Rationale and Objectives
In spite of various therapies developed, hepatocellular carcinoma still shows poor prognosis. In this study, we introduced ethylbromopyruvate (EBrP), a hydrophobic derivative of 3-bromopyruvate, as an agent for intraarterial therapy of hepatocellular carcinoma.
Materials and Methods
In in vitro study, we evaluated whether EBrP induced apoptotic cell death in Huh-BAT cells. Chemical degradation products of EBrP were identified by performing proton nuclear magnetic resonance spectroscopy and thin layer chromatography. VX2 carcinoma was implanted and grown in the liver of 25 rabbits for in vivo study. By transfemoral intraarterial approach, 0.4 mL of 10 mM and 40 mM EBrP dissolved in an iodized oil (Lipiodol) was infused into the proper hepatic artery in 8 and 10 rabbits, respectively. In the remaining seven rabbits, 0.4 mL of Lipiodol alone was intraarterially injected as a control. One week later, tumor necrosis rate was calculated with histopathologic examination and hepatotoxicity was evaluated with biochemical analysis.
Results
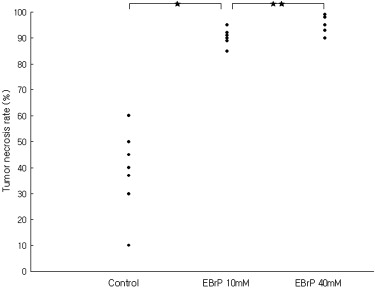
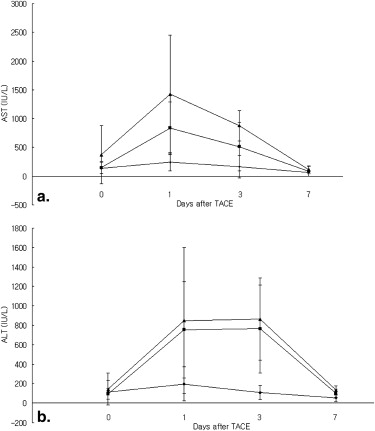
EBrP induced apoptosis in human HCC cells via mitochondrial apoptotic signaling cascades. EBrP dissociated into 3-bromopyruvate and ethanol in the aqueous environment. In VX2 liver tumor models, the group of intraarterial delivery of 40 mM EBrP/Lipiodol solution showed higher tumor necrosis rates (96.1% ± 3.8) than the other groups (38.9% ± 15.9 of a control, 90.5% ± 2.9 in 10 mM) ( P < .05). There was transient elevation of AST and ALT enzyme levels without any mortality.
Conclusions
Intraarterial infusion of EBrP/Lipiodol solution is a feasible intraarterial therapy for liver tumors with potent antitumor effects and transient hepatotoxicity.
Hepatocellular carcinoma (HCC) is an aggressive cancer showing a poor prognosis even though various treatments have been developed. It is one of the most common, lethal malignancies around the world, and about 10% of patients survive 5 years after a diagnosis and the median survival period in patients with unresectable tumors is 4–6 months . Surgical resection is still the treatment of choice in patients with a good residual liver function, but only 15%–25% of HCC in patients with liver cirrhosis are resectable as most patients have a poor hepatic function and advanced stage of disease. Therefore, in advanced stage of HCC, various palliative or investigational therapies have been performed. They include intraarterial chemotherapy such as transcatheter arterial chemoembolization or infusion chemotherapy, internal or external radiotherapy, and newly developed systemic anticancer drugs targeting tumor angiogenesis. Hepatic intraarterial chemotherapy, which is based on the dominant blood supply of HCC from the hepatic artery and the direct delivery of high-dose anticancer agents, is widely used, especially in Asian countries, to treat HCC using various chemotherapeutic drugs such as mitomycin, doxorubicin, cisplatin, 5-fluorouracil, and epirubicin. However, its therapeutic efficacy is still limited and its overall results remain unsatisfactory .
