ 2.3 to 8.3 7.7 ∗ 5.4 to 12 ΔHb, μmol/L −0.4 −1.2 to 0.1 −0.5 −1.8 to 0.7 CVR DCS , % 20 ∗ 14.3 to 25.1 26.1 ∗ 13.9 to 32 CVR TCD , % 10.7 ∗ −6.8 to 27.5 27.8 ∗ 19 to 42.7
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 3
MCA CVR (TCD-CVR) Versus Local CVR (DCS-CVR) and ΔHbO 2
Parameter Responsive by TCD Nonresponsive by TCD Responsive by DCS 15 (45) 10 (30) Nonresponsive by DCS 5 (15) 3 (9) Responsive by NIRS-DOS, ΔHbO 2 13 (39) 4 (12) Nonresponsive by NIRS-DOS, ΔHbO 2 7 (21) 9 (27)
Data are indicated as the number of hemispheres/arteries and as percentage of the total [No. (%)].
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Barnett H., Taylor D., Eliasziw M., et. al.: Benefit of carotid endarterectomy in patients with symptomatic moderate or severe stenosis. N Engl J Med 1998; 339: pp. 1415-1425.
2. Gupta A., Chazen J.L., Hartman M., et. al.: Cerebrovascular reserve and stroke risk in patients with carotid stenosis or occlusion: A systematic review and meta-analysis. Stroke 2012; 43: pp. 2884-2891.
3. Gibbs J., Leenders K., Wise R., et. al.: Evaluation of cerebral perfusion reserve in patients with carotid-artery occlusion. Lancet 1984; 323: pp. 310-314.
4. Kistler J., Ropper A., Heros R.: Therapy of ischemic cerebral vascular disease due to atherothrombosis. N Engl J Med 1984; 311: pp. 27-34.
5. Caplan L., Hennerici M.: Impaired clearance of emboli (washout) is an important link between hypoperfusion, embolism, and ischemic stroke. Arch Neurol 1998; 55: pp. 1475.
6. Markus H., Cullinane M.: Severely impaired cerebrovascular reactivity predicts stroke and TIA risk in patients with carotid artery stenosis and occlusion. Brain 2001; 124: pp. 457-467.
7. Herzig R., Hlustík P., Urbánek K., et. al.: Can we identify patients with carotid occlusion who would benefit from EC/IC bypass?. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2004; 148: pp. 119-122.
8. Vernieri F., Tibuzzi F., Pasqualetti P., et. al.: Transcranial Doppler and near-infrared spectroscopy can evaluate the hemodynamic effect of carotid artery occlusion. Stroke 2004; 35: pp. 64-70.
9. Russell S., Woo H., Siller K., et. al.: Evaluating middle cerebral artery collateral blood flow reserve using acetazolamide transcranial Doppler ultrasound in patients with carotid occlusive disease. Surg Neurol 2008; 70: pp. 466-470.
10. Ozgur H.T., Walsh T.K., Masaryk A., et. al.: Correlation of cerebrovascular reserve as measured by acetazolamide-challenged SPECT with angiographic flow patterns and intra- or extracranial arterial stenosis. Am J Neuroradiol 2001; 22: pp. 928-936.
11. Romano J.G., Liebeskind D.S.: Revascularization of collaterals for hemodynamic stroke insight on pathophysiology from the Carotid Occlusion Surgery Study. Stroke 2012; 43: pp. 1988-1991.
12. T Durduran, AG Yodh, Diffuse correlation spectroscopy for noninvasive, microvascular cerebral blood flow measurement, NeuroImage, 2013 in Press.
13. Boas D., Campbell L., Yodh A.: Scattering and imaging with diffusing temporal field correlations. Phys Rev Lett 1995; 75: pp. 1855-1858.
14. Boas D.A., Yodh A.G.: Spatially varying dynamical properties of turbid media probed with diffusing temporal light correlation. J Opt Soc Am A 1997; 14: pp. 192-215.
15. Durduran T., Choe R., Baker W., et. al.: Diffuse optics for tissue monitoring and tomography. Rep Progr Phys 2010; 73: pp. 076701.
16. Mesquita R., Durduran T., Yu G., et. al.: Direct measurement of tissue blood flow and metabolism with diffuse optics. Phil Trans R Soc 2011; 369: pp. 4390-4406.
17. Zirak P., Delgado-Mederos R., Martí-Fàbregas J., et. al.: Effects of acetazolamide on the microand macrovascular cerebral hemodynamics: a diffuse optical and transcranial Doppler ultrasound study. Biomed Opt Exp 2010; 1: pp. 1443-1459.
18. Grant E., Benson C., Moneta G., et. al.: Carotid artery stenosis: gray-scale and Doppler us diagnosis society of radiologists in ultrasound consensus conference1. Radiology 2003; 229: pp. 340-346.
19. North American Symptomatic Carotid Endarterectomy Trial Collaborators: Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med 1991; 325: pp. 445-453.
20. Gibbons J.D.: Nonparametric Statistics: An Introduction1993.SageCalifornia, USA
21. R Development Core Team: R: A Language and Environment for Statistical Computing.2009.R Foundation for Statistical ComputingVienna, Austria
22. Brauer P., Kochs E., Werner C., et. al.: Correlation of transcranial Doppler sonography mean flow velocity with cerebral blood flow in patients with intracranial pathology. J Neurosurg Anesthesiol 1998; 10: pp. 80.
23. Pindzola R., Balzer J., Nemoto E., et. al.: Cerebrovascular reserve in patients with carotid occlusive disease assessed by stable xenon-enhanced CT cerebral blood flow and transcranial Doppler. Stroke 2001; 32: pp. 1811-1817.
24. Bokkers R., van Osch M., van der Worp H., et. al.: Symptomatic carotid artery stenosis: impairment of cerebral autoregulation measured at the brain tissue level with arterial spin-labeling MR imaging. Radiology 2010; 256: pp. 201-208.
25. Henderson R., Eliasziw M., Fox A., et. al.: Angiographically defined collateral circulation and risk of stroke in patients with severe carotid artery stenosis. Stroke 2000; 31: pp. 128-132.
26. Yamaguchi H., Yamauchi H., Hazama S., et. al.: Correlation between cerebral oxygen metabolism and cerebral blood flow simultaneously measured before and after acetazolamide administration. J Biomed Optics 1999; 4: pp. 418.
27. Tachtsidis I., Tisdall M., Delpy D., et. al.: Measurement of cerebral tissue oxygenation in young healthy volunteers during acetazolamide provocation: a transcranial Doppler and near-infrared spectroscopy investigation. Oxygen Transport to Tissue XXIX; Advances In Experimental Medicine And Biology 2008; 614: pp. 389-396.