Rationale and Objectives
To retrospectively assess the value of spectral presaturation by inversion-recovery (SPIR) magnetic resonance (MR) imaging sequence after gadolinium injection to differentiate fibrotic scar tissue and tumoral infiltration within the mesorectal fat in patients with rectal carcinoma undergoing MR restaging after neoadjuvant chemo- and radiation therapy (CRT).
Materials and Methods
Forty-three consecutive patients (mean age, 65.8 years; range, 46–85 years; male:female, 29:14) with locally advanced rectal carcinoma underwent CRT followed by surgery. MR imaging was performed before and after completion of CRT by using T2-weighted turbo spin-echo and T1-weighted SPIR sequences before and after gadolinium injection, and MR images were assessed by two radiologists in consensus. Logistic regression was conducted to test the significance of the MR image findings with histology.
Results
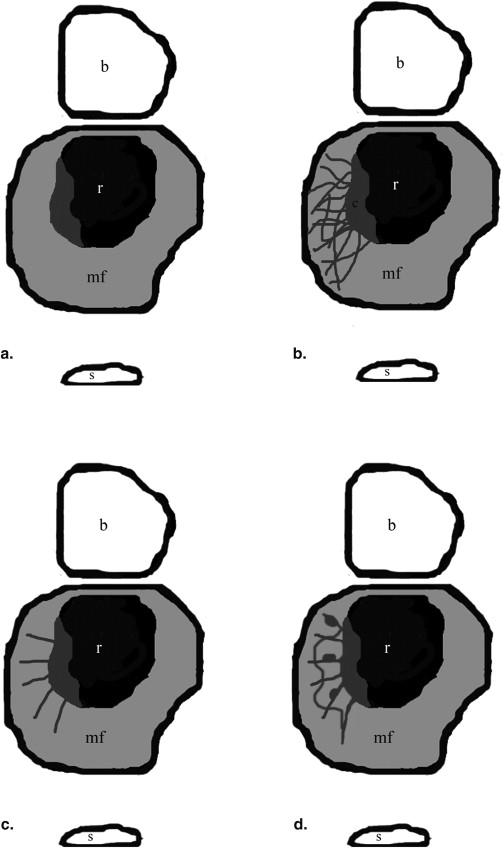
After CRT the disease was either limited to the rectal wall ( n = 18 patients) or presented perirectal infiltration ( n = 25) on histology. In 21 patients, mesorectal enhancing strands were observed. Reticular-shaped enhancing strands reaching the mesorectal fascia presented the highest correlation with tumor infiltration of the mesorectal fat (OR 130.33, 95% CI: 4.1–4220.29; logistic regression), whereas linear-shaped enhancing strands either reaching or not reaching the mesorectal fascia (OR 0.25 or 0.1, 95% CI: 0.024–2.6 or 0.01–1.07) revealed the lowest correlation.
Conclusions
Reticular-shaped enhancing strands on SPIR MR imaging after gadolinium injection are associated with tumor infiltration of the mesorectal fat.
Total mesorectal excision is widely accepted as standard surgical practice for rectal cancer , and it provides the best chance of a tumor-free circumferential resection margin . Local recurrence is directly related to incomplete resection of the tumor and the most important parameter related to local recurrence is the distance between the tumor and the mesorectal fascia . Locally advanced rectal cancer with extramural spread (T3 tumor) has high frequency of local recurrence and metastasis. Nowadays, the standard treatment for locally advanced rectal cancer consists of preoperative neoadjuvant concomitant radiation and chemotherapy (CRT) followed by standard resection of the rectum and resection of the surrounding organs .
The classic approach of defining the T stage of the tumor is based on magnetic resonance (MR) imaging or endoscopic ultrasound and may lead to staging failures in the differentiation of T2 and borderline T3 tumors . The measurement of tumor extent relative to the mesorectal fascia and the prediction of tumor-free resection margins on the basis of MR imaging may be more feasible than defining the T stage . MR imaging has an accuracy of 66% in predicting resection margin involvement during restaging of irradiated rectal cancers (sensitivity, 100%; specificity, 35%; positive predictive value, 58%; negative predictive value, 100%) . The reported overall accuracy of MR imaging in predicting the pathologic stage of nonirradiated rectal cancer is 71%–91% (mean, 85%) for T staging, and 43%–85% (75%) for N staging . However, the reported overall accuracy of MR imaging in the restaging of irradiated rectal cancer is much lower being 47%–54% (50%) for T staging and 64%–68% (65%) for N staging , and it is even worse for mucin-producing tumors.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Get Radiology Tree app to read full this article<
Patients
Get Radiology Tree app to read full this article<
Neoadjuvant Treatment
Get Radiology Tree app to read full this article<
MR Imaging
Get Radiology Tree app to read full this article<
Consensual Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
T Stage Before and After Neoadjuvant Therapy
Tumor Before CRT ∗ After CRT † T0/T2 0 18 ‡ T3 38 23 T4 5 2
Note:–Neoadjuvant treatment refers to radiation therapy with concomitant chemotherapy.
