Rationale and Objectives
Task-evoked functional MRI (fMRI) has been used successfully in the study of brain function and clinically for presurgical localization of eloquent brain regions prior to the performance of brain surgery. This method requires patient cooperation and is not useful in young children or if the patient has cognitive dysfunction or physical impairment. An alternative method that can overcome some of these disadvantages measures the intrinsic function of the brain using resting-state fMRI. This method does not require any task performance and measures the spontaneous low-frequency (<0.1 Hz) fluctuations of the fMRI signal over time. Our objective in the present work is to provide preliminary information on the possible clinical utility of this technique for presurgical planning and on possible future applications.
Materials and Methods
Data from prior fMRI resting-state studies were reviewed for their potential use in preoperative mapping. Structural and resting-state fMRI data from normal subjects and patients with brain tumors were preprocessed and seed regions were placed in key regions of the brain; the related functional networks were identified using correlation analysis.
Results
Several key functional networks can be identified in patients with brain tumors from resting-state fMRI data.
Conclusion
Resting-state fMRI data can provide valuable presurgical information in many patients who cannot benefit from traditional task-based fMRI. Adoption of this method has the potential to improve individualized patient-centered care.
Blood oxygen level–dependent (BOLD) functional magnetic resonance imaging (fMRI) measures neuronal activity using the ratio of oxyhemoglobin to deoxyhemoglobin as a contrast mechanism. In a typical application, the patient alternates between a passive resting state and performing a task, such as finger tapping, that activates a region of interest in the cerebral cortex. During these periods, the BOLD signal is measured in the magnetic resonance scanner, and the two images are subtracted from each other to reveal areas of the brain that were activated during the prescribed task ( Fig 1 A ). This technique has been an invaluable research tool in the laboratory and has helped increase our understanding of normal and abnormal brain function. Clinical applications of fMRI have focused on localizing areas of critical function for presurgical planning . The accurate localization of eloquent cortex (eg, somatomotor, language) in relation to a tumor mass can help optimize resection and minimize morbidity and mortality. Functional foci identified using fMRI have been shown to correlate well with foci identified using more invasive techniques, such as intraoperative electrophysiology and Wada testing . Furthermore, the distance from an fMRI-identified functional region to the surgical margin has been shown to correlate with loss of function postoperatively . Although there is much accumulated evidence for the benefit of fMRI use in presurgical planning, large-scale studies to demonstrate improved patient outcomes have not been performed.
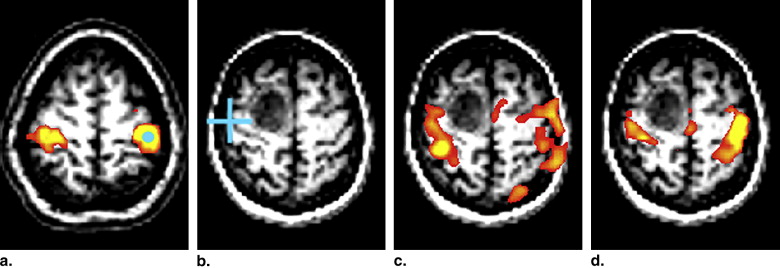
Figure 1
The blood oxygen level–dependent (BOLD) signal during task-based functional magnetic resonance imaging (fMRI) (a) and during intrinsic resting-state fMRI (b) . (a) The subject focused on a flickering checkerboard pattern that was periodically turned on and off. The measurement was made in the V1 region of the visual cortex (red circle) . Ongoing spontaneous fluctuations can be seen in this task paradigm. (b) Spontaneous fluctuations from two regions of the dorsal attention system. The regions are the intraparietal sulcus (IPS) and the frontal eye fields (FEF) (red circles) . The two time curves demonstrate a high degree of correlation ( r = 0.60).
Get Radiology Tree app to read full this article<
Spontaneous bold fluctuation fMRI
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Assessing a new fMRI paradigm
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Acknowledgment
Get Radiology Tree app to read full this article<
References
1. Bandettini P.A., Wong E.C., Hinks R.S., Tikofsky R.S., Hyde J.S.: Time course EPI of human brain function during task activation. Magn Reson Med 1992; 25: pp. 390-397.
