Rationale and Objectives
This study aimed to assess whether different breast cancer subspecialty physicians can be trained to distinguish non-suspicious from suspicious areas of post-lumpectomy specimen margin in patients with breast cancer using optical coherence tomography (OCT) images (a near-infrared based imaging technique) with final histology as the reference standard.
Materials and Methods
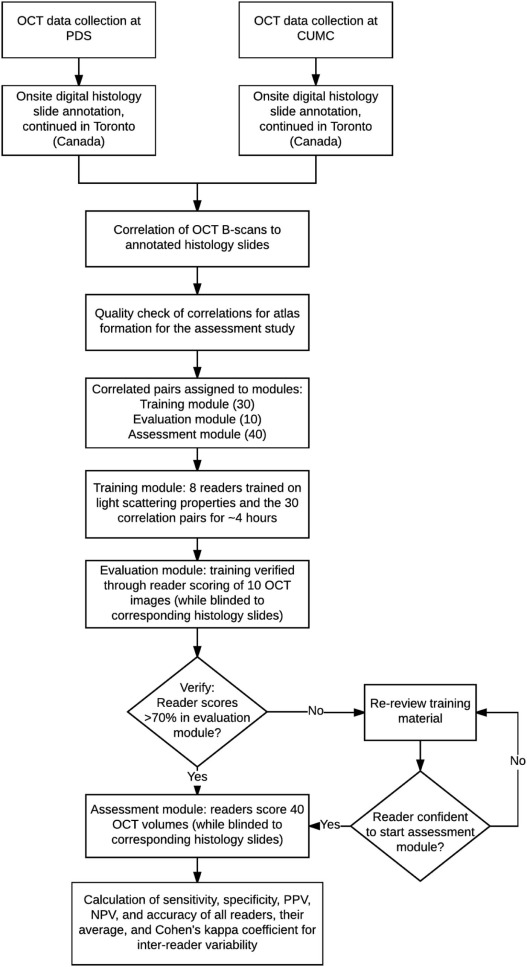
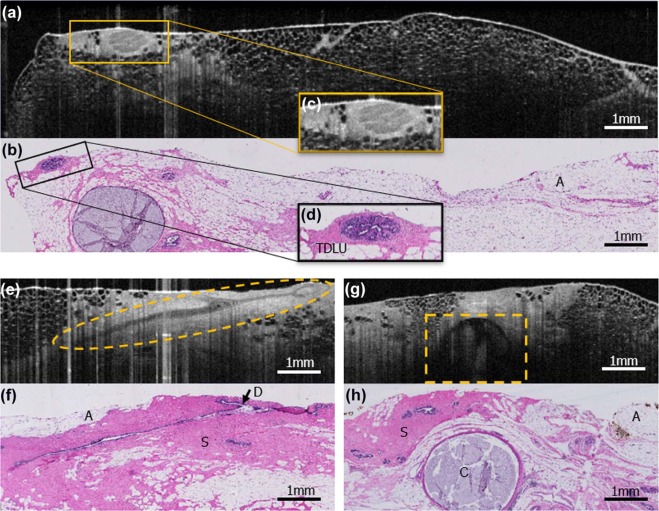
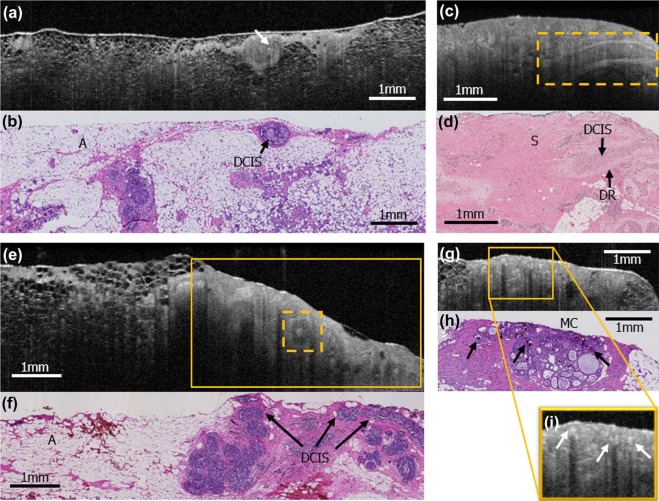
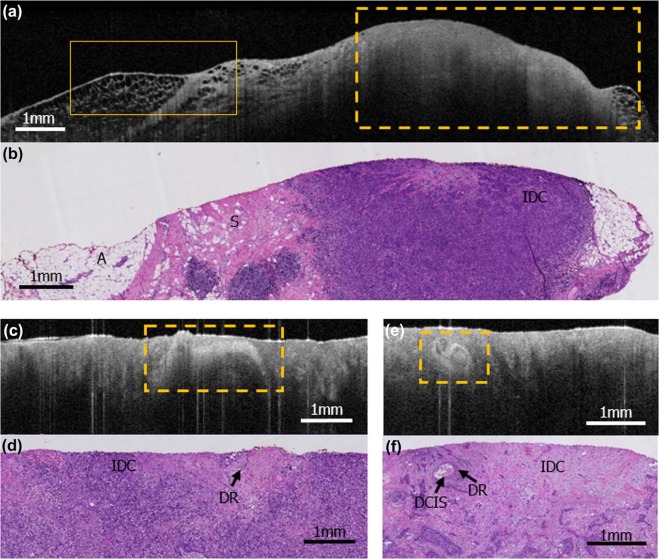
This institutional review board-exempt, Health Insurance Portability and Accountability Act-compliant study was performed on 63 surgically excised breast specimens from 35 female patients, creating a 90-case atlas containing both non-suspicious and suspicious areas for cancer. OCT images of the specimens were performed, providing 6.5–15 µm resolution with tissue visualization 1–2 mm subsurface. From the 90-case atlas, 40 cases were chosen for training and 40 were randomly selected for reader assessment. Three breast imaging radiologists, two pathologists, two breast surgeons, and one non-clinical reader were trained and assessed for ability to distinguish non-suspicious from suspicious findings blinded to clinical data and corresponding histology slides. Duration of training and assessment, sensitivity, specificity, positive predictive value, negative predictive value, and the area under the curve for each reader were calculated as well as averages by subspecialty.
Results
The average training time was 3.4 hours (standard deviation, 1.2). The average assessment time was 1.9 hours (standard deviation, 0.7). The overall average reader sensitivity, specificity, and accuracy for detecting suspicious findings with histologic confirmation of cancer at the surgical margin for all eight readers were 80%, 87%, and 87%, respectively. Radiologists demonstrated the highest average among the disciplines, 85%, 93%, and 94%, followed by pathologists, 79%, 90%, and 84%, and surgeons, 76%, 84%, and 82% respectively.
Conclusions
With relatively short training (3.4 hours), readers from different medical specialties were able to distinguish suspicious from non-suspicious OCT imaging findings in ex vivo breast tissue as confirmed by histology. These results support the potential of OCT as a real-time intraoperative tool for post-lumpectomy specimen margin assessment.
