Rationale and Objectives
In this study, we investigated the diagnostic and grading value of diffusion tensor imaging (DTI) in patients with carpal tunnel syndrome (CTS).
Materials and Methods
Of the 120 subjects included in the present study, 72 were in the CTS group and 48 were in the healthy control group. In addition, the patients with CTS were further divided into three subgroups based on severity (mild, moderate, and severe) according to electrophysiological studies (EPS). DTI-derived parameters (fractional anisotropy [FA] and apparent diffusion coefficient [ADC]) were evaluated at four median nerve levels. The mean FA and ADC values of the CTS groups and healthy controls were compared separately. Correlations and possible relationships between DTI parameters and EPS results were analyzed. Receiver operating characteristics analysis was used to calculate the FA and ADC cutoff values for CTS diagnosis and grading.
Results
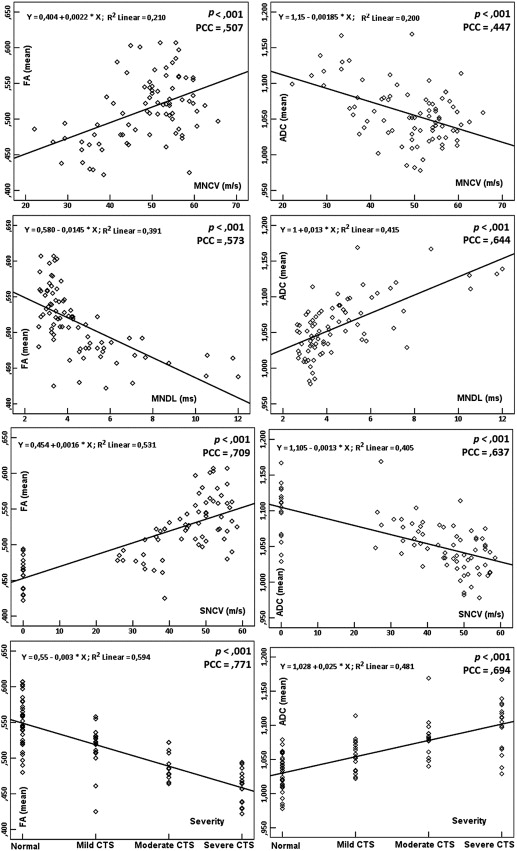
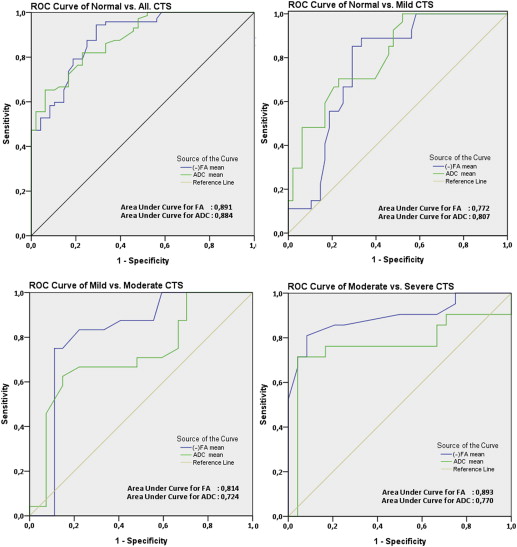
Statistically significant differences were observed in mean FA and ADC between the normal and mild, mild and moderate, and moderate and severe subgroups. Significant correlations were found between DTI parameters and EPS measurements based on severity. FA and ADC threshold values, as well as the sensitivity and specificity levels, for diagnosing and grading CTS were determined.
Conclusions
DTI parameters can provide helpful information for CTS. The correlations of FA and ADC measurements versus EPS measurements based on severity were significant. Moreover, FA and ADC threshold values were sufficient for the diagnosis and grading of CTS.
Carpal tunnel syndrome (CTS) is the most common peripheral neuropathy, caused by entrapment of the median nerve at the carpal tunnel level . Reportedly, the prevalence of CTS is 3.8% in the general population and is more common in women than in men, occurring predominantly in subjects aged between 40 and 60 years . Combined use of clinical symptoms, physical examinations, and electrophysiological studies (EPS) are considered as the reference standard for diagnosis of CTS. However, in some cases discrepancies in the diagnosis and severity of CTS may be because of the different measured clinical and EPS parameters . Imaging methods such as ultrasound (US) and conventional magnetic resonance imaging (MRI) may resolve these discrepancies . In particular, conventional MRI may show enlargement of the median nerve, nerve flattening, increased nerve signal intensity, and bowing of the transverse carpal ligament in patients with CTS . However, the sensitivity and specificity of conventional MRI findings are inadequate for diagnosis of CTS and provide insufficient diagnostic data .