CRT, neoadjuvant chemo- and radiation therapy.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Surgery and Histopathologic Evaluation
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Patient-by-Patient MR Imaging Findings
Patient Number Sex Age (y) T/N Stage ∗ Enhancing Nodules Reticular Enhancing Strands Linear-shaped Enhancing Strands Mesorectal Fat Infiltration on Histology 1 M 60 T3/N0 − − − + 2 M 74 T2/N1 − − − − 3 F 71 T2/N0 − − + − 4 M 70 T3/N2 − + − + 5 M 71 T2/N0 − − + − 6 F 64 T3/N0 − − − + 7 M 62 T2/N0 − − + − 8 F 78 T2/N0 − − + − 9 M 64 T3/N1 − − − + 10 M 60 T3/N2 + + − + 11 M 68 T3/N1 − + − + 12 F 60 T3/N0 − + − + 13 M 72 T1/N0 − + − − 14 F 77 T1/N0 − − − − 15 F 56 T3/N0 − − − + 16 M 78 T3/N1 − + − + 17 M 66 T3/N0 − − − + 18 F 80 T3/N2 − − − + 19 F 52 T3/N1 − + − + 20 M 69 T2/N1 − − − − 21 M 63 T3/N1 − − − + 22 M 74 T3/N0 − − + + 23 M 56 T0/N0 − − − − 24 M 85 T3/N2 − + − + 25 M 73 T2/N1 − − − − 26 M 52 T1/N0 + − + − 27 M 60 T4/N1 − + − + 28 M 68 T3/N1 − − − + 29 F 67 T1/N0 − − + − 30 F 61 T3/N0 − − − + 31 M 60 T2/N0 + + − − 32 F 46 T2/N0 − − − − 33 M 56 T3/N0 − + − + 34 M 63 T4/N1 − − − + 35 F 63 T3/N0 − − − + 36 M 65 T2/N0 − − − − 37 F 65 T3/N0 − + − + 38 M 60 T3/N0 − + − + 39 M 79 T3/N0 − − − + 40 M 74 T2/N0 − − − − 41 M 60 T2/N0 − − − − 42 M 74 T3/N0 − − + + 43 F 71 T2/N0 − − − −
Note:–Correlation of MR imaging features and presence of mesorectal fat infiltration after neoadjuvant chemo- and radiation therapy. +, present; −, absent; F, female; M, male; MR, magnetic resonance.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 3
Univariate Logistic Regression Analysis
MR Imaging Findings Odds Ratio (95% CI)P Enhancing nodules .295 (.024–3.58) > .05 Enhancing strands 1.33 (.4–4.43) > .05 Reticular enhancing strands reaching the mesorectal fascia 5.95 (1.09–32.57)
28.52 (1.34–604.1) < .05
< .05 Separate enhancing strands reaching the mesorectal fascia .08 (.009–.76)
.18 0 (.019–1.78) > .05
> .05
Note:–Odds ratio according to the different MR imaging parameters.
Enhancing nodules or strands were defined as focal or linear enhancing areas appearing more intense than the surrounding mesorectal fat.
CI, confidence intervals; MR, magnetic resonance.
Table 4
Multivariate Logistic Regression Analysis
MR Imaging Findings Odds Ratio (95% CI)P Enhancing nodules .095 (.003–2.87) > .05 Enhancing strands .16 (.017–1.58) > .05 Reticular enhancing strands reaching the mesorectal fascia 62.14 (3.34–1154.26)
130.33 (4.1–4220.29) < .05
< .05 Separate enhancing strands reaching the mesorectal fascia .1 (.01–1.07)
.25 (.024–2.6) > .05
> .05
Note:–Odds ratio according to the different MR imaging parameters.
Enhancing nodules or strands were defined as focal or linear enhancing areas appearing more intense than the surrounding mesorectal fat.