2. Kwong K.K., Belliveau J.W., Chesler D.A., et. al.: Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc Natl Acad Sci U S A 1992; 89: pp. 5675-5679.
3. Ogawa S., Tank D.W., Menon R., et. al.: Intrinsic signal changes accompanying sensory stimulation: functional brain mapping with magnetic resonance imaging. Proc Natl Acad Sci U S A 1992; 89: pp. 5951-5955.
4. Matthews P.M., Honey G.D., Bullmore E.T.: Applications of fMRI in translational medicine and clinical practice. Nat Rev Neurosci 2006; 7: pp. 732-744.
5. Vlieger E.J., Majoie C.B., Leenstra S., Den Heeten G.J.: Functional magnetic resonance imaging for neurosurgical planning in neurooncology. Eur Radiol 2004; 14: pp. 1143-1153.
6. Adcock J.E., Wise R.G., Oxbury J.M., Oxbury S.M., Matthews P.M.: Quantitative fMRI assessment of the differences in lateralization of language-related brain activation in patients with temporal lobe epilepsy. Neuroimage 2003; 18: pp. 423-438.
7. Binder J.R., Swanson S.J., Hammeke T.A., et. al.: Determination of language dominance using functional MRI: a comparison with the Wada test. Neurology 1996; 46: pp. 978-984.
8. Haberg A., Kvistad K.A., Unsgard G., Haraldseth O.: Preoperative blood oxygen level-dependent functional magnetic resonance imaging in patients with primary brain tumors: clinical application and outcome. Neurosurgery 2004; 54: pp. 902-915.
9. Pujol J., Conesa G., Deus J., Lopez-Obarrio L., Isamat F., Capdevila A.: Clinical application of functional magnetic resonance imaging in presurgical identification of the central sulcus. J Neurosurg 1998; 88: pp. 863-869.
10. Fox M.D., Raichle M.E.: Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci 2007; 8: pp. 700-711.
11. Biswal B., Yetkin F., Haughton V., Hyde J.: Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 1995; 34: pp. 537-541.
12. Cordes D., Haughton V.M., Arfanakis K., et. al.: Mapping functionally related regions of brain with functional connectivity MR imaging. Am J Neuroradiol 2000; 21: pp. 1636-1644.
13. Cordes D., Haughton V.M., Arfanakis K., et. al.: Frequencies contributing to functional connectivity in the cerebral cortex in “resting-state” data. Am J Neuroradiol 2001; 22: pp. 1326-1333.
14. De Luca M., Smith S.M., De Stefano N., Federico A., Matthews P.M.: Blood oxygenation level dependent contrast resting state networks are relevant to functional activity in the neocortical sensorimotor system. Exp Brain Res 2005; 167: pp. 587-594.
15. Fox M.D., Snyder A.Z., Zacks J.M., Raichle M.E.: Coherent spontaneous activity accounts for trial-to-trial variability in human evoked brain responses. Nat Neurosci 2006; 9: pp. 23-25.
16. Lowe M.J., Mock B.J., Sorenson J.A.: Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage 1998; 7: pp. 119-132.
17. Xiong J., Parsons L.M., Gao J.H., Fox P.T.: Interregional connectivity to primary motor cortex revealed using MRI resting state images. Hum Brain Mapp 1999; 8: pp. 151-156.
18. Hampson M., Peterson B.S., Skudlarski P., Gatenby J.C., Gore J.C.: Detection of functional connectivity using temporal correlations in MR images. Hum Brain Mapp 2002; 15: pp. 247-262.
19. Fukunaga M., Horovitz S.G., Van Gelderen P., et. al.: Large-amplitude, spatially correlated fluctuations in BOLD fMRI signals during extended rest and light sleep. Magn Reson Imaging 2006; 24: pp. 979-992.
