Rationale and Objectives
This study aims to evaluate if navigator-echo respiratory-triggered magnetic resonance acquisition can acquire supine high-quality breast magnetic resonance imaging (MRI).
Materials and Methods
Supine respiratory-triggered magnetic resonance imaging (Trig-MRI) was compared to supine non-Trig-MRI to evaluate breathing-induced motion artifacts (group 1), and to conventional prone non-Trig-MRI (group 2, 16-channel breast coil), all at 3T. A 32-channel thorax coil was placed on top of a cover to prevent breast deformation. Ten volunteers were scanned in each group, including one patient. The acquisition time was recorded. Image quality was compared by visual examination and by calculation of signal-to-noise ratio (SNR), contrast-to-noise ratio (CNR), and image sharpness (IS).
Results
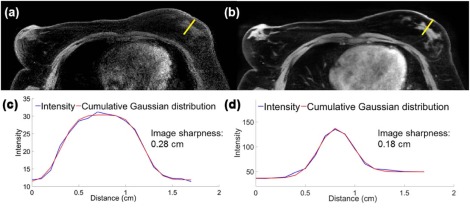
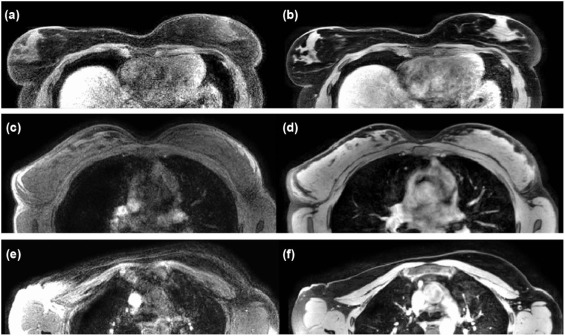
Scan time increased from 56.5 seconds (non-Trig-MRI) to an average of 306 seconds with supine Trig-MRI (range: 120–540 seconds). In group 1, the median values (interquartile range) of SNR, CNR, and IS improved from 11.5 (6.0), 7.3 (3.1), and 0.23 (0.2) cm on supine non-Trig-MRI to 38.1 (29.1), 32.8 (29.7), and 0.12 (0) cm (all P < 0.01) on supine Trig-MRI . All qualitative image parameters in group 1 improved on supine Trig-MRI (all P < 0.01) . In group 2, SNR and CNR improved from 14.7 (6.8) and 12.6 (5.6) on prone non-Trig-MRI to 36.2 (12.2) and 32.7 (12.1) (both P < 0.01) on supine Trig-MRI. IS was similar: 0.10 (0) cm vs 0.11 (0) cm ( P = 0.88) .
Conclusions
Acquisition of high-quality supine breast MRI is possible when respiratory triggering is applied, in a similar setup as during subsequent treatment. Image quality improved when compared to supine non-triggered breast MRI and prone breast MRI, but at the cost of increased acquisition time.
Introduction
Breast-conserving surgery can be challenging in patients with small or non-palpable tumors, where tumor palpation is hampered . Therefore, accurate tumor localization during surgery is essential for achieving negative resection margins and for avoiding re-excisions and local recurrences. Several technologies have been developed to guide the surgeon, such as wire-guided localization and radioactive seed localization . Both methods have led to a decrease in positive resection margins but provide only surrogate position information about the tumor, not the actual 3-D shape and position information with respect to the tumor border.
Recently, Fichtinger et al. have proposed the use of an electromagnetic navigation system for intraoperative tumor tracking in combination with wire-guided localization . Preoperatively, an electromagnetically tracked localization needle is placed over the wire guide under ultrasound guidance. Subsequently, a tracked ultrasound system is used for delineation of the tumor. During surgery, the tracked tumor is shown in combination with a tracked cautery on the navigation screen. This system was found to be safe and feasible by evaluation on six palpable tumors. However, non-palpable tumors are often occult on ultrasound , challenging tumor visualization and delineation.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and Methods
Imaging Protocols
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Imaging Subjects
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Qualitative Image Assessment
Get Radiology Tree app to read full this article<
Quantitative Image Assessment
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Group 1
Get Radiology Tree app to read full this article<
TABLE 1
Qualitative Evaluation of the Supine Trig- and Non-Trig-MRI Scans of the 10 Volunteers of Group 1 by Four Different Radiologists
Supine Non-Trig-MRI ( n = 40) Supine Trig-MRI ( n = 40)P Value Overall Image Quality <0.01 Good 0 33 Moderate 16 7 Poor 24 0 Distinction between fibroglandular and fatty tissues <0.01 Good 0 38 Moderate 13 2 Poor 27 0 Evaluation of breast <0.01 Good 0 32 Moderate 8 7 Poor 32 1 Evaluation of axilla <0.01 Good 1 34 Moderate 16 5 Poor 23 1 Breathing-induced motion artifacts <0.01 Absent 1 20 Moderate 18 17 Severe 21 3
MRI, magnetic resonance imaging; Trig-MRI, triggered magnetic resonance imaging.
