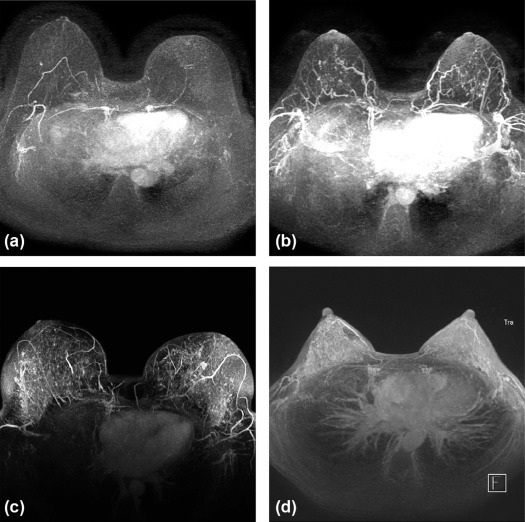
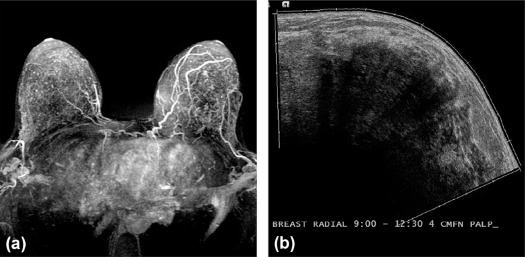
Rationale and Objectives
To determine if background parenchymal enhancement (BPE) on screening breast magnetic resonance imaging (MRI) in high-risk women correlates with future cancer.
Materials and Methods
All screening breast MRIs ( n = 1039) in high-risk women at our institution from August 1, 2004, to July 30, 2013, were identified. Sixty-one patients who subsequently developed breast cancer were matched 1:2 by age and high-risk indication with patients who did not develop breast cancer ( n = 122). Five fellowship-trained breast radiologists independently recorded the BPE. The median reader BPE for each case was calculated and compared between the cancer and control cohorts.
Results
Cancer cohort patients were high-risk because of a history of radiation therapy (10%, 6 of 61), high-risk lesion (18%, 11 of 61), or breast cancer (30%, 18 of 61); BRCA mutation (18%, 11 of 61); or family history (25%, 15 of 61). Subsequent malignancies were invasive ductal carcinoma (64%, 39 of 61), ductal carcinoma in situ (30%, 18 of 61) and invasive lobular carcinoma (7%, 4of 61). BPE was significantly higher in the cancer cohort than in the control cohort ( P = 0.01). Women with mild, moderate, or marked BPE were 2.5 times more likely to develop breast cancer than women with minimal BPE (odds ratio = 2.5, 95% confidence interval: 1.3–4.8, P = .005). There was fair interreader agreement (κ = 0.39).
Conclusions
High-risk women with greater than minimal BPE at screening MRI have increased risk of future breast cancer.
Introduction
Breast cancer is the most common and second most deadly cancer in women, responsible for over 230,000 new diagnoses and 40,000 deaths yearly in the United States . Although there are many factors that may increase an individual woman’s breast cancer risk, women are generally considered high-risk if their lifetime risk of breast cancer is greater than or equal to 20% . For these women, the American Cancer Society and the American College of Radiology recommend supplemental screening with magnetic resonance imaging (MRI) . Published meta-analyses demonstrate a sensitivity of greater than 90% for MRI . Furthermore, screening with MRI in high-risk women is cost-effective, and the effectiveness increases with increased patient risk . As a result, screening MRI for high-risk women is one of the more common indications for breast MRI in clinical practice today.
