Rationale and Objectives
The purpose of this study was to evaluate the diagnostic value of Micropure Imaging (MI) in breast lesions differentiation by comparison with B-mode ultrasonography (B-US) and Doppler ultrasonography (DU).
Materials and Methods
A total of 135 consecutive patients (all females) with 135 suspicious lesions were examined and skin marked by MI before mammotome biopsies. All patients (age range, 20–86 years; mean age, 42.5 ± 15.6 years) were the first onset, not in the pregnancy or lactation and had no history of radiation or chemotherapy. The maximum diameter of lesions ranged from 0.2 to 9.6 cm (average 1.98 ± 1.3 cm). Their final diagnoses were obtained by histologic examination. The study protocol was approved by the hospital review board; each patient gave written informed consent.
Results
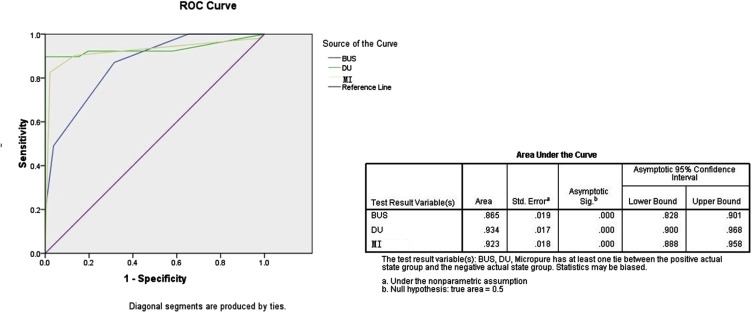
One hundred thirty-five breast lesions were classified into 90 nonmalignant and 45 malignant types. To 86 breast lesions with microcalcification, MI showed more microcalcification and coincided better with pathology results than B-US did ( P < .05). The specificity of MI was higher than that of B-US and DU; the sensitivity of DU was significantly higher than that of B-US and MI ( P < .001). By combining B-US, DU, and MI, the detection accuracy was 86.7%. Receiver-operator characteristic curves showed the area under the curve of B-US, DU, and MI was 0.865, 0.934, and 0.923( P = .000), respectively. Moreover, the interobserver agreements of MI were the highest, 0.922 (observer 1 vs. observer 2), 0.866 (observer 1 vs. observer 3), and 0.916 (observer 2 vs. observer 3).
Conclusions
MI as an adjunct ultrasound modality holds some promise in locating and differentiating breast lesions.
Instruction
Breast cancer is one of the most malignant tumors in women. Early detection often spells excellent prognosis. Numerous pathologic subtypes of breast cancer with suspicious microcalcification are proved either ductal carcinoma in situ (DCIS) or invasive cancer, which could be an early stage of breast cancer . So microcalcification plays an important role in diagnosing breast diseases and leads to extensive research in digital mammography . However, not all microcalcifications in mammography are malignant, and digital mammography does not eliminate fundamental limitations of detecting breast cancers without calcification, especially in dense parenchyma . Owing to the potential carcinogenicity, whether and when we intensify screening mammography programs on breast cancer are controversial. To seek a more objective and accessible method to check, suspicious microcalcification has become a new research trend. Many research showed Asian women had small dense breast tissue and fit for screening by ultrasonography rather than mammography . Microcalcification is also a common finding and symbolized by bright spot on ultrasonography. Because the speckle pattern and some tissue structures look like microcalcification, traditional ultrasound imaging is not good enough in clinical evaluation of suspicious microcalcification to date. Micropure Imaging (MI; USA Patent No.8,696,575) is a patent that Toshiba company invented and added on traditional gray scale ultrasonography. The apparatus removes a speckle pattern using information on a direction (depth direction) that is substantially orthogonal to a plurality of ultrasonic images included in three-dimensional image data. Micropure technique highlights microcalcification (less than 0.5 μm) represented by white spot through suppressing surrounding tissue with blue overlay image . Thus far, although a few articles proved MI had a possibility to increase detectability of the microcalcification , no articles evaluated the capabilities of the application in differentiating breast lesions with pathologic tests as gold standard. The purpose of our study was to assess the capabilities of MI in revealing the nature of breast lesions and targeting at suspicious microcalcification in biopsy. The results were compared with those of B-mode ultrasonography (B-US) and Doppler ultrasonography (DU). DU included color Doppler flow imaging (CDFI) and spectral Doppler (SD).
Materials and methods
Patients, Procedure, and Examination Technique
The study was conducted in a general hospital from November 2013 to March 2015, 135 consecutive patients (all females) with 135 nonpalpable suspicious breast lesions were examined by B-US and MI before mammotome biopsy and after surgery dissection. All patients (age range, 20–86 years; mean age, 42.5 ± 15.6 years) were the first onset, not in the pregnancy or lactation and had no history of radiation or chemotherapy. The maximum diameter of lesions ranged from 0.2 to 9.6 cm (average, 1.98 ± 1.3 cm). Their final diagnoses were obtained by means of histologic examination. The study protocol was approved by the hospital review board; each patient gave written informed consent.