Data are presented as numbers of evaluations.
TABLE 2
SNR, CNR, and Image Sharpness of the MRI of the Volunteers in Groups 1 and 2
Median SNR (IQR) Median CNR (IQR) Median Image Sharpness (cm) (IQR) Group 1 Supine non-Trig-MRI 11.5 (6.0) 7.3 (3.1) 0.23(0.2) Supine Trig-MRI 38.1 (29.1) 32.8 (29.7) 0.12(0)P value <0.01 <0.01 <0.01 Group 2 Prone non-Trig-MRI 14.7 (6.8) 12.6 (5.6) 0.10(0) Supine Trig-MRI 36.2 (12.2) 32.7 (12.1) 0.11(0)P value <0.01 <0.01 0.88
SNR, signal-to-noise ratio; CNR, contrast-to-noise ratio; IQR, interquartile range; MRI, magnetic resonance imaging, Trig-MRI, triggered magnetic resonance imaging.
Get Radiology Tree app to read full this article<
Group 2
Get Radiology Tree app to read full this article<
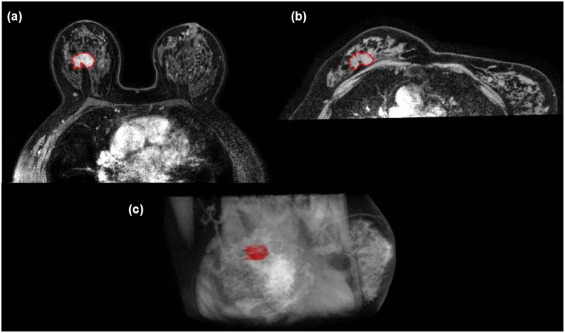
Patient Example
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Acknowledgment
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Pleijhuis R.G., Graafland M., de Vries J., et. al.: Obtaining adequate surgical margins in breast-conserving therapy for patients with early-stage breast cancer: current modalities and future directions. Ann Surg Oncol 2009; 16: pp. 2717-2730.
2. Lovrics P.J., Goldsmith C.H., Hodgson N., et. al.: A multicentered, randomized, controlled trial comparing radioguided seed localization to standard wire localization for nonpalpable, invasive and in situ breast carcinomas. Ann Surg Oncol 2011; 18: pp. 3407-3414.
3. Janssen N.N.Y., Nijkamp J., Alderliesten T., et. al.: Radioactive seed localization in breast cancer treatment. Br J Surg 2016; 103: pp. 70-80.
4. Mariscal Martínez A., Solà M., de Tudela A.P., et. al.: Radio-guided localization of nonpalpable breast cancer lesions: randomized comparisonwith wire localization in patients undergoing conservative surgery and sentinel node biopsy. AJR Am J Roentgenol 2009; 193: pp. 1001-1009.
5. Ungi T., Gauvin G., Yeo C.T., et. al.: Real time navigation in breast tumor surgery. Int J Comput Assist Radiol Surg 2015; 10: pp. S59-S60.
6. Gordon P.B., Goldenberg S.L.: Malignant breast masses detected only by ultrasound. A retrospective review. Cancer 1995; 76: pp. 626-630.
7. Warren R.M.L., Pointon L., Thompson D., et. al.: Reading protocol for dynamic contrast-enhanced MR images of the breast: sensitivity and specificity analysis. Radiology 2005; 236: pp. 779-788.
8. Peters N.H.G.M., Borel Rinkes I.H.M., Zuithoff N.P.A., et. al.: Meta-analysis of MR imaging in the diagnosis of breast lesions. Radiology 2008; 246: pp. 116-124.
9. Houssami N., Ciatto S., Macaskill P., et. al.: Accuracy and surgical impact of magnetic resonance imaging in breast cancer staging: systematic review and meta-analysis in detection of multifocal and multicentric cancer. J Clin Oncol 2008; 26: pp. 3248-3258.