CI, confidence intervals; MR, magnetic resonance.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Jemal A., Siegel R., Ward E., et. al.: Cancer statistics 2007. CA Cancer J Clin 2007; 57: pp. 43-66.
2. Nagtegaal I.D., Gosens M.: Combinations of tumor and treatment parameters are more discriminative for prognosis than the present TNM system in rectal cancer. J Clin Oncol 2007; 25: pp. 1647-1650.
3. Adam I.J., Mohamed M.O., Martin I.G., et. al.: Role of circumferential margin involvement in the local recurrence of rectal cancer. Lancet 1994; 344: pp. 707-711.
4. Quirke P., Durdey P., Dixon M.F., et. al.: Local recurrence of rectal adenocarcinoma due to inadequate surgical resection: histopathological study of lateral tumor spread and surgical excision. Lancet 1986; 2: pp. 996-999.
5. Heald R.J., Ryall R.D.H.: Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1986; 1: pp. 1479-1482.
6. Reynolds J.V., Joyce W.P., Dolan J., et. al.: Pathological evidence in support of total mesorectal excision in the management of rectal cancer. Br J Surg 1996; 83: pp. 1112-1115.
7. Kapiteijn E., Marijnen C.A.M., Nagtegaal I.D., et. al.: Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med 2001; 345: pp. 638-646.
8. Swedish Rectal Cancer Trial. Improved survival with preoperative radiotherapy in resectable rectal cancer. N Engl J Med 1997; 336: pp. 980-987.
9. Allen S.D., Padhani A.R., Dzik-Jurasz A.S., et. al.: Rectal carcinoma: MRI with histologic correlation before and after chemoradiation therapy. AJR Am J Roentgenol 2007; 188: pp. 442-451.
10. Beets-Tan R.G., Beets G.L.: Rectal cancer: review with emphasis on MR imaging. Radiology 2004; 232: pp. 335-346.
11. Beets-Tan R.G., Beets G.L., Vliegen R.F., et. al.: Accuracy of magnetic resonance imaging in prediction of tumor-free resection margin in rectal cancer surgery. Lancet 2001; 357: pp. 497-504.
12. Vliegen R.F., Beets G.L., Lammering G., et. al.: Mesorectal fascia invasion after neoadjuvant chemotherapy and radiation therapy for locally advanced rectal cancer: accuracy of MR imaging for prediction. Radiology 2008; 246: pp. 454-462.
13. Kuo L.J., Chern M.C., Tsou M.H., et. al.: Interpretation of magnetic resonance imaging for locally advanced rectal carcinoma after preoperative chemoradiation therapy. Dis Colon Rectum 2005; 48: pp. 23-28.
14. Chen C.C., Lee R.C., Lin J.K., et. al.: How accurate is magnetic resonance imaging in restaging rectal cancer in patients receiving preoperative combined chemoradiotherapy?. Dis Colon Rectum 2005; 48: pp. 722-728.
15. Reerink O., Verschueren R.C., Szabo B.G., et. al.: A favourable pathological stage after neoadjuvant radiochemotherapy in patients with initially irresectable rectal cancer correlates with a favourable prognosis. Eur J Cancer 2003; 39: pp. 192-195.
16. Dresen R.C., Beets G.L., Rutten H.J., et. al.: Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part I. Are we able to predict tumor confined to the rectal wall?. Radiology 2009; 252: pp. 71-80.
17. World Medical Association : World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. Bull World Health Organ 2001; 79: pp. 373-374.
18. Bipat S., Glas A.S., Slors F.J., et. al.: Rectal cancer: local staging and assessment of lymph node involvement with endoluminal US, CT, and MR imaging. Radiology 2004; 232: pp. 773-783.
19. Barbaro B., Vitale R., Leccisotti L., et. al.: Restaging locally advanced rectal cancer with MR imaging after chemoradiation therapy. Radiographics 2010; 30: pp. 699-721.
20. Wood D.A.: Clinical staging and end results classification: TNM system of clinical classification as applicable to carcinoma of the colon and rectum. Cancer 1971; 28: pp. 109-114.
21. Gareen I.F., Gatsonis C.: Primer on multiple regression models for diagnostic imaging research. Radiology 2003; 229: pp. 305-310.