Get Radiology Tree app to read full this article<
B-US, DU, and MI Evaluation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Pathologic Examination
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Pathology
Get Radiology Tree app to read full this article<
Table 1
Pathologic Types and Characteristics
Pathologic Types Number MI Microcalcification Display Rate, % (n/N) B-US Microcalcification Display Rate, % (n/N) Fibroadenoma 56 33.9 (19/56) 1.8 (1/56) Intraductal papilloma 10 90 (9/10) 80 (8/10) Usual ductal hyperplasia 6 83.3 (5/6) 50 (3/6) Atypical ductal hyperplasia 6 100 (6/6) 66.7 (4/6) Tubular adenoma 4 50 (2/4) 25 (1/4) Sclerosing adenosis 3 66.7 (2/3) 33.3 (1/3) Benign phyllodes tumor 1 100 (1/1) 0 Hamartoma 1 100 (1/1) 0 Breast tuberculosis 1 100 (1/1) 0 Breast abscess 1 100 (1/1) 0 Ductal adenoma 1 100 (1/1) 100 (1/1) NST 36 91.7 (33/36) 77.8 (28/36) DCIS 5 100 (5/5) 60 (3/5) Mucinous carcinoma 2 0 0 Medullary carcinoma 1 0 0 Invasive lobular carcinoma 1 0 0
B-US, B-mode ultrasonography; DCIS, ductal carcinoma in situ; MI, Micropure Imaging; NST, invasive carcinomas of no special type.
Get Radiology Tree app to read full this article<
B-US, DU, and MI
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Interobserver Variability
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Stomper C., Geradts J., Edge S., et. al.: Mammographic predictors of the presence and size of invasive carcinomas associated with malignant microcalcification lesions without a mass. AJR Am J Roentgenol 2003; 181: pp. 1674-1684.
2. Bundred S.M., Zhou J., Whiteside S., et. al.: Impact of full-field digital mammography on pre-operative diagnosis and surgical treatment of mammographic microcalcification. Breast Cancer Res Treat 2014; 143: pp. 359-366.
3. Li X., Weaver O., Desouki M.M., et. al.: Microcalcification is an important factor in the management of breast intraductal papillomas diagnosed on core biopsy. Am J Clin Pathol 2012; 138: pp. 789-795.
4. Colbert J.A., kaine E.M., Bigby J.A., et. al.: The age at which women begin mammographic screening. Cancer 2004; 101: pp. 1850-1859.
5. Teo S.Y., Chuwa E., Latha S., et. al.: Young breast cancer in a specialised breast unit in Singapore: clinical, radiological and pathological factors. Ann Acad Med Singapore 2014; 43: pp. 79-85.
6. Yamaguchi T., Hachiya H., Kato K., et. al.: Extraction of quantitative three-dimensional information from ultrasonic volumetric images of cirrhotic liver. Japan Journal of Applied Physics 2000; 39: pp. 3266-3269.
7. Wang T.C., Karayiannis N.B.: Detection of microcalcifications in digital mammograms using wavelets. IEEE Transactions on Medical Imaging 1998; 17: pp. 498-509.
8. Kamiyama N., Okamura Y., Kakee A., et. al.: Investigation of ultrasound image processing to improve perceptibility of microcalcifications. J Med Ultrason 2008; 35: pp. 97-105.
9. Machado P., Eisenbrey J.R., Cavanaugh B., et. al.: New image processing technique for evaluating breast microclacification: a comparative study. J Ultrasound Med 2012; 31: pp. 885-893.
10. Machado P., Eisenbrey J.R., Cavanaugh B., et. al.: Microcalcifications versus artifacts: initial evaluation of a new ultrasound image processing technique to identify breast microcalcifications in a screening population. Ultrasound Med Biol 2014; 40: pp. 2321-2324.
11. American College of Radiology: Breast imaging reporting and data system (BI-RADS).2003.American College of RadiologyReston, VA
12. Alder D.D., Carson P.L., Rubin J.M., et. al.: Doppler ultrasound color flow imaging in the study of breast cancer: preliminary findings. Ultrasound Med Biol 1999; 16: pp. 553-559.
13. Choi H.Y., Kim H.Y., Baek S.Y., et. al.: Significance of resistive index in color Doppler ultrasonogram: differentiation between benign and malignant breast masses. Clin Imaging 1999; 23: pp. 284-288.
14. Yang F., Li J.: WHO classification of tumors of the breast. Zhonghua Wai Ke Za Zhi 2014; 52: pp. 1-3.
15. Byrne D.W.: Publishing your medical research paper what they don’t teach in medical school.1st ed.2002.Lippincot Williams&wilkins and Peking Union Medical College PressBeijinpp. 88-96.
16. Vivona L., Cascio D., Fauci F., et. al.: Fuzzy technique for microcalcifications clustering in digital mammograms. BMC Medical Imaging 2014; 14: pp. 23.
17. Langen H.J., Koehler S., Bielmeier J., et. al.: Microradiography of microcalcifications in breast specimen: a new histological correlation procedure and the effect of improved resolution on diagnostic validity. Radiology Research and Practice 2012; 2012: pp. 1-13.
18. Tse G.M., Tan P.H., Cheung H.S., et. al.: Intermediate to highly suspicious calcification in breast lesions: a radio-pathologic correlation. Breast Cancer Res Treat 2008; 110: pp. 1-7.
19. Baker R., Rogers K.D., Shepherd N., et. al.: New relationships between breast microcalcifications and cancer. Br J Cancer 2010; 103: pp. 1034-1039.
20. Peters—Eng C., Med M., Leodoter S.: The use of colour-coded and spectral Doppler ultrasound in the differentiation of benign and malignant breast lesions. Br J Cancer 1995; 71: pp. 137-139.