10. Carbonaro L.A., Tannaphai P., Trimboli R.M., et. al.: Contrast enhanced breast MRI: spatial displacement from prone to supine patient’s position. Preliminary results. Eur J Radiol 2012; 81: pp. e771-e774.
11. Siegler P., Holloway C.M.B., Causer P., et. al.: Supine breast MRI. J Magn Reson Imaging 2011; 34: pp. 1212-1217.
12. Kim B.S., Kim J.H., Choi G.M., et. al.: Comparison of three free-breathing T2-weighted MRI sequences in the evaluation of focal liver lesions. AJR Am J Roentgenol 2008; 190: pp. 19-27.
13. Klessen C., Asbach P., Kroencke T.J., et. al.: Magnetic resonance imaging of the upper abdomen using a free-breathing T2-weighted turbo spin echo sequence with navigator triggered prospective acquisition correction. J Magn Reson Imaging 2005; 21: pp. 576-582.
14. Vasanawala S.S., Iwadate Y., Church D.G., et. al.: Navigated abdominal T1-W MRI permits free-breathing image acquisition with less motion artifact. Pediatr Radiol 2010; 40: pp. 340-344.
15. Itatani R., Namimoto T., Kajihara H., et. al.: Preoperative evaluation of the cystic duct for laparoscopic cholecystectomy: comparison of navigator-gated prospective acquisition correction- and conventional respiratory-triggered techniques at free-breathing 3D MR cholangiopancreatography. Eur Radiol 2013; 23: pp. 1911-1918.
16. Ivancevic M.K., Kwee T.C., Takahara T., et. al.: Diffusion-weighted MR imaging of the liver at 3.0 Tesla using tracking only navigator echo (TRON): a feasibility study. J Magn Reson Imaging 2009; 30: pp. 1027-1033.
17. Inoue Y., Hata H., Nakajima A., et. al.: Optimal techniques for magnetic resonance imaging of the liver using a respiratory navigator-gated three-dimensional spoiled gradient-recalled echo sequence. Magn Reson Imaging 2014; 32: pp. 9750-9980.
18. Tokuda J., Morikawa S., Haque H.A., et. al.: Adaptive 4D MR imaging using navigator-based respiratory signal for MRI-guided therapy. Magn Reson Med 2008; 59: pp. 1051-1061.
19. van der Leij F., Bosma S.C.J., van de Vijver M.J., et. al.: First results of the preoperative accelerated partial breast irradiation (PAPBI) trial. Radiother Oncol 2015; 114: pp. 322-327.
20. den Hartogh M.D., Philippens M.E.P., van Dam I.E., et. al.: MRI and CT imaging for preoperative target volume delineation in breast-conserving therapy. Radiat Oncol 2014; 9: pp. 63.
21. Siegler P., Ebrahimi M., Holloway C.M.B., et. al.: Supine breast MRI and assessment of future clinical applications. Eur J Radiol 2012; 81: pp. S153-S155.
22. Han L., Hipwell J., Mertzanidou T., et. al.: A hybrid fem-based method for aligning prone and supine images for image guided breast surgery. Proc IEEE Int Symp Biomed Imaging 2011; pp. 1239-1242.
23. Eiben B., Vavourakis V., Hipwell J.H., et. al.: Symmetric biomechanically guided prone-to-supine breast image registration. Ann Biomed Eng 2015; 44: pp. 154-173.
24. Salem D.S., Kamal R.M., Mansour S.M., et. al.: Breast imaging in the young: the role of magnetic resonance imaging in breast cancer screening, diagnosis and follow-up. J Thorac Dis 2013; 5:
25. Ledger A.E.W., Scurr E.D., Hughes J., et. al.: Comparison of Dixon sequences for estimation of percent breast fibroglandular tissue. PLoS ONE 2016; 11: pp. e0152152.
26. Ungi T., Gauvin G., Lasso A., et. al.: Navigated breast tumor excision using electromagnetically tracked ultrasound and surgical instruments. IEEE Trans Biomed Eng 2016; 63: pp. 600-606.
27. Alderliesten T., Loo C., Paape A., et. al.: On the feasibility of MRI-guided navigation to demarcate breast cancer for breast-conserving surgery. Med Phys 2010; 37: pp. 2617-2626.