Introduction
Wide local excision (WLE) is a surgical technique intended to achieve complete removal of malignant neoplasms with negative margins. WLE procedures are performed as part of the preferred surgical management of solid tumors including melanomas , cancers of colon or rectum , prostate , and breast . In the context of breast cancer, WLE is also known as breast-conserving surgery (BCS) . The success of BCS has been linked with the adequacy of disease-free (negative) margin widths on the primary resected specimen . Failure to achieve negative margins during the primary surgery necessitates a re-excision surgery to remove any residual disease, so increasing health-care costs and surgery-associated physical or psychological morbidity . Currently, the average re-excision rate among the patients who undergo BCS is about 20% , with re-excision rates as high as 60% reported in the literature . The high degree of variation in re-excision rates could be reduced with a reliable real-time intraoperative tissue assessment tool for margin involvement of the tumor .
Current intraoperative breast lumpectomy assessment tools are either histopathology based (such as touch preparation cytology and frozen section ) or non-destructive imaging based (including ultrasound and specimen radiography ). Although histopathology-based methods provide high-resolution representation of the tissue and have shown high sensitivity or specificity in detecting invasive carcinoma , they are associated with a number of limitations, including being time-intensive, destructive, prone to sampling error, lacking subsurface information, low sensitivity or specificity in detecting ductal carcinoma in situ (DCIS), and requiring the presence of a pathologist in proximity to the operating room . The imaging-based margin assessment techniques have the advantage of being faster compared to the histopathology-based tools; however, it has been shown that imaging has a much lower sensitivity than histopathology in detecting breast cancer within the margin . Both imaging techniques (ie, ultrasound and specimen radiography) perform poorly in detecting DCIS without microcalcifications .