20. Horovitz S.G., Fukunaga M., de Zwart J.A., et. al.: The default-mode network connectivity during awake and early sleep: a simultaneous EEG-BOLD-fMRI study. Presented at: Organization for Human Brain Mapping Annual Meeting; Florence, Italy 2006;
21. Kiviniemi V., Kantola J.H., Jauhiainen J., Hyvarinen A., Tervonen O.: Independent component analysis of nondeterministic fMRI signal sources. Neuroimage 2003; 19: pp. 253-260.
22. Peltier S.J., Kerssens C., Hamann S.B., Sebel P.S., Byas-Smith M., Hu X.: Functional connectivity changes with concentration of sevoflurane anaesthesia. Neuroreport 2005; 16: pp. 285-288.
23. Vincent J.L., Patel G.H., Fox M.D., et. al.: Intrinsic functional architecture in the anaesthetized monkey brain. Nature 2007; 447: pp. 83-86.
24. Zhang D, Johnston JM, Fox MD, et al. Preoperative sensorimotor mapping in brain tumor patients using spontaneous fluctuations in neuronal activity imaged with fMRI: initial experience. Neurosurgery. In press.
25. Rombouts S.A., Stam C.J., Kuijer J.P., Scheltens P., Barkhof F.: Identifying confounds to increase specificity during a “no task condition.” Evidence for hippocampal connectivity using fMRI. Neuroimage 2003; 20: pp. 1236-1245.
26. Vincent J.L., Snyder A.Z., Fox M.D., et. al.: Coherent spontaneous activity identifies a hippocampal-parietal memory network. J Neurophysiol 2006; 96: pp. 3517-3531.
27. Fox M.D., Snyder A.Z., Vincent J.L., Corbetta M., Van Essen D.C., Raichle M.E.: The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A 2005; 102: pp. 9673-9678.
28. Fransson P.: Spontaneous low-frequency BOLD signal fluctuations: an fMRI investigation of the resting-state default mode of brain function hypothesis. Hum Brain Mapp 2005; 26: pp. 15-29.
29. Greicius M.D., Krasnow B., Reiss A.L., Menon V.: Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci U S A 2003; 100: pp. 253-258.
30. Laufs H., Krakow K., Sterzer P., et. al.: Electroencephalographic signatures of attentional and cognitive default modes in spontaneous brain activity fluctuations at rest. Proc Natl Acad Sci U S A 2003; 100: pp. 11053-11058.
31. Fox M.D., Corbetta M., Snyder A.Z., Vincent J.L., Raichle M.E.: Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc Natl Acad Sci U S A 2006; 103: pp. 10046-10051.
32. Bettus G, Guedj E, Joyeux F, et al. Decreased basal fMRI functional connectivity in epileptogenic networks and contralateral compensatory mechanisms. Hum Brain Mapp. In press.
33. Sanai N., Mirzadeh Z., Berger M.S.: Functional outcome after language mapping for glioma resection. N Engl J Med 2008; 358: pp. 18-27.
34. De Luca M., Beckmann C.F., De Stefano N., Matthews P.M., Smith S.M.: fMRI resting state networks define distinct modes of long-distance interactions in the human brain. Neuroimage 2006; 29: pp. 1359-1367.
35. van de Ven V.G., Formisano E., Prvulovic D., Roeder C.H., Linden D.E.: Functional connectivity as revealed by spatial independent component analysis of fMRI measurements during rest. Hum Brain Mapp 2004; 22: pp. 165-178.
36. Hollingworth W., Jarvik J.G.: Technology assessment in radiology: putting the evidence in evidence-based radiology. Radiology 2007; 244: pp. 31-38.
37. Mackenzie R., Dixon A.K.: Measuring the effects of imaging: an evaluative framework. Clin Radiol 1995; 50: pp. 513-518.
38. Johnston J.M., Vaishnavi S.N., Smyth M.D., et. al.: Loss of resting interhemispheric functional connectivity after complete section of the corpus callosum. J Neurosci 2008; 28: pp. 6453-6458.