Rationale and Objectives
The aim of this study was to explore the effects of blood supply on high-intensity focused ultrasound (HIFU) applied to rabbit hepatic VX2 tumors of different ages.
Materials and Methods
Eighteen rabbits with VX2 hepatic tumors were randomly divided into three groups according to the time of sacrifice after tumor implantation: 10, 15, or 20 days. Contrast-enhanced ultrasound was performed immediately before HIFU ablation. The same settings for HIFU dose parameters were used to ablate the central tumor area in each group, and the real-time temperature of the targeted site of the tumor was measured. After HIFU, the coagulation necrosis volumes of tumor tissue and the microvascular density of residual tumor tissue were determined.
Results
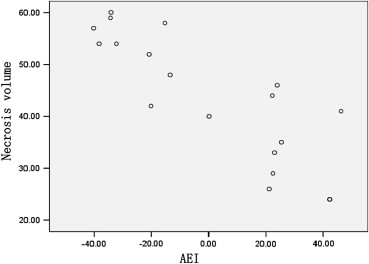
Histopathologic analysis showed that the extent of a tumor’s blood supply followed the order 10-day group > 15-day group > 20-day group ( P < .01). Contrast-enhanced ultrasound showed the same results. There was no statistically significant difference among the three groups in terms of temperature-increase parameters during HIFU treatment ( P > .05). However, there were statistically significant differences between the groups in terms of temperature-decrease parameters during HIFU treatment and in terms of necrosis volumes after HIFU treatment ( P < .05). Necrosis volume was inversely related to absolute enhanced intensity ( r = −0.823, P < .001).
Conclusions
The extent of a tumor’s blood supply had a significant effect on the temperature-decrease phase but not on the temperature-increase phase during HIFU treatment. The longer the temperature-decrease phase, the more slowly heat dissipated after HIFU, resulting in larger coagulation necrosis volumes.
High-intensity focused ultrasound (HIFU) is a rapidly developing noninvasive technology for thermal treatment. HIFU ablation of tumors is based on the absorption of ultrasound energy in a focal zone located at a distance from the therapeutic ultrasound transducer. In recent years, the technique has been used clinically to treat a variety of solid tumors, with promising prospects . Nevertheless, many studies of HIFU ablation have shown that tissue heating and lesion coagulation are affected by many factors, such as overlying ribs, abdominal gas, and the extent of blood perfusion within tumors . Tumor blood supply has been identified as one of the most important predictive factors. Early studies reported that HIFU ablation was less effective in the presence of abundant tumor blood perfusion, which was able to carry part of the focused ultrasound energy away. This was especially true in malignant tumors with high neovascularity and vascular invasion . Thus, the extent of blood perfusion within the focused region can limit ablation size and alter ablation shape by lowering the maximal temperature induced with HIFU .
