Rationale and Objectives
Evaluation of nodal involvement in early-stage breast cancers (T1 or T2) changed following the Z11 trial; however, not all patients meet the Z11 inclusion criteria. Hence, the relevance of ultrasound imaging of the axilla and fine-needle aspiration biopsy (FNA) in early-stage breast cancers was investigated.
Materials and Methods
In this single-center, retrospective study, 758 subjects had pathology-verified breast cancer diagnosis over a 3-year period, of which 128 subjects with T1 or T2 breast tumors had abnormal axillary lymph nodes on ultrasound, had FNA, and proceeded to axillary surgery. Ultrasound images were reviewed and analyzed using multivariable logistic regression to identify the features predictive of positive FNA. Accuracy of FNA was quantified as the area under the receiver operating characteristic curve with axillary surgery as reference standard.
Results
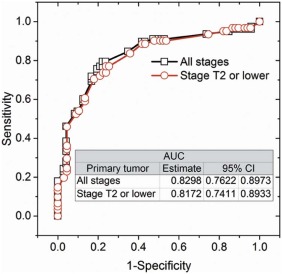
Of 128 subjects, 61 were positive on FNA and 65 were positive on axillary surgery. Sensitivity, specificity, positive predictive value, and negative predictive value of FNA were 52 of 65 (80%), 54 of 63 (85.7%), 52 of 61(85.2%), and 54 of 67 (80.5%), respectively. After adjusting for neoadjuvant chemotherapy between FNA and surgery, a positive FNA was associated with higher likelihood for positive axillary surgery (odds ratio: 22.7; 95% confidence interval [CI]: 7.2–71.3, P < .0001), and the accuracy of FNA was 0.801 (95% CI: 0.727–0.876). Among ultrasound imaging features, cortical thickness and abnormal hilum were predictive ( P < .017) of positive FNA with accuracy of 0.817 (95% CI: 0.741–0.893).
Conclusions
Ultrasound imaging and FNA can play an important role in the management of early breast cancers even in the post-Z11 era. Higher weightage can be accorded to cortical thickness and hilum during ultrasound evaluation.
Introduction
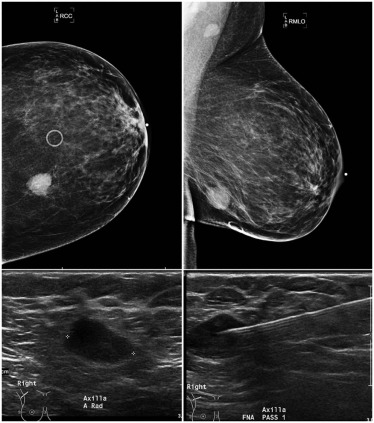