Recently, 3-bromopyruvate (3-BrP), a hexokinase II inhibitor, was introduced as a novel intraarterial therapy for liver cancer and its potential anticancer effects have been studied . Highly malignant cells such as HCC spend more energy in keeping their functions and growth than normal cells, so they are always short of energy called ATP no matter how much energy they produce effectively through an oxygen-dependent mitochondrial ATP-generating system. The shortage spurs them to conduct a salvage pathway called a glycolytic system converting glucose into lactate in a cell, which is then taken out of the cell by specialized transporters . Hexokinase is the first-step enzyme in the pathway and it is essential in maintaining the high glycolytic phenotype. Hexokinase II among the four mammalian types (I-IV) is the predominantly overexpressed form in HCC . 3-BrP selectively blocks hexokinase II to inhibit the pathway and induces depletion of ATP and loss of cell viability . 3-BrP is known also to inhibit glyceraldehyde-3-phosphate dehydrogenase and to overcome protective autophagy of tumor cells, a cellular response to stress resisting to chemotherapy, and induce apoptosis in the end . In vitro and in vivo studies presented that 3-BrP has anticancer effects and that it can be an effective anticancer agent in advanced malignant tumors . Nonetheless, 3-BrP was rapidly neutralized in vivo and had a single-pass effect during rather short intraarterial infusion . Therefore, it will be meaningful to find out chemical agents analogous to 3-BrP will take a role as a hexokinase II inhibitor and stay in the tumor neovasculature with prolonged release and action.
Get Radiology Tree app to read full this article<
Materials and methods
Preparation of EBrP/Lipiodol Solutions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Stability of EBrP Solutions
Get Radiology Tree app to read full this article<
In Vitro Study
Cell line and culture
Get Radiology Tree app to read full this article<
Quantitation of apoptosis
Get Radiology Tree app to read full this article<
Preparation of mitochondrial and cytosolic extracts
Get Radiology Tree app to read full this article<
Immunoblot analysis
Get Radiology Tree app to read full this article<
In Vivo Study
Rabbit VX2 liver tumor model and experimental groups
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Transcatheter arterial embolization
Get Radiology Tree app to read full this article<
Evaluation of antitumor effects
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Measurement of plasma biochemicals
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
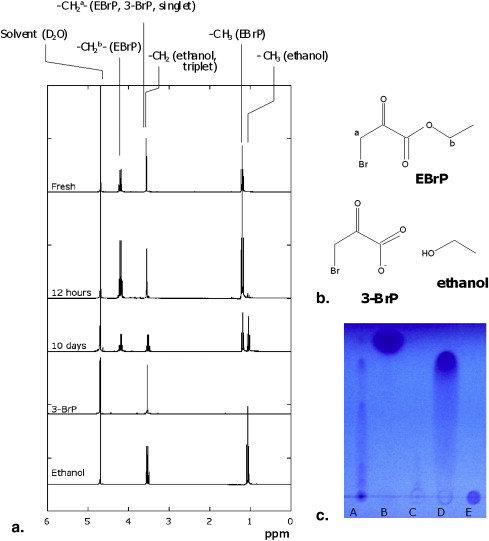
Degradation of Ethylbromopyruvate in Aqueous Solutions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
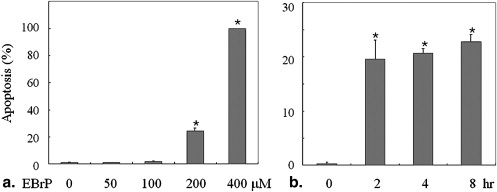
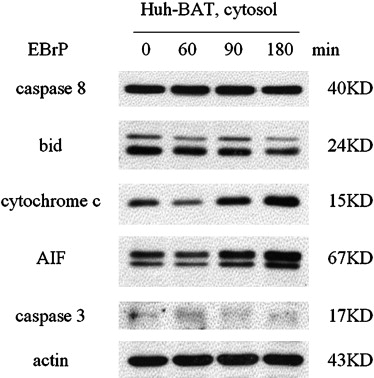
EBrP Induces Apoptosis in Human HCC Cells via Mitochondrial Apoptotic Signaling Cascades
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
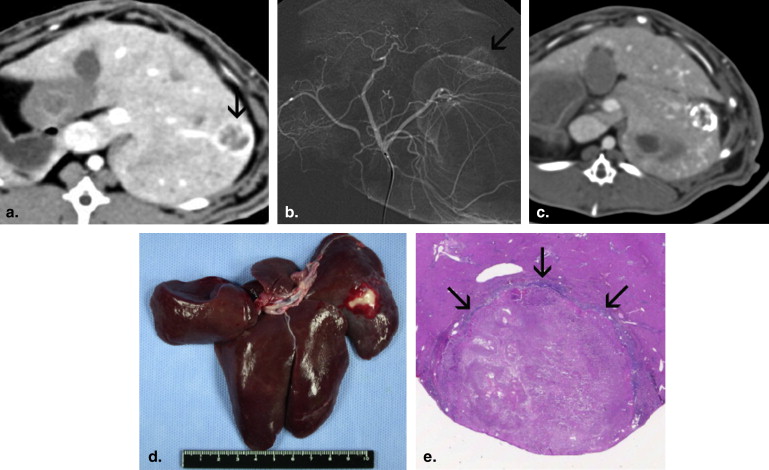
Antitumor Effects and Hepatotoxicity of EBrP
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Di Bisceglie A.M., Rustgi V.K., Hoofnagle J.H., et. al.: NIH conference. Hepatocellular carcinoma. Ann Intern Med 1988; 108: pp. 390-401.