Get Radiology Tree app to read full this article<
TABLE 1
Requirements for an Ideal Clinically Useful Intraoperative Ex Vivo Breast Tissue Assessment Tool
Required Feature Description and Rationale Adjunctive information Assessment tool output can be combined with the clinician’s judgment (based on the current standard of care) and all patient-specific clinical factors Provides subsurface context Allows clinicians to review, measure, and differentiate near-surface tissue microstructures, including distinctive features and different breast tissue types, specifically: adipose tissue, fibrous stroma, breast lobules and ducts, as well as in situ and invasive carcinomas High spatial resolution Able to discern the subsurface features with 6.5–15 µm resolution (close-to-histology) Automated acquisition Standardizes data collection of the whole specimen to eliminate sampling errors, ideally with no increased operator workload (ie, from manipulating an imaging probe) or even minimizing or eliminating the need for an operator Non-destructive Preserves entire tissue sample for postoperative pathology. Does not require special subsectioning, staining, or other preparation of the tissue specimen Rapid intraoperative results Does not substantially extend the time required for the surgical procedure Useable by diverse clinicians Enables use of tool by surgeons, radiologists, or pathologists, as appropriate per institution-specific workflow needs High accuracy Provides improved quantitative clinical outcomes (ie, sensitivity, specificity, PPV, NPV, and accuracy) compared to the current standard of care
NPV, negative predictive value; PPV, positive predictive value.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and Methods
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Breast Specimen Collection and Imaging
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
OCT Image Assessment Study
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 2
Statistical Analysis of the Results for Distinguishing Non-suspicious From Suspicious Breast Tissue
Reader Time Sensitivity 95% CI Specificity 95% CI PPV NPV Accuracy R#1 90 min 81% 0.62–1.00 92% 0.81–1.00 0.87 0.88 0.89 R#2 94 min 81% 0.62–1.00 96% 0.88–1.00 0.93 0.88 0.97 R#3 87 min 94% 0.82–1.00 92% 0.81–1.00 0.88 0.96 0.96 P#1 189 min 94% 0.82–1.00 83% 0.68–0.98 0.79 0.95 0.89 P#2 150 min 63% 0.39–0.86 96% 0.88–1.00 0.91 0.79 0.80 S#1 180 min 88% 0.71–1.00 75% 0.58–0.92 0.70 0.90 0.81 S#2 90 min 63% 0.39–0.86 92% 0.81–1.00 0.83 0.79 0.84 NC 137 min 75% 0.54–0.96 71% 0.53–0.89 0.63 0.81 0.82
CI, confidence interval; NC, non-clinician; NPV, negative predictive value; P1–P2, pathologist 1–2; PPV, positive predictive value; R1–R3, radiologist 1–3; S1–S2, surgeon 1–2.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 3
Average Assessment Results for Distinguishing Non-suspicious From Suspicious Breast Tissue for Readers From Different Disciplines
Reader Discipline Sensitivity Specificity PPV NPV Accuracy Radiology 85% 93% 0.89 0.91 0.94 Pathology 79% 90% 0.85 0.87 0.84 Surgery 76% 84% 0.77 0.85 0.82 The 7 clinician readers 81% 89% 0.84 0.88 0.88 All 8 readers 80% 87% 0.82 0.87 0.87
NPV, negative predictive value; PPV, positive predictive value.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Bolshinsky V., Lin M.J., Serpell J., et. al.: Frequency of residual melanoma in wide local excision (WLE) specimens after complete excisional biopsy. J Am Acad Dermatol 2016; 74: pp. 102-107.
2. Pessaux P., Pocard M., Elias D., et. al.: Surgical management of primary anorectal melanoma. Br J Surg 2004; 91: pp. 1183-1187.
3. Moore B.M., Savdie R., Pebenito R.A., et. al.: The impact of nerve sparing on incidence and location of positive surgical margins in radical prostatectomy. BJU Int 2012; 109: pp. 533-538.
4. Wong J.S., Kaelin C.M., Troyan S.L., et. al.: Prospective study of wide excision alone for ductal carcinoma in situ of the breast. J Clin Oncol 2006; 24: pp. 1031-1036.
5. Clough K.B., Lewis J.S., Couturaud B., et. al.: Oncoplastic techniques allow extensive resections for breast-conserving therapy of breast carcinomas. Ann Surg 2003; 237: pp. 26-34.
6. McCready D., Holloway C., Shelley W., et. al.: Surgical management of early stage invasive breast cancer: a practice guideline. Can J Surg 2005; 48: pp. 185-194.
7. Landercasper J., Attai D., Atisha D., et. al.: Toolbox to reduce lumpectomy reoperations and improve cosmetic outcome in breast cancer patients: the American Society of Breast Surgeons Consensus Conference. Ann Surg Oncol 2015; pp. 1-10.
8. Houssami N., Macaskill P., Luke Marinovich M., et. al.: The association of surgical margins and local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol 2014; 21: pp. 717-730.