The splanchnic circulation in rabbits is similar to that in humans . Rabbit hepatic VX2 tumors are highly vascularized by the hepatic artery because of the induction of neovascularity in the host animals, thereby resembling human hepatic carcinoma . This tumor model has thus been widely used for research on interventional therapy and imaging evaluation. Lorelius and Stridbeck found age-related changes in VX2 tumor vascularity of the rabbit hind leg. We propose that rabbit hepatic VX2 tumors of different ages may also possess age-related changes in blood supply.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Experimental Setup and Animal Handling
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
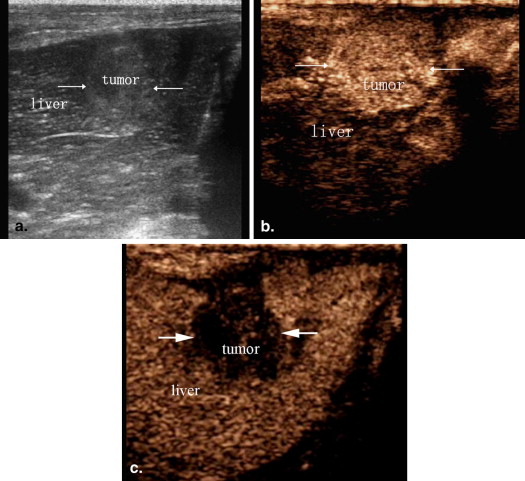
CEUS Examinations
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
HIFU Treatments
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
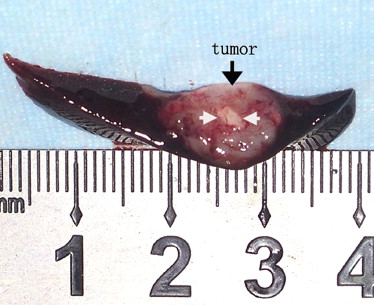
Histology
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
CEUS Evaluation of Blood Supply to VX2 Liver Tumors
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Comparison of AEI in VX2 Rabbit Tumor Tissues
Group_n_ AEI (Level) 10 day 6 30.04 ± 11.11 15 day 6 0.60 ± 20.38 ∗ 20 day 6 −32.36 ± 8.88 ∗ †
AEI, absolute enhanced intensity.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
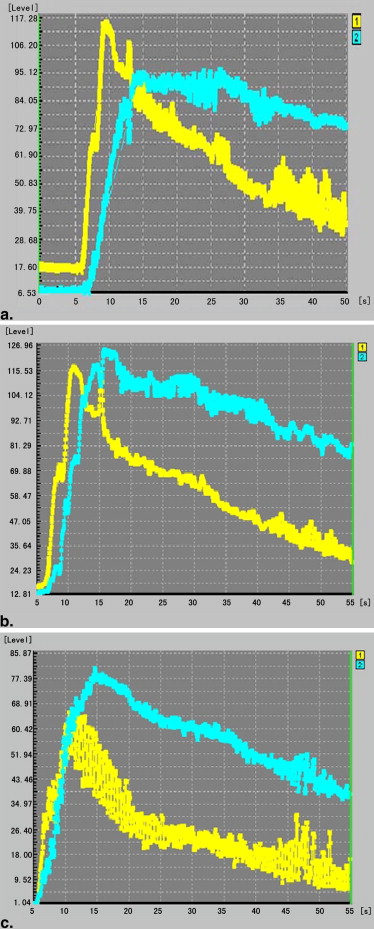
Temperature Changes in Tumors Targeted Site During HIFU Treatment
Get Radiology Tree app to read full this article<
Table 2
TTC Parameters at Different Times During VX2 Rabbit Tumors Undergoing HIFU Irradiation
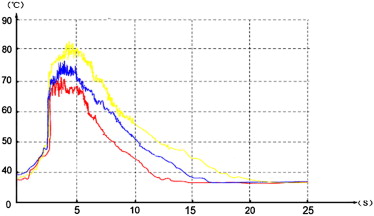
Group_n__T_ max (°C)t 1 (s)t 2 (s)k 1 (°C/s)k 2 (°C/s) 10 day 6 79.7 ± 2.60 4.3 ± 0.69 12.5 ± 0.47 9.7 ± 2.10 4.9 ± 0.36 15 day 6 79.8 ± 3.92 4.3 ± 0.50 14.1 ± 0.66 ∗ 9.5 ± 1.12 4.2 ± 0.70 ∗ 20 day 6 83.2 ± 5.58 4.5 ± 0.55 17.5 ± 1.21 ∗ † 9.9 ± 1.75 3.4 ± 0.36 ∗ †
HIFU, high-intensity focused ultrasound; TTC, time-temperature curve.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Pathologic Analysis of Rabbit Hepatic VX2 Tumors after HIFU Treatment
Get Radiology Tree app to read full this article<
Table 3
Comparison of Volume of Coagulation Necrosis in VX2 Rabbit Tumor Tissues