Although the primary goal of high-risk screening MRI is to identify current cancer, the ability to predict future breast cancer would improve risk stratification for women and could lead to more personalized screening strategies. The association between increased breast density on mammography and future breast cancer is well established, but the corresponding relationship between background parenchymal enhancement (BPE) on MRI and breast cancer risk is less well known . Use of computer vision algorithms to quantify various BPE metrics have had mixed results in predicting future breast cancer, in part because of different methods of quantifying BPE . Two reader studies evaluating high-risk screening MRI suggest that increased BPE is associated with future breast cancer, but these studies were limited based on small sample sizes and reliance on single or dual readers, which limits the generalizability because of interobserver variability in BPE measurements . To build on this promising work, additional studies with more patients as well as multiple readers are needed.
Get Radiology Tree app to read full this article<
Materials and Methods
Study Population
Get Radiology Tree app to read full this article<
Image Review
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Characteristics of the Study Group
Get Radiology Tree app to read full this article<
TABLE 1
Patient Demographics for the Cancer and Control Cohort Matched 1:2 by Age and High-risk Indication
Cancer Cohort ( n = 61) Control Cohort ( n = 122)P Value Age at index MRI, mean (range) 50.5 years (27.4–73.9) 50.9 (28.4–75.9) .81 Race/ethnicity .26 White or Caucasian 48 (79) 91 (75) Black or African American 11 (18) 13 (11) Other or not reported 2 (3) 18 (15) Breast density .79 Predominately fatty 1 (2) 2 (2) Scattered fibroglandular 19 (31) 30 (25) Heterogeneously dense 29 (48) 67 (55) Extremely dense 11 (18) 22 (18) Mammogram not available 1 (2) 1 (1) High-risk indication 1.00 History of mediastinal radiation 6 (10) 12 (10) Personal history of high-risk lesion 11 (18) 22 (18) BRCA mutation 11 (18) 22 (18) Family history of breast cancer 15 (25) 30 (25) Personal history of breast cancer 18 (30) 36 (30) Patients with a personal history of breast cancer Original treatment .33 Lumpectomy 9 (50) 23 (64) Mastectomy 9 (50) 13 (36) Subsequent cancer site Ipsilateral breast 6 (33) N/A Contralateral breast 12 (66) N/A Treated with radiation therapy 13 (72) 26 (72) 1.00 Chemoprevention therapy at time of first study MRI 8 (44) 19 (53) .56 Duration from initial cancer to first study MRI, mean (range) 6.4 years (0.1–14.5) 2.3 (0.5–9.0) <.01 Study follow up duration, mean (range) 2.0 years (0–6.2) 4.5 (2.1–6.7) <.01 Subsequent cancer diagnosis DCIS 18 (30) N/A Invasive lobular carcinoma 4 (7) N/A Invasive ductal carcinoma 39 (64) N/A
DCIS, ductal carcinoma in situ; MRI, magnetic resonance imaging.
Unless otherwise indicated, data are numbers of subjects, with percentages in parentheses.
Get Radiology Tree app to read full this article<
Cancer vs Control Cohorts
Get Radiology Tree app to read full this article<
TABLE 2
Odds Ratios of Breast Cancer by Median BPE Level
Characteristic Cancer Cohort ( n = 61) Control Cohort ( n = 122) Odds Ratio \* P Value BPE .01 Minimal 21 (34) 69 (57) 1.0 Mild 31 (51) 36 (30) 2.8 (1.4–5.7) Moderate 7 (11) 16 (13) 1.4 (0.5–3.9) Marked 2 (3) 1 (1) 6.6 (0.6–145.5) BPE (dichotomous) .004 Minimal 21 (34) 69 (57) 1.0 Mild, moderate, or marked 40 (66) 53 (43) 2.5 (1.3–4.8)
BPE, background parenchymal enhancement.