9. Houssami N., Morrow M.: Margins in breast conservation: a clinician’s perspective and what the literature tells us. J Surg Oncol 2014; 110: pp. 2-7.
10. Azu M., Abrahamse P., Katz S.J., et. al.: What is an adequate margin for breast-conserving surgery? Surgeon attitudes and correlates. Ann Surg Oncol 2010; 17: pp. 558-563.
11. Xue D.Q., Qian C., Yang L., et. al.: Risk factors for surgical site infections after breast surgery: a systematic review and meta-analysis. Eur J Surg Oncol 2012; 38: pp. 375-381.
12. Al-Ghazal S.K., Fallowfield L., Blamey R.W.: Does cosmetic outcome from treatment of primary breast cancer influence psychosocial morbidity?. Eur J Surg Oncol 1999; 25: pp. 571-573.
13. Landercasper J., Tafra L.: The relationship between quality and cost during the perioperative breast cancer episode of care. Breast 2010; 19: pp. 289-296.
14. Jeevan R., Cromwell D.A., Trivella M., et. al.: Reoperation rates after breast conserving surgery for breast cancer among women in England: retrospective study of hospital episode statistics. BMJ 2012; 345: pp. 4505.
15. Landercasper J., Whitacre E., Degnim A.C., et. al.: Reasons for re-excision after lumpectomy for breast cancer: insight from the American Society of Breast Surgeons Mastery (SM) database. Ann Surg Oncol 2014; 21: pp. 3185-3191.
16. Morrow M., Jagsi R., Alderman A.K., et. al.: Surgeon recommendations and receipt of mastectomy for treatment of breast cancer. JAMA 2009; 302: pp. 1551-1556.
17. Waljee J.F., Hu E.S., Newman L.A., et. al.: Predictors of re-excision among women undergoing breast-conserving surgery for cancer. Ann Surg Oncol 2008; 15: pp. 1297-1303.
18. McCahill L.E.: Variability in reexcision following breast conservation surgery. JAMA 2012; 307: pp. 467-475.
19. Moran M.S., Schnitt S.J., Giuliano A.E., et. al.: Society of Surgical Oncology–American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Ann Surg Oncol 2014; 21: pp. 704-716.
20. Lovrics P.J., Gordon M., Cornacchi S.D., et. al.: Practice patterns and perceptions of margin status for breast conserving surgery for breast carcinoma: National Survey of Canadian General Surgeons. Breast 2012; 21: pp. 730-734.
21. Parvez E., Hodgson N., Cornacchi S.D., et. al.: Survey of American and Canadian general surgeons’ perceptions of margin status and practice patterns for breast conserving surgery. Breast J 2014; 20: pp. 481-488.
22. Creager A.J., Shaw J.A., Young P.R., et. al.: Intraoperative evaluation of lumpectomy margins by imprint cytology with histologic correlation: a community hospital experience. Arch Pathol Lab Med 2002; 126: pp. 846-848.
23. Sumiyoshi K., Nohara T., Iwamoto M., et. al.: Usefulness of intraoperative touch smear cytology in breast-conserving surgery. Exp Ther Med 2010; 1: pp. 641-645.
24. Thill M., Baumann K., Barinoff J.: Intraoperative assessment of margins in breast conservative surgery—still in use?. J Surg Oncol 2014; 110: pp. 15-20.
25. Lester S.C.: Manual of surgical pathology: expert consult.2010.Elsevier SaundersPhiladelphia, PA
26. Krekel N.M.A., Haloua M.H., Lopes Cardozo A.M.F., et. al.: Intraoperative ultrasound guidance for palpable breast cancer excision (COBALT trial): a multicentre, randomised controlled trial. Lancet Oncol 2013; 14: pp. 48-54.
27. St John E.R., Al-Khudairi R., Ashrafian H., et. al.: Diagnostic accuracy of intraoperative techniques for margin assessment in breast cancer surgery: a meta-analysis. Ann Surg 2016; 265: pp. 300-310.
28. Mazouni C., Rouzier R., Balleyguier C., et. al.: Specimen radiography as predictor of resection margin status in non-palpable breast lesions. Clin Radiol 2006; 61: pp. 789-796.
29. Thill M., Röder K., Diedrich K., et. al.: Intraoperative assessment of surgical margins during breast conserving surgery of ductal carcinoma in situ by use of radiofrequency spectroscopy. Breast 2011; 20: pp. 579-580.
30. Cendán J.C., Coco D., Copeland E.M.: Accuracy of intraoperative frozen-section analysis of breast cancer lumpectomy-bed margins. J Am Coll Surg 2005; 201: pp. 194-198.