Group_n_ Volume of Coagulation Necrosis (mm 3 ) 10 day 6 31.33 ± 6.28 15 day 6 45.00 ± 4.39 ∗ 20 day 6 57.00 ± 2.55 ∗ †
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
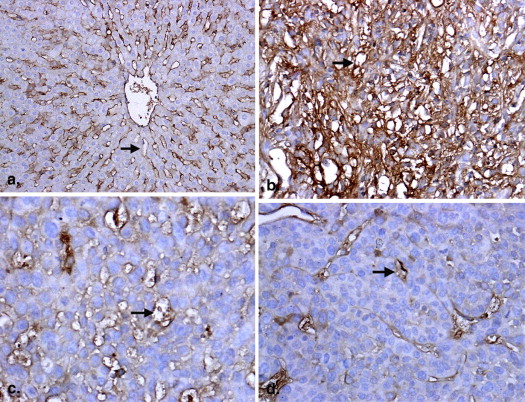
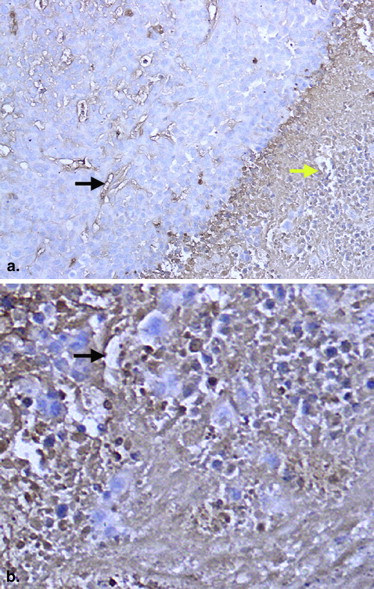
Table 4
Comparison of MVD in Rabbit Hepatic VX2 Tumors and Normal Tissues
MVD 10-Day Group
( n = 6) 15-Day Group
( n = 6) 20-Day Group
( n = 6) Tumor 37.71 ± 6.13 ∗ 25.63 ± 4.33 † 15.31 ± 3.17 Normal liver tissue 18.55 ± 3.33 19.63 ± 2.62 18.21 ± 2.07
MVD, microvessel density.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
References
1. Wu F., Wang Z.B., Chen W.Z., et. al.: Extracorporeal focused ultrasound surgery for treatment of human solid carcinomas: early Chinese clinical experience. Ultrasound Med Biol 2004; 30: pp. 245-260.
2. Wu F., Chen W.Z., Bai J., et. al.: Pathological changes in human malignant carcinoma treated with high-intensity focused ultrasound. Ultrasound Med Biol 2001; 27: pp. 1099-1106.
3. Li C., Wu P., Zhang L., et. al.: Osteosarcoma: limb salvaging treatment by ultrasonographically guided high-intensity focused ultrasound. Cancer Biol Ther 2009; 8: pp. 1102-1108.
4. Li C., Zhang W., Zhang R., et. al.: Therapeutic effects and prognostic factors in high-intensity focused ultrasound combined with chemoembolisation for larger hepatocellular carcinoma. Eur J Cancer 2010; 46: pp. 2513-2521.
5. Li J.J., Gu M.F., Luo G.Y., et. al.: Complications of high intensity focused ultrasound for patients with hepatocellular carcinoma. Technol Cancer Res Treat 2009; 8: pp. 217-224.
6. Thuroff S., Chaussy C.: High-intensity focused ultrasound: complications and adverse events. Mol Urol 2000; 4: pp. 183-187.
7. Wu F., Wang Z.B., Chen W.Z., et. al.: Extracorporeal high intensity focused ultrasound ablation in the treatment of patients with large hepatocellular carcinoma. Ann Surg Oncol 2004; 11: pp. 1061-1069.
8. Luo W., Zhou X., He G., et. al.: Ablation of high intensity focused ultrasound combined with SonoVue on rabbit VX2 liver tumors: assessment with conventional gray-scale US, conventional color/power Doppler US, contrast-enhanced color Doppler US, and contrast-enhanced pulse-inversion harmonic US. Ann Surg Oncol 2008; 15: pp. 2943-2953.