Unless otherwise indicated, data are numbers of subjects, with percentages in parentheses.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 3
Odds Ratios of Breast Cancer by Threshold BPE Levels for Individual Readers
Characteristic Cancer Cohort ( n = 61) Control Cohort ( n = 122) Odds Ratio \* P Value Reader 1 0.001 Minimal 18 (30) 61 (50) 1.0 Mild, moderate, or marked 43 (70) 61 (50) 2.6 (1.3–4.7) Reader 2 0.059 Minimal 27 (44) 72 (59) 1.0 Mild, moderate, or marked 34 (56) 50 (41) 1.8 (1.0–3.4) Reader 3 0.249 Minimal 26 (43) 63 (52) 1.0 Mild, moderate, or marked 35 (57) 59 (48) 1.4 (0.8–2.7) Reader 4 0.019 Minimal 19 (31) 60 (49) 1.0 Mild, moderate, or marked 42 (69) 62 (51) 2.1 (1.1–4.2) Reader 5 0.010 Minimal 30 (49) 84 (69) 1.0 Mild, moderate, or marked 31 (51) 38 (31) 2.3 (1.2–4.3)
BPE, background parenchymal enhancement.
Unless otherwise indicated, data are numbers of subjects, with percentages in parentheses.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Interreader Agreement
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Centers for Disease Control and Prevention : Breast Cancer Statistics. Available at: https://www.cdc.gov/cancer/breast/statistics/
2. Mainiero M.B., Lourenco A., Mahoney M.C., et. al.: ACR Appropriateness Criteria breast cancer screening. J Am Coll Radiol 2016; 13: pp. R45-R49.
3. Saslow D., Boetes C., Burke W., et. al.: American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin 2007; 57: pp. 75-89.
4. Berg W.A.: Tailored supplemental screening for breast cancer: what now and what next?. AJR Am J Roentgenol 2009; 192: pp. 390-399.
5. Phi X.A., Houssami N., Obdeijn I.M., et. al.: Magnetic resonance imaging improves breast screening sensitivity in BRCA mutation carriers age >/= 50 years: evidence from an individual patient data meta-analysis. J Clin Oncol 2015; 33: pp. 349-356.
6. Medeiros L.R., Duarte C.S., Rosa D.D., et. al.: Accuracy of magnetic resonance in suspicious breast lesions: a systematic quantitative review and meta-analysis. Breast Cancer Res Treat 2011; 126: pp. 273-285.
7. Plevritis S.K., Kurian A.W., Sigal B.M., et. al.: Cost-effectiveness of screening BRCA1/2 mutation carriers with breast magnetic resonance imaging. JAMA 2006; 295: pp. 2374-2384.
8. Taneja C., Edelsberg J., Weycker D., et. al.: Cost effectiveness of breast cancer screening with contrast-enhanced MRI in high-risk women. J Am Coll Radiol 2009; 6: pp. 171-179.
9. Engmann N.J., Golmakani M.K., Miglioretti D.L., et. al.: Population-attributable risk proportion of clinical risk factors for breast cancer. JAMA Oncol 2017; 3: pp. 1228-1236.
10. Jeffers A.M., Sieh W., Lipson J.A., et. al.: Breast cancer risk and mammographic density assessed with semiautomated and fully automated methods and BI-RADS. Radiology 2017; 282: pp. 348-355.
11. Pettersson A., Graff R.E., Ursin G., et. al.: Mammographic density phenotypes and risk of breast cancer: a meta-analysis. J Natl Cancer Inst 2014; 106:
12. Hu X., Jiang L., Li Q., et. al.: Quantitative assessment of background parenchymal enhancement in breast magnetic resonance images predicts the risk of breast cancer. Oncotarget 2017; 8: pp. 10620-10627.
13. Bennani-Baiti B., Dietzel M., Baltzer P.A.: MRI Background parenchymal enhancement is not associated with breast cancer. PLoS ONE 2016; 11: e0158573
14. Mazurowski M.A., Zhang J., Grimm L.J., et. al.: Radiogenomic analysis of breast cancer: luminal B molecular subtype is associated with enhancement dynamics at MR imaging. Radiology 2014; 273: pp. 365-372.
15. King V., Brooks J.D., Bernstein J.L., et. al.: Background parenchymal enhancement at breast MR imaging and breast cancer risk. Radiology 2011; 260: pp. 50-60.