31. Britton P.D., Sonoda L.I., Yamamoto A.K., et. al.: Breast surgical specimen radiographs: how reliable are they?. Eur J Radiol 2011; 79: pp. 245-249.
32. Drexler W., Fujimoto J.G.: Optical coherence tomography: technology and applications.2nd ed.2015.Springer International Publishing
33. Tearney G.J., Regar E., Akasaka T., et. al.: Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J Am Coll Cardiol 2012; 59: pp. 1058-1072.
34. Guagliumi G., Sirbu V.: Optical coherence tomography: high resolution intravascular imaging to evaluate vascular healing after coronary stenting. Catheter Cardiovasc Interv 2008; 72: pp. 237-247.
35. Nguyen F.T., Zysk A.M., Chaney E.J., et. al.: Intraoperative evaluation of breast tumor margins with optical coherence tomography. Cancer Res 2009; 69: pp. 8790-8796.
36. Kennedy B.F., McLaughlin R.A., Kennedy K.M., et. al.: Investigation of optical coherence microelastography as a method to visualize cancers in human breast tissue. Cancer Res 2015; 75: pp. 3236-3245.
37. South F.A., Chaney E.J., Marjanovic M., et. al.: Differentiation of ex vivo human breast tissue using polarization-sensitive optical coherence tomography. Biomed Opt Express 2014; 5: pp. 3417-3426.
38. Nguyen F.T., Zysk A.M., Chaney E.J., et. al.: Optical coherence tomography: the intraoperative assessment of lymph nodes in breast cancer. IEEE Eng Med Biol Mag 2010; 29: pp. 63-70.
39. Vakoc B.J., Fukumura D., Jain R.K., et. al.: Cancer imaging by optical coherence tomography: preclinical progress and clinical potential. Nat Rev Cancer 2012; 12: pp. 363-368.
40. Yao X., Gan Y., Chang E., et. al.: Visualization and tissue classification of human breast cancer images using ultrahigh-resolution OCT. Lasers Surg Med 2017; 49: pp. 258-269.
41. Obuchowski N.A.: Receiver operating characteristic curves and their use in radiology. Radiology 2003; 229: pp. 3-8.
42. Altman D.G.: Practical statistics for medical research.1999.Chapman and HallLondon
43. Papa M., Allweis T., Karni T., et. al.: An intraoperative MRI system for margin assessment in breast conserving surgery: initial results from a novel technique. J Surg Oncol 2016; pp. 22-26.
44. Dashevsky B.Z., D’Alfonso T., Sutton E.J., et. al.: The potential of high resolution magnetic resonance microscopy in the pathologic analysis of resected breast and lymph tissue. Sci Rep 2015; 5: pp. 174351-174358.
45. Dixon J.M., Renshaw L., Young O., et. al.: Intra-operative assessment of excised breast tumour margins using ClearEdge imaging device. Eur J Surg Oncol 2016; pp. 1-7.
46. Haka A.S., Volynskaya Z., Gardecki J.A., et. al.: In vivo margin assessment during partial mastectomy breast surgery using Raman spectroscopy. Cancer Res 2006; 66: pp. 3317-3322.
47. Brachtel E.F., Johnson N.B., Huck A.E., et. al.: Spectrally encoded confocal microscopy for diagnosing breast cancer in excision and margin specimens. Lab Invest 2016; 96: pp. 459-467.
48. Nichols B.S., Schindler C.E., Brown J.Q., et. al.: A quantitative diffuse reflectance imaging (QDRI) system for comprehensive surveillance of the morphological landscape in breast tumor margins. PLoS ONE 2015; 10: pp. 1-26.
49. Mohs A.M., Mancini M.C., Singhal S., et. al.: Hand-held spectroscopic device for in vivo and intraoperative tumor detection: contrast enhancement, detection sensitivity, and tissue penetration. Anal Chem 2010; 82: pp. 9058-9065.
50. Zysk A.M., Chen K., Gabrielson E., et. al.: Intraoperative assessment of final margins with a handheld optical imaging probe during breast-conserving surgery may reduce the reoperation rate: results of a multicenter study. Ann Surg Oncol 2015; pp. 3356-3362.
51. Assayag O., Antoine M., Sigal-Zafrani B.: Large field, high resolution full field optical coherence tomography: a pre-clinical study of human breast tissue and cancer assessment. Technol Cancer Res Treat 2014; 13: pp. 455-468.
52. Schnabel F., Boolbol S.K., Gittleman M., et. al.: A randomized prospective study of lumpectomy margin assessment with use of MarginProbe in patients with nonpalpable breast malignancies. Ann Surg Oncol 2014; 21: pp. 1589-1595.