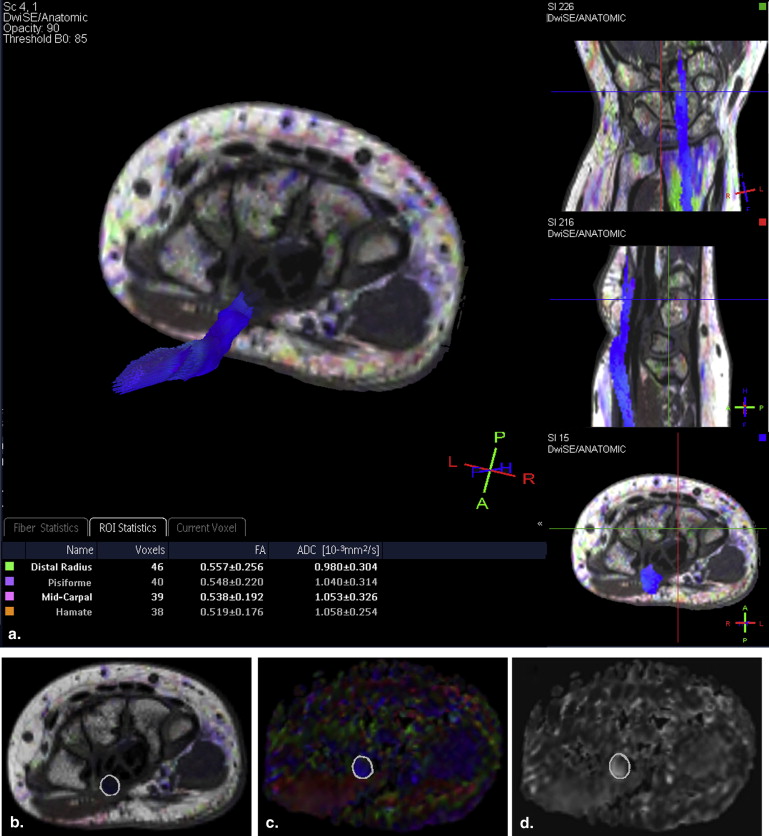
Diffusion tensor imaging (DTI) is an advanced MRI method used to measure the diffusion of water in tissue. The water diffusion anisotropy can be measured with DTI. Directional anisotropy of water diffusibility can be measured by microstructural parameters such as fractional anisotropy (FA; quantitative index used to characterize the degree of diffusion anisotropy) and apparent diffusion coefficient (ADC; one-third of the trace of the diffusion tensor) . The main clinical application of DTI is white matter tract visualization , which can be useful for imaging the median nerve . Studies showing that DTI is a feasible diagnostic method for CTS diagnosis are limited .
Get Radiology Tree app to read full this article<
Materials and methods
Subjects
Get Radiology Tree app to read full this article<
Electrophysiological Studies
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
MRI Studies
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Methods
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Table 1
Demographic Data of Normal and CTS Groups
Group Number of Subjects Age (Mean ± SD) Gender CTS Mild 27 40.00 ± 8.53 7M/20F Moderate 24 45.67 ± 4.87 6M/18F Severe 21 44.05 ± 7.12 6M/15F Total 72 43.07 ± 7.40 19M/53F Normal 48 41.85 ± 7.81 11M/37F All subjects 120 42.58 ± 7.56 30M/90F
CTS, carpal tunnel syndrome; F, female; M, male.
Table 2
ICC for Interreader Agreement in FA and ADC Measurements
Group FA ADC Patients with CTS ( n = 72) 0.915 0.897 Healthy volunteers ( n = 48) 0.899 0.838
ADC, apparent diffusion coefficient; CTS, carpal tunnel syndrome; FA, fractional anisotropy; ICC, intraclass correlation coefficients.
The following ICC categories were used for interpretation: 0.01–0.20 = slight; 0.21–0.40 = fair; 0.41–0.60 = moderate; 0.61–0.80 = substantial; and 0.81–1.00 = almost perfect agreement.