22. Campbell M.J., Machin D.: Medical statistics, a commonsense approach.1999.WileyChichester, UK 133–134
23. Sistrom C.L., Garvan C.W.: Proportions, odds, and risk. Radiology 2004; 230: pp. 12-19.
24. Cawthorn S.J., Parums D.V., Gibbs N.M., et. al.: Extent of mesorectal spread and involvement of lateral resection margin as prognostic factors after surgery for rectal cancer. Lancet 1990; 335: pp. 1055-1059.
25. Willett C.G., Badizadegan K., Ancukiewicz M., et. al.: Prognostic factors in stage T3N0 rectal cancer: do all patients require postoperative pelvic irradiation and chemotherapy?. Dis Colon Rectum 1999; 42: pp. 167-173.
26. Harrison J.C., Dean P.J., el-Zeky F., et. al.: From Dukes through Jass: pathological prognostic indicators in rectal cancer. Hum Pathol 1994; 25: pp. 498-505.
27. Sauer R., Becker H., Hohenberger W., et. al.: Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004; 351: pp. 1731-1740.
28. Brown C.L., Ternent C.A., Thorson A.G., et. al.: Response to preoperative chemoradiation in stage II and III rectal cancer. Dis Colon Rectum 2003; 46: pp. 1189-1193.
29. Glimelius B., Grönberg H., Järhult J., et. al.: A systemic overview of radiation therapy effects in rectal cancer. Acta Oncol 2003; 42: pp. 476-492.
30. Glimelius B., Holm T., Blomquist L.: Chemotherapy in addition to preoperative radiotherapy in locally advanced rectal cancer: a systematic overview. Rev Recent Clin Trials 2008; 3: pp. 204-211.
31. Barbaro B., Fiorucci C., Tebala C., et. al.: Locally advanced rectal cancer: MR imaging in prediction of response after preoperative chemotherapy and radiation therapy. Radiology 2009; 250: pp. 730-739.
32. Dworak O., Keilholz L., Hoffmann A.: Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 1997; 12: pp. 19-23.
33. Torkzad M.R., Lindholm J., Martling A., et. al.: MRI after preoperative radiotherapy for rectal cancer; correlation with histopathology and the role of volumetry. Eur Radiol 2007; 17: pp. 1566-1573.
34. Kim D.J., Kim J.H., Lim J.S., et. al.: Restaging of rectal cancer with MR imaging after concurrent chemotherapy and radiation therapy. Radiographics 2010; 30: pp. 503-516.
35. Vliegen R.F., Beets G.L., von Meyenfeldt M.F., et. al.: Rectal cancer: MR imaging in local staging–is gadolinium–based contrast material helpful?. Radiology 2005; 234: pp. 179-188.
36. Hawnaur J.M., Zhu X.P., Hutchinson C.E.: Quantitative dynamic contrast-enhanced MRI of recurrent pelvic masses in patients treated for cancer. Br J Radiol 1998; 71: pp. 1136-1142.
37. Dicle O., Obuz F., Çakmakci H.: Differentiation of recurrent rectal cancer and scarring with dynamic MR imaging. Br J Radiol 1999; 72: pp. 1155-1159.
38. Rudisch A., Kremser C., Judmaier W., et. al.: Dynamic contrast-enhanced magnetic resonance imaging: a non-invasive method to evaluate significant differences between malignant and normal tissue. Eur J Radiol 2005; 53: pp. 514-519.
39. Atkin G., Taylor N.J., Daley F.M., et. al.: Dynamic contrast-enhanced magnetic resonance imaging is a poor measure of rectal cancer angiogenesis. Br J Surg 2006; 93: pp. 992-1000.
40. Dinter D.J., Horisberger K., Zechmann C., et. al.: Can dynamic MR imaging predict response in patients with rectal cancer undergoing cetuximab-based neoadjuvant chemoradiation?. Onkologie 2009; 32: pp. 86-93.
41. Wieder H.A., Rosenberg R., Lordick F., et. al.: Rectal cancer: MR imaging before neoadjuvant chemotherapy and radiation therapy for prediction of tumor-free circumferential resection margins and long-term survival. Radiology 2007; 243: pp. 744-751.
