Rationale and Objectives
The objective of this study was to evaluate the ultrasound features of thyroid nodules and their association with malignancy, focusing on establishing feature-oriented ultrasound criteria to determine proper management of a thyroid nodule.
Materials and Methods
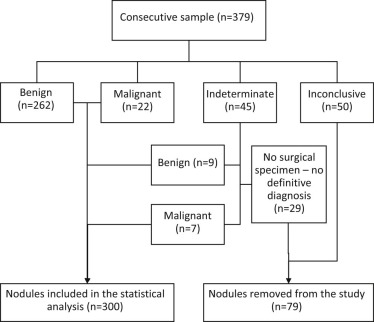
A sample of 379 thyroid nodules were biopsied (from a total of 357 patients aged 59.8 ± 14.8 years) and 300 were included in the final study (271 benign nodules and 29 malignant ones). Ultrasound features were recorded for each nodule: size, echogenicity, homogeneity, contours, shape, texture, peripheral halo, calcifications, and the presence of adenopathy. Statistical analysis of the data was performed using the Mann-Whitney U test and chi-square test. The sensitivity and the specificity of variables seen to have a statistically significant association with the malignancy of nodules were assessed and a logistic regression was performed.
Results
A taller-than-wide shape, an ill-defined contour, the presence of a halo, microcalcifications, and adenopathy were found to have a statistically significant relationship ( P < 0.05) with malignancy, although with a low sensitivity and a high specificity. The presence of at least one suspicious feature yields great sensitivity (89.7%) in detecting malignant disease.
Conclusions
The ultrasound features of thyroid nodules alone do not allow the radiologist to make a confident diagnosis regarding the malignancy of a nodule without performing a biopsy. However, a nodule showing a taller-than-wide shape, microcalcifications, a peripheral halo, an ill-defined contour, or associated adenopathy should be considered for cytology.
Introduction
There is an increasing rate of diagnosis of thyroid nodules in the general population, much due to the widespread availability of ultrasound equipment and better health care worldwide . Up to two-thirds of the general population has thyroid nodules at an ultrasound examination. Although only a small percentage of nodules are malignant, prognosis for patients with thyroid cancer is good, attaining a 5-year survival rate of 98.1% in the United States, mainly related to the early diagnosis of small lesions .
Ultrasound examination, although very sensitive in identifying thyroid nodules, lacks the accuracy to differentiate between benign and malignant nodules . Fine-needle aspiration cytology (FNAC) technique under ultrasound guidance is the most used method to obtain biologic material from a thyroid nodule, leading to a definite benign or malignant result in about 90% of cases .
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and Methods
Patients
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Ultrasound Technique
Get Radiology Tree app to read full this article<
Ultrasound Interpretation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Histologic Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
TABLE 1
Demographic Features of the Sample \*
Demographic Features Malignant Nodules ( n = 29) Benign Nodules ( n = 271)P Value Gender 0.339 Male 6 (15.4) 33 (84.6) Female 23 (8.8) 238 (91.2) Age 54.1 y (37.8–66.1) 60.44 y (50.2–72.1) 0.034
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 2
Frequency and Statistical Analysis of the Ultrasound Features \*
