Rationale and Objectives
Based on their association with malignant proliferation, using noninvasive phosphorus MR spectroscopic imaging ( 31 P MRSI), we measured the tumor content of the phospholipid-related phosphomonoesters (PME), phosphoethanolamine and phospholcholine, and its correlation with treatment outcome in newly diagnosed patients with diffuse large B-cell lymphoma (DLBCL) receiving standard first-line chemotherapy.
Experimental Design
The PME value normalized to nucleoside triphosphates (PME/NTP) was measured using 31 P MRSI in tumor masses of 20 patients with DLBCL before receiving standard first-line chemotherapy. Response at 6 months was complete in 13 patients and partial in seven. Time to treatment failure (TTF) was ≤11 months in eight patients, from 18 to 30 months in three, and ≥60 months in nine.
Results
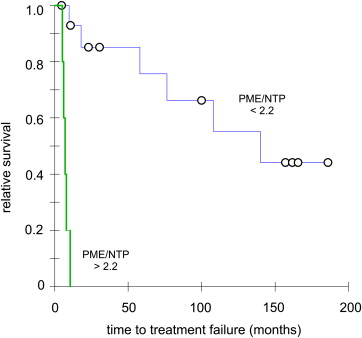
On a t test, the pretreatment tumor PME/NTP mean value (SD, n ) of patients with a complete response at 6 months was 1.42 (0.41, 13), which was significantly different from the value of 2.46 (0.40, 7) in patients with partial response ( P < .00001). A Fisher test significantly correlated the PME/NTP values with response at 6 months (sensitivity and specificity at 0.85, P < .004) while a Cox proportional hazards regression significantly correlated the PME/NTP values with TTF (hazard ratio = 5.21, P < .02). A Kaplan–Meier test set apart a group entirely composed of patients with TTF ≤ 11 months (hazard ratio = 8.66, P < .00001).
Conclusions
The pretreatment tumor PME/NTP values correlated with response to treatment at 6 months and time to treatment failure in newly diagnosed patients with DLBCL treated with first-line chemotherapy, and therefore they could be used to predict treatment outcome in these patients.
For many cancer patients, established first-line therapies are either ineffective or initially effective but not curative. For instance, 63% of patients with diffuse large B-cell lymphoma (DLBCL) show a complete response to first-line therapy, but only 40% have prolonged survival . An a priori method to identify unresponsive patients to standard treatments would be of extreme value to offer these patients alternate treatment options, thereby maximizing therapeutic success, sparing toxicity, and lowering health care costs.
Newly diagnosed patients with DLBCL receive equivalent first-line chemotherapy regardless of the substandard treatment outcome on a large number of patients. Over the years, the most common regimen has been CHOP, a doxorubicin-based drug cocktail with added cyclophosphamide, vincristine, and prednisone . Other first-line multidrug treatments can be used to treat DLBCL but their mechanisms of action and response rates are similar to CHOP; therefore, they are considered comparable (CHOP-like therapy). Only recently, rituximab has been added to first-line regimens to treat DLBCL (ie, R-CHOP) increasing relapse-free and overall survival . However, even though rituximab has improved treatment outcome, still a significant proportion of patients experience early treatment failure, partial response, or recurrence. A method that could appropriately and timely identify high-risk newly diagnosed patients with DLBCL bound to be unresponsive to the established first-line therapies should allow early consideration of alternate treatments, like high-dose therapy, autologous stem cell transplantation , and/or promising new agents that target cellular signaling processes , with the aim to maximize therapeutic success in high-risk patients without compromising those patients who will respond to the present standard of care.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Patients and methods
Patients
Get Radiology Tree app to read full this article<
Study Design
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Assay Method
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<