2. Rustgi V.K.: Epidemiology of hepatocellular carcinoma. Gastroenterol Clin North Am 1987; 16: pp. 545-551.
3. Okuda K., Ohtsuki T., Obata H., et. al.: Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer 1985; 56: pp. 918-928.
4. Bruix J.: Treatment of hepatocellular carcinoma. Hepatology 1997; 25: pp. 259-262.
5. Lin C.P., Yu H.C., Cheng J.S., et. al.: Clinical effects of intra-arterial infusion chemotherapy with cisplatin, mitomycin C, leucovorin and 5-flourouracil for unresectable advanced hepatocellular carcinoma. J Chin Med Assoc 2004; 67: pp. 602-610.
6. Liapi E., Geschwind J.F.: Intra-arterial therapies for hepatocellular carcinoma: where do we stand?. Ann Surg Oncol 2010; 17: pp. 1234-1246.
7. Geschwind J.F., Ko Y.H., Torbenson M.S., et. al.: Novel therapy for liver cancer: direct intraarterial injection of a potent inhibitor of ATP production. Cancer Res 2002; 62: pp. 3909-3913.
8. Ko Y.H., Smith B.L., Wang Y., et. al.: Advanced cancers: eradication in all cases using 3-bromopyruvate therapy to deplete ATP. Biochem Biophys Res Commun 2004; 324: pp. 269-275.
9. Kim W., Yoon J.H., Jeong J.M., et. al.: Apoptosis-inducing antitumor efficacy of hexokinase II inhibitor in hepatocellular carcinoma. Mol Cancer Ther 2007; 6: pp. 2554-2562.
10. Chang J.M., Chung J.W., Jae H.J., et. al.: Local toxicity of hepatic arterial infusion of hexokinase II inhibitor, 3-bromopyruvate: in vivo investigation in normal rabbit model. Acad Radiol 2007; 14: pp. 85-92.
11. Gwak G.Y., Yoon J.H., Kim K.M., et. al.: Hypoxia stimulates proliferation of human hepatoma cells through the induction of hexokinase II expression. J Hepatol 2005; 42: pp. 358-364.
12. Bustamante E., Pedersen P.L.: High aerobic glycolysis of rat hepatoma cells in culture: role of mitochondrial hexokinase. Proc Natl Acad Sci U S A 1977; 74: pp. 3735-3739.
13. Bustamante E., Morris H.P., Pedersen P.L.: Energy metabolism of tumor cells. Requirement for a form of hexokinase with a propensity for mitochondrial binding. J Biol Chem 1981; 256: pp. 8699-8704.
14. Moreno-Sánchez R., Rodríguez-Enríquez S., Marín-Hernández A., et. al.: Energy metabolism in tumor cells. FEBS J 2007; 274: pp. 1393-1418.
15. Nakashima R.A., Paggi M.G., Scott L.J., et. al.: Purification and characterization of a bindable form of mitochondrial bound hexokinase from the highly glycolytic AS-30D rat hepatoma cell line. Cancer Res 1988; 48: pp. 913-919.
16. Mathupala S.P., Rempel A., Pedersen P.L.: Glucose catabolism in cancer cells. Isolation, sequence, and activity of the promoter for type II hexokinase. J Biol Chem 1995; 270: pp. 16918-16925.
17. Ko Y.H., McFadden B.A.: Alkylation of isocitrate lyase from Escherichia coli by 3-bromopyruvate. Arch Biochem Biophys 1990; 278: pp. 373-380.
18. Ko Y.H., Pedersen P.L., Geschwind J.F.: Glucose catabolism in the rabbit VX2 tumor model for liver cancer: characterization and targeting hexokinase. Cancer Lett 2001; 173: pp. 83-91.