Table 3
FA and ADC Values in Normal Subjects and in Patients with Mild, Moderate, and Severe CTS
Group FA ∗ † ‡ § (Mean ± SD) ADC (×10 −3 mm 2 /s) ‖ ¶ # ∗∗ (Mean ± SD) Normal ( n = 48) 0.548 ± 0.034 1.026 ± 0.027 CTS all ( n = 72) 0.488 ± 0.034 1.080 ± 0.037 Mild ( n = 27) 0.514 ± 0.032 1.059 ± 0.025 Moderate ( n = 24) 0.488 ± 0.018 1.077 ± 0.028 Severe ( n = 21) 0.456 ± 0.020 1.111 ± 0.039
ADC, apparent diffusion coefficient; CTS, carpal tunnel syndrome; FA, fractional anisotropy.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 4
ROC Analysis of FA and ADC for All Groups
Group Cutoff Value Area Under Curve 95% Confidence Interval Sensitivity (%) Specificity (%) Normal versus all CTS FA 0.532 0.901 0.834–0.948 94.4 70.8 ADC 1.047 0.884 0.827–0.940 81.9 77.1 Normal versus mild CTS FA 0.532 0.772 0.666–0.877 85.2 70.8 ADC 1.047 0.807 0.709–0.904 70.4 77.1 Mild versus moderate CTS FA 0.507 0.814 0.684–0.944 83.3 77.8 ADC 1.076 0.724 0.579–0.869 66.7 77.8 Moderate versus severe CTS FA 0.475 0.893 0.790–0.995 85.7 79.2 ADC 1.093 0.770 0.610–0.930 76.2 83.3
ADC, apparent diffusion coefficient; CTS, carpal tunnel syndrome; FA, fractional anisotropy; ROC, receiver operating characteristics.
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Andreisek G., Crook D.W., Burg D., et. al.: Peripheral neuropathies of the median, radial, and ulnar nerves: MR imaging features. Radiographics 2006; 26: pp. 1267-1287.
2. Atroshi I., Gummesson C., Johnsson R., et. al.: Prevalence of carpal tunnel syndrome in a general population. JAMA 1999; 282: pp. 153-158.
3. Phalen G.S.: The carpal-tunnel syndrome. Seventeen years’ experience in diagnosis and treatment of six hundred fifty-four hands. J Bone Joint Surg Am 1966; 48: pp. 211-228.
4. Graham B.: The value added by electrodiagnostic testing in the diagnosis of carpal tunnel syndrome. J Bone Joint Surg Am 2008; 90: pp. 2587-2593.
5. Bland J.D.: A neurophysiological grading scale for carpal tunnel syndrome. Muscle Nerve 2000; 23: pp. 1280-1283.
6. Bower J.A., Stanisz G.J., Keir P.J.: An MRI evaluation of carpal tunnel dimensions in healthy wrists: implications for carpal tunnel syndrome. Clin Biomech (Bristol, Avon) 2006; 21: pp. 816-825.
7. Martins R.S., Siqueira M.G., Simplicio H., et. al.: Magnetic resonance imaging of idiopathic carpal tunnel syndrome: correlation with clinical findings and electrophysiological investigation. Clin Neurol Neurosurg 2008; 110: pp. 38-45.
8. Cudlip S.A., Howe F.A., Clifton A., et. al.: Magnetic resonance neurography studies of the median nerve before and after carpal tunnel decompression. J Neurosurg 2002; 96: pp. 1046-1051.
9. Jarvik J.G., Yuen E., Haynor D.R., et. al.: MR nerve imaging in a prospective cohort of patients with suspected carpal tunnel syndrome. Neurology 2002; 58: pp. 1597-1602.
10. Kang S., Kwon H.K., Kim K.H., et. al.: Ultrasonography of median nerve and electrophysiologic severity in carpal tunnel syndrome. Ann Rehabil Med 2012; 36: pp. 72-79.
11. Basser P.J., Mattiello J., LeBihan D.: MR diffusion tensor spectroscopy and imaging. Biophys J 1994; 66: pp. 259-267.
12. Le Bihan D.: Molecular diffusion, tissue microdynamics and microstructure. NMR Biomed 1995; 8: pp. 375-386.
13. Le Bihan D., Mangin J.F., Poupon C., et. al.: Diffusion tensor imaging: concepts and applications. J Magn Reson Imaging 2001; 13: pp. 534-546.
14. Wang C.K., Jou I.M., Huang H.W., et. al.: Carpal tunnel syndrome assessed with diffusion tensor imaging: comparison with electrophysiological studies of patients and healthy volunteers. Eur J Radiol 2012; 81: pp. 3378-3383.