![Figure 2, A 55-year-old woman with a rectal cancer. (a,b) Magnetic resonance (MR) imaging before radiation therapy with concomitant chemotherapy. (a) Turbo spin-echo T2-weighted sequence (repetition time [TR]/echo time [TE], 4947/130) and (b) spectral presaturation by inversion recovery T1-weighted sequence (TR/TE, 598/27) after gadolinium injection. Transverse plane. Rectal cancer of the left wall ( large arrow ) without evidence of mesorectal strands. (c,d) MR imaging after radiation therapy with concomitant chemotherapy. (c) Turbo spin-echo T2-weighted sequence (TR/TE, 4947/130) and (d) spectral presaturation by inversion recovery T1-weighted sequence (TR/TE, 598/27) after gadolinium injection. Transverse plane. There is a clear reduction of the tumor volume after neoadjuvant chemo- and radiation therapy with evidence of reticular enhancing strands within the mesorectal fat ( small arrows ).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SpectralPresaturationInversionRecoveryMRImagingSequenceafterGadoliniumInjectiontoDifferentiateFibroticScarTissueandNeoplasticStrandsintheMesorectalFatinPatientsUndergoingRestagingofRectalCarcinomaafterNeoadjuvantChemoandRadiationTherapy/1_1s20S1076633211003576.jpg)
![Figure 3, A 60-year-old woman with a rectal cancer treated by radiation therapy with concomitant chemotherapy. Tumor infiltration of the perirectal fat. (a,b) Turbo spin-echo T2 weighted sequence (repetition time [TR]/echo time [TE], 4947/130) and (c,d) spectral presaturation by inversion recovery T1-weighted sequence (TR/TE, 598/27) after gadolinium injection. Transverse plane. Reticular enhancing strands ( arrows ) within the mesorectal fat. (e) The corresponding transversely sliced pathologic specimen shows residual tumor in the rectal wall ( large black arrow ) and perirectal strands extending into the perirectal fat as ( small black arrows ). (f,g) Histologic evaluation showed perirectal fibrotic strands extending within the mesorectal fat with evidence of tumoral infiltration ( small black arrows ). Hematoxylin-eosin stain; original magnification, ×35. L: rectal lumen.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SpectralPresaturationInversionRecoveryMRImagingSequenceafterGadoliniumInjectiontoDifferentiateFibroticScarTissueandNeoplasticStrandsintheMesorectalFatinPatientsUndergoingRestagingofRectalCarcinomaafterNeoadjuvantChemoandRadiationTherapy/2_1s20S1076633211003576.jpg)
![Figure 4, A 55-year-old woman with a rectal cancer treated by radiation therapy with concomitant chemotherapy. Fibrotic reaction of the perirectal fat after neoadjuvant radiochemotherapy. (a) Turbo spin-echo T2-weighted sequence (repetition time [TR]/echo time [TE], 4947/130) and (b) spectral presaturation by inversion recovery T1-weighted sequence (TR/TE, 598/27) after gadolinium injection. Transverse plane. Linear-shaped enhancing strands ( arrows ) traveling separately within the mesorectal fat. (c) Histologic evaluation showed fibrotic strands ( arrows ) extending within the mesorectal fat without evidence of tumoral infiltration. Hematoxylin-eosin stain; original magnification, ×35. L: rectal lumen. There is reactive hypertrophy of the muscular layer of the rectal wall.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SpectralPresaturationInversionRecoveryMRImagingSequenceafterGadoliniumInjectiontoDifferentiateFibroticScarTissueandNeoplasticStrandsintheMesorectalFatinPatientsUndergoingRestagingofRectalCarcinomaafterNeoadjuvantChemoandRadiationTherapy/3_1s20S1076633211003576.jpg)
![Figure 5, A 60-year-old man with a rectal cancer. (a-d) Magnetic resonance imaging examination after radiation therapy with concomitant chemotherapy. (a,b) Turbo spin-echo T2 weighted sequence (repetition time [TR]/echo time [TE], 4947/130) and (c,d) spectral presaturation by inversion recovery T1-weighted sequence (TR/TE, 598/27) after gadolinium injection. Transverse plane. Reticular enhancing strands ( black arrows ) within the mesorectal fat associated with concomitant enhancing nodules ( white arrows ) projecting from the rectal wall toward the mesorectal fat. (e) Histologic evaluation showed perirectal fibrotic strands extending within the mesorectal fat with tumoral infiltration ( black arrows ). Hematoxylin-eosin stain; original magnification, ×35. There is reactive hypertrophy of the muscular layer of the rectal wall.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SpectralPresaturationInversionRecoveryMRImagingSequenceafterGadoliniumInjectiontoDifferentiateFibroticScarTissueandNeoplasticStrandsintheMesorectalFatinPatientsUndergoingRestagingofRectalCarcinomaafterNeoadjuvantChemoandRadiationTherapy/4_1s20S1076633211003576.jpg)