19. Ganapathy-Kanniappan S., Geschwind J.F., Kunjithapatham R., et. al.: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is pyruvylated during 3-bromopyruvate mediated cancer cell death. Anticancer Res 2009; 29: pp. 4909-4918.
20. Ganapathy-Kanniappan S., Geschwind J.F., Kunjithapatham R., et. al.: 3-Bromopyruvate induces endoplasmic reticulum stress, overcomes autophagy and causes apoptosis in human HCC cell lines. Anticancer Res 2010; 30: pp. 923-935.
21. Vossen J.A., Buijs M., Syed L., et. al.: Development of a new orthotopic animal model of metastatic liver cancer in the rabbit VX2 model: effect on metastases after partial hepatectomy, intra-arterial treatment with 3-bromopyruvate and chemoembolization. Clin Exp Metastasis 2008; 25: pp. 811-817.
22. Vali M., Vossen J.A., Buijs M., et. al.: Targeting of VX2 rabbit liver tumor by selective delivery of 3-bromopyruvate: a biodistribution and survival study. J Pharmacol Exp Ther 2008; 327: pp. 32-37.
23. Jae H.J., Chung J.W., Park H.S., et. al.: The antitumor effect and hepatotoxicity of a hexokinase II inhibitor 3-bromopyruvate: in vivo investigation of intraarterial administration in a rabbit VX2 hepatoma model. Korean J Radiol 2009; 10: pp. 596-603.
24. Higuchi H., Bronk S.F., Takikawa Y., et. al.: The bile acid glycochenodeoxycholate induces trail-receptor 2/DR5 expression and apoptosis. J Biol Chem 2001; 276: pp. 38610-38618.
25. Choi S.H., Chung J.W., Kim H.C., et. al.: The role of perfusion CT as a follow-up modality after transcatheter arterial chemoembolization: an experimental study in a rabbit model. Invest Radiol 2010; 45: pp. 427-436.
26. Greiner E.F., Guppy M., Brand K.: Glucose is essential for proliferation and the glycolytic enzyme induction that provokes a transition to glycolytic energy production. J Biol Chem 1994; 269: pp. 31484-31490.
27. Mathupala S.P., Rempel A., Pedersen P.L.: Glucose catabolism in cancer cells: identification and characterization of a marked activation response of the type II hexokinase gene to hypoxic conditions. J Biol Chem 2001; 276: pp. 43407-43412.
28. Shinohara Y., Ichihara J., Terada H.: Remarkably enhanced expression of the type II hexokinase in rat hepatoma cell line AH130. FEBS Lett 1991; 291: pp. 55-57.
29. Pedersen P.L., Mathupala S., Rempel A., et. al.: Mitochondrial bound type II hexokinase: a key player in the growth and survival of many cancers and an ideal prospect for therapeutic intervention. Biochim Biophys Acta 2002; 1555: pp. 14-20.
30. Park H.S., Chung J.W., Jae H.J., et. al.: FDG-PET for evaluating the antitumor effect of intraarterial 3-bromopyruvate administration in a rabbit VX2 liver tumor model. Korean J Radiol 2007; 8: pp. 216-224.
31. Cammà C., Schepis F., Orlando A., et. al.: Transarterial chemoembolization for unresectable hepatocellular carcinoma: meta-analysis of randomized controlled trials. Radiology 2002; 224: pp. 47-54.
32. Yoon C.J., Chung J.W., Park J.H., et. al.: Transcatheter arterial chemoembolization with paclitaxel-lipiodol solution in rabbit VX2 liver tumor. Radiology 2003; 229: pp. 126-131.
33. Chao Y., Chan W.K., Birkhofer M.J., et. al.: Phase II and pharmacokinetic study of paclitaxel therapy for unresectable hepatocellular carcinoma patients. Br J Cancer 1998; 78: pp. 34-39.
34. Petrasch S., Welt A., Reincher A., et. al.: Chemotherapy with cisplatin and paclitaxel in patients with locally advanced, recurrent or metastatic oesophageal cancer. Br J Cancer 1998; 78: pp. 511-514.
35. Dombernowski P., Giaccone G., Sandler A., et. al.: Gemcitabine and paclitaxel combinations in non-small cell lung cancer. Semin Oncol 1998; 25: pp. 44-50.
36. Yamamoto K., Kikichi Y., Kudoh K., et. al.: Treatment with paclitaxel alone rather than combination with paclitaxel and cisplatin may be selective for cisplatin-resistant ovarian carcinoma. Jpn J Clin Oncol 2000; 30: pp. 446-449.