Ultrasound Features Malignant Nodules ( n = 29) Benign Nodules ( n = 271)P Value Diameter 22 mm (14–26) 24 mm (16–31) 0.387 Echogenicity 0.361 Hypoechoic 21 (72.4) 160 (59.0) Isoechoic 6 (20.7) 77 (28.4) Hyperechoic 2 (6.9) 34 (12.5) Homogeneity 0.282 Homogeneous 7 (24.1) 44 (16.2) Heterogeneous 22 (75.9) 227 (83.8) Texture 0.071 Solid 25 (79.3) 169 (62.4) Cystic or mixed type 6 (20.7) 102 (37.6) Contours 0.041 Well defined 13 (44.8) 174 (64.2) Ill defined 16 (55.2) 97 (35.8) Shape 0.007 Parallel to gland plane or spherical 26 (89.7) 266 (98.2) Taller-than-wide shape 3 (10.3) 5 (1.8) Peripheral halo 0.044 Absence 17 (58.6) 204 (75.8) Presence 12 (42.4) 65 (24.2) Calcifications 0.010 Absence or gross calcifications 22 (75.9) 247 (91.1) Microcalcifications 7 (24.1) 24 (8.9) Adenopathy 0.001 Absence 27 (93.1) 270 (99.6) Presence 2 (6.9) 1 (0.4) At least one of the ultrasound features associated with malignancy 0.017 None 3 (10.3) 86 (31.7) At least one 26 (89.7) 185 (68.3)
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 3
Sensitivity, Specificity, and Accuracy of Each of the Statistical Significant Ultrasound Features
Ultrasound Features Sensitivity ( n = 29) Specificity ( n = 271) Accuracy ( n = 300) Ill-defined contour 16 (55.2) 174 (64.2) 190 (63.3) Taller-than-wide shape 3 (10.3) 266 (98.2) 269 (89.7) Peripheral halo 12 (41.4) 204 (75.8) 216 (72.0) Microcalcifications 7 (24.1) 247 (91.1) 254 (84.7) Adenopathy 2 (6.9) 270 (99.6) 272 (90.7) At least one of the ultrasound features associated with malignancy 26 (89.7) 86 (31.7) 112 (37.3)
Data presented as number (percentage).
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
TABLE 4
Results of Multiple Regression Analysis for Detection of Malignant Nodules
Ultrasound Features β ± Standard Error_Z_ Value Odds Ratios 95% Confidence Interval_P_ Value Ill-defined contour 1.887 ± 0.51 3.701 6.598 2.538–19.054 <0.01 Taller-than-wide shape 2.271 ± 0.854 2.659 9.692 1.634–51.904 0.008 Peripheral halo 1.649 ± 0.509 3.238 5.201 1.961–14.727 0.001 Microcalcifications 1.222 ± 0.553 2.21 3.393 1.081–9.765 0.027 Adenopathies 2.811 ± 1.495 1.88 16.63 0.97–513.899 0.06 Age −0.035 ± 0.011 −3.077 0.966 0.944–0.987 0.002 Female gender −0.56 ± 0.56 −1 0.571 0.2–1.863 0.317
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Moon W.J., Jung S.L., Lee J.H., et. al.: Benign and malignant thyroid nodules: US differentiation—multicenter retrospective study. Radiology 2008; 247: pp. 762-770.
2. Ram N., Hafeez S., Qamar S., et. al.: Diagnostic validity of ultrasonography in thyroid nodules. J Pak Med Assoc 2015; 65: pp. 875-878.
3. US Preventive Services Task Force, Bibbins-Domingo K., Grossman D.C., et. al.: Screening for thyroid cancer: US preventive services task force recommendation statement. JAMA 2017; 317: pp. 1882-1887.
4. Iannuccilli J.D., Cronan J.J., Monchik J.M.: Risk for malignancy of thyroid nodules as assessed by sonographic criteria: the need for biopsy. J Ultrasound Med 2004; 23: pp. 1455-1464.
5. Wienke J.R., Chong W.K., Fielding J.R., et. al.: Sonographic features of benign thyroid nodules: interobserver reliability and overlap with malignancy. J Ultrasound Med 2003; 22: pp. 1027-1031.
6. Frates M.C., Benson C.B., Charboneau J.W., et. al.: Management of thyroid nodules detected at US: Society of Radiologists in Ultrasound consensus conference statement. Radiology 2005; 237: pp. 794-800.
7. Kwak J.Y., Han K.H., Yoon J.H., et. al.: Thyroid imaging reporting and data system for US features of nodules: a step in establishing better stratification of cancer risk. Radiology 2011; 260: pp. 892-899.