15. Jellison B.J., Field A.S., Medow J., et. al.: Diffusion tensor imaging of cerebral white matter: a pictorial review of physics, fiber tract anatomy, and tumor imaging patterns. AJNR Am J Neuroradiol 2004; 25: pp. 356-369.
16. Andreisek G., White L.M., Kassner A., et. al.: Evaluation of diffusion tensor imaging and fiber tractography of the median nerve: preliminary results on intrasubject variability and precision of measurements. AJR Am J Roentgenol 2010; 194: pp. W65-W72.
17. Meek M.F., Stenekes M.W., Hoogduin H.M., et. al.: In vivo three-dimensional reconstruction of human median nerves by diffusion tensor imaging. Exp Neurol 2006; 198: pp. 479-482.
18. Skorpil M., Karlsson M., Nordell A.: Peripheral nerve diffusion tensor imaging. Magn Reson Imaging 2004; 22: pp. 743-745.
19. Hiltunen J., Suortti T., Arvela S., et. al.: Diffusion tensor imaging and tractography of distal peripheral nerves at 3 T. Clin Neurophysiol 2005; 116: pp. 2315-2323.
20. Kabakci N., Gurses B., Firat Z., et. al.: Diffusion tensor imaging and tractography of median nerve: normative diffusion values. AJR Am J Roentgenol 2007; 189: pp. 923-927.
21. Cauley K.A., Filippi C.G.: Diffusion-tensor imaging of small nerve bundles: cranial nerves, peripheral nerves, distal spinal cord, and lumbar nerve roots—clinical applications. AJR Am J Roentgenol 2013; 201: pp. W326-W335.
22. Stein D., Neufeld A., Pasternak O., et. al.: Diffusion tensor imaging of the median nerve in healthy and carpal tunnel syndrome subjects. J Magn Reson Imaging 2009; 29: pp. 657-662.
23. Guggenberger R., Markovic D., Eppenberger P., et. al.: Assessment of median nerve with MR neurography by using diffusion-tensor imaging: normative and pathologic diffusion values. Radiology 2012; 265: pp. 194-203.
24. Hiltunen J., Kirveskari E., Numminen J., et. al.: Pre- and post-operative diffusion tensor imaging of the median nerve in carpal tunnel syndrome. Eur Radiol 2012; 22: pp. 1310-1319.
25. Khalil C., Hancart C., Le Thuc V., et. al.: Diffusion tensor imaging and tractography of the median nerve in carpal tunnel syndrome: preliminary results. Eur Radiol 2008; 18: pp. 2283-2291.
26. Tasdelen N., Gurses B., Kilickesmez O., et. al.: Diffusion tensor imaging in carpal tunnel syndrome. Diagn Interv Radiol 2012; 18: pp. 60-66.
27. Stevens J.C.: AAEM minimonograph #26: the electrodiagnosis of carpal tunnel syndrome. American Association of Electrodiagnostic Medicine. Muscle Nerve 1997; 20: pp. 1477-1486.
28. Preston D.C., Shapiro B.E.: Electromyography and neuromuscular disorders.2nd ed2005.ElsevierPhiladelphia, PA
29. Shrout P.E., Fleiss J.L.: Intraclass correlations: uses in assessing rater reliability. Psychol Bull 1979; 86: pp. 420-428.
30. Landis J.R., Koch G.G.: The measurement of observer agreement for categorical data. Biometrics 1977; 33: pp. 159-174.
31. Koyuncuoglu H.R., Kutluhan S., Yesildag A., et. al.: The value of ultrasonographic measurement in carpal tunnel syndrome in patients with negative electrodiagnostic tests. Eur J Radiol 2005; 56: pp. 365-369.
32. Deniz F.E., Oksüz E., Sarikaya B., et. al.: Comparison of the diagnostic utility of electromyography, ultrasonography, computed tomography, and magnetic resonance imaging in idiopathic carpal tunnel syndrome determined by clinical findings. Neurosurgery 2012; 70: pp. 610-616.
33. Mesgarzadeh M., Schneck C.D., Bonakdarpour A., et. al.: Carpal tunnel: MR imaging. Part II. Carpal tunnel syndrome. Radiology 1989; 171: pp. 749-754.
34. Seyfert S., Boegner F., Hamm B., et. al.: The value of magnetic resonance imaging in carpal tunnel syndrome. J Neurol 1994; 242: pp. 41-46.