16. Dontchos B.N., Rahbar H., Partridge S.C., et. al.: Are qualitative assessments of background parenchymal enhancement, amount of fibroglandular tissue on MR images, and mammographic density associated with breast cancer risk?. Radiology 2015; 276: pp. 371-380.
17. Ikeda D.M., Hylton N.M., Kuhl C.K., et. al.: BI-RADS: Magnetic Resonance Imaging.D’Orsi C.J.Mendelson E.B.Ikeda D.M.Breast Imaging Reporting and Data System: ACR BI-RADS—Breast Imaging Atlas.2013.American College of RadiologyReston, VA:
18. Landis J.R., Koch G.G.: The measurement of observer agreement for categorical data. Biometrics 1977; 33: pp. 159-174.
19. Sprague B.L., Gangnon R.E., Burt V., et. al.: Prevalence of mammographically dense breasts in the United States. J Natl Cancer Inst 2014; 106:
20. Melsaether A., McDermott M., Gupta D., et. al.: Inter- and intrareader agreement for categorization of background parenchymal enhancement at baseline and after training. AJR Am J Roentgenol 2014; 203: pp. 209-215.
21. DeLeo M.J., Domchek S.M., Kontos D., et. al.: Breast MRI fibroglandular volume and parenchymal enhancement in BRCA1 and BRCA2 mutation carriers before and immediately after risk-reducing salpingo-oophorectomy. AJR Am J Roentgenol 2015; 204: pp. 669-673.
22. King V., Gu Y., Kaplan J.B., et. al.: Impact of menopausal status on background parenchymal enhancement and fibroglandular tissue on breast MRI. Eur Radiol 2012; 22: pp. 2641-2647.
23. Preibsch H., Wanner L., Bahrs S.D., et. al.: Background parenchymal enhancement in breast MRI before and after neoadjuvant chemotherapy: correlation with tumour response. Eur Radiol 2016; 26: pp. 1590-1596.
24. Price E.R., Brooks J.D., Watson E.J., et. al.: The impact of bilateral salpingo-oophorectomy on breast MRI background parenchymal enhancement and fibroglandular tissue. Eur Radiol 2014; 24: pp. 162-168.
25. Scaranelo A.M., Carrillo M.C., Fleming R., et. al.: Pilot study of quantitative analysis of background enhancement on breast MR images: association with menstrual cycle and mammographic breast density. Radiology 2013; 267: pp. 692-700.
26. Tagliafico A., Bignotti B., Tagliafico G., et. al.: Quantitative evaluation of background parenchymal enhancement (BPE) on breast MRI. A feasibility study with a semi-automatic and automatic software compared to observer-based scores. Br J Radiol 2015; 88: pp. 20150417.
27. Yoon H.J., Kim Y., Lee J.E., et. al.: Background 99mTc-methoxyisobutylisonitrile uptake of breast-specific gamma imaging in relation to background parenchymal enhancement in magnetic resonance imaging. Eur Radiol 2015; 25: pp. 32-40.
28. Grimm L.J., Anderson A.L., Baker J.A., et. al.: Interobserver variability between breast imagers using the fifth edition of the BI-RADS MRI Lexicon. AJR Am J Roentgenol 2015; 204: pp. 1120-1124.
29. National Cancer Institute : Breast Cancer Risk Assessment Tool. Available at: https://www.cancer.gov/bcrisktool/
30. Breast Cancer Surveillance Consortium : Breast Cancer Surveillance Consortium Risk Calculator. Available at: https://tools.bcsc-scc.org/bc5yearrisk/calculator.htm
31. International Breast Cancer Intervention Study : Tyrer-Cuzick Model Breast Cancer Risk Evaluation Tool. Available at: http://ibis.ikonopedia.com/
32. Vreemann S., Gubern-Merida A., Borelli C., et. al.: The correlation of background parenchymal enhancement in the contralateral breast with patient and tumor characteristics of MRI-screen detected breast cancers. PLoS ONE 2018; 13: e0191399