8. Jegerlehner S., Bulliard J.-L., Aujesky D., et. al.: Overdiagnosis and overtreatment of thyroid cancer: a population-based temporal trend study. PLoS ONE 2017; 12: e0179387
9. Ahn H.S., Kim H.J., Welch H.G.: Korea’s thyroid-cancer “epidemic”—screening and overdiagnosis. N Engl J Med 2014; 371: pp. 1765-1767.
10. Khoo M.L., Asa S.L., Witterick I.J., et. al.: Thyroid calcification and its association with thyroid carcinoma. Head Neck 2002; 24: pp. 651-655.
11. Papini E., Guglielmi R., Bianchini A., et. al.: Risk of malignancy in nonpalpable thyroid nodules: predictive value of ultrasound and color-Doppler features. J Clin Endocrinol Metab 2002; 87: pp. 1941-1946.
12. Marqusee E., Benson C.B., Frates M.C., et. al.: Usefulness of ultrasonography in the management of nodular thyroid disease. Ann Intern Med 2000; 133: pp. 696-700.
13. Yoo J., Ahn H.S., Kim S.J., et. al.: Evaluation of diagnostic performance of screening thyroid ultrasonography (US) and imaging findings of screening-detected thyroid cancer. Cancer Res Treat 2017;
14. Na D.G., Kim J.H., Kim D.S., et. al.: Thyroid nodules with minimal cystic changes have a low risk of malignancy. Ultrasonography 2016; 35: pp. 153-158.
15. Patricio M., Oliveira C., Caseiro-Alves F.: Differentiating malignant thyroid nodule with statistical classifiers based on demographic and ultrasound features. 2017 IEEE 5th Portuguese Meeting on Bioengineering (ENBENG)2017.
16. Moon H.J., Kwak J.Y., Kim E.K., et. al.: A taller-than-wide shape in thyroid nodules in transverse and longitudinal ultrasonographic planes and the prediction of malignancy. Thyroid 2011; 21: pp. 1249-1253.
17. Tessler F.N., Middleton W.D., Grant E.G., et. al.: ACR Thyroid Imaging, Reporting and Data System (TI-RADS): white paper of the ACR TI-RADS Committee. J Am Coll Radiol 2017; 14: pp. 587-595.
18. O’Malley M.E., Weir M.M., Hahn P.F., et. al.: US-guided fine-needle aspiration biopsy of thyroid nodules: adequacy of cytologic material and procedure time with and without immediate cytologic analysis. Radiology 2002; 222: pp. 383-387.
19. Klinck G.H., Winship T.: Psammoma bodies and thyroid cancer. Cancer 1959; 12: pp. 656-662.
20. Meissner W.A., Adler A.: Papillary carcinoma of the thyroid; a study of the pathology of two hundred twenty-six cases. AMA Arch Pathol 1958; 66: pp. 518-525.
21. Dean D.S., Gharib H.: Endotext. De Groot L.J.; et al.2000.
22. Ahn H.S., Welch H.G.: South Korea’s thyroid-cancer “epidemic”—turning the tide. N Engl J Med 2015; 373: pp. 2389-2390.
23. Lee Y.H., Baek J.H., Jung S.L., et. al.: Ultrasound-guided fine needle aspiration of thyroid nodules: a consensus statement by the Korean Society of Thyroid Radiology. Korean J Radiol 2015; 16: pp. 391-401.
24. Haugen B.R., Alexander E.K., Bible K.C., et. al.: 2015 American Thyroid Association Management Guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016; 26: pp. 1-133.
25. Lim-Dunham J.E., Erdem Toslak I., Alsabban K., et. al.: Ultrasound risk stratification for malignancy using the 2015 American Thyroid Association Management Guidelines for Children with Thyroid Nodules and Differentiated Thyroid Cancer. Pediatr Radiol 2017;