9. Li J.J., Xu G.L., Gu M.F., et. al.: Complications of high intensity focused ultrasound in patients with recurrent and metastatic abdominal tumors. World J Gastroenterol 2007; 13: pp. 2747-2751.
10. Chen L., ter Haar G., Hill C.R., et. al.: Effect of blood perfusion on the ablation of liver parenchyma with high-intensity focused ultrasound. Phys Med Biol 1993; 38: pp. 1661-1673.
11. Bonte F.J., Curry G.C., Parkey R.W.: Experimental studies of the splanchnic circulation of the rabbit after ligation of the superior mesenteric artery. I. Angiographic and physiologic studies. Am J Roentgenol Radium Ther Nucl Med 1969; 106: pp. 691-699.
12. Kuszyk B.S., Boitnott J.K., Choti M.A., et. al.: Local tumor recurrence following hepatic cryoablation: radiologic-histopathologic correlation in a rabbit model. Radiology 2000; 217: pp. 477-486.
13. Lorelius L.E., Stridbeck H.: Age related changes in tumor vascularity. Angiography and vascular casts of the VX2 tumor in the rabbit hind leg. Acta Radiol Diagn (Stockh) 1984; 25: pp. 529-533.
14. Schwarz K.Q., Chen X., Steinmetz S., et. al.: Harmonic imaging with Levovist. J Am Soc Echocardiogr 1997; 10: pp. 1-10.
15. Xu H.X., Lu M.D., Xie X.H., et. al.: Treatment response evaluation with three-dimensional contrast-enhanced ultrasound for liver cancer after local therapies. Eur J Radiol 2010; 76: pp. 81-88.
16. Li Q., Du J., Yu M., et. al.: Transmission electron microscopy of VX2 liver tumors after high-intensity focused ultrasound ablation enhanced with SonoVue. Adv Ther 2009; 26: pp. 117-125.
17. Luo W., Zhou X., Zheng X., et. al.: Role of sonography for implantation and sequential evaluation of a VX2 rabbit liver tumor model. J Ultrasound Med 2010; 29: pp. 51-60.
18. Divkovic G.W., Liebler M., Braun K., et. al.: Thermal properties and changes of acoustic parameters in an egg white phantom during heating and coagulation by high intensity focused ultrasound. Ultrasound Med Biol 2007; 33: pp. 981-986.
19. Lechat P., Levassort C.: Modification of rabbit body temperature induced by the simultaneous injection of gonococcus vaccine and various phenothiazine neuroleptics. Pharmacology 1969; 2: pp. 100-112.
20. ter Haar G., Rivens I., Chen L., et. al.: High intensity focused ultrasound for the treatment of rat tumours. Phys Med Biol 1991; 36: pp. 1495-1501.
21. Weidner N.: Current pathologic methods for measuring intratumoral microvessel density within breast carcinoma and other solid tumors. Breast Cancer Res Treat 1995; 36: pp. 169-180.
22. Gandhi D., Hoeffner E.G., Carlos R.C., et. al.: Computed tomography perfusion of squamous cell carcinoma of the upper aerodigestive tract. Initial results. J Comput Assist Tomogr 2003; 27: pp. 687-693.
23. Schacherer D., Girlich C., Wiest R., et. al.: Semiquantitative characterization of hepatocellular carcinoma (HCC)—perfusion with contrast-enhanced ultrasound and perfusion analysis. Clin Hemorheol Microcirc 2010; 44: pp. 97-105.
24. Giorgio A., Ferraioli G., Tarantino L., et. al.: Contrast-enhanced sonographic appearance of hepatocellular carcinoma in patients with cirrhosis: comparison with contrast-enhanced helical CT appearance. AJR Am J Roentgenol 2004; 183: pp. 1319-1326.
25. Boxer G.M., Tsiompanou E., Levine T., et. al.: Immunohistochemical expression of vascular endothelial growth factor and microvessel counting as prognostic indicators in node-negative colorectal cancer. Tumour Biol 2005; 26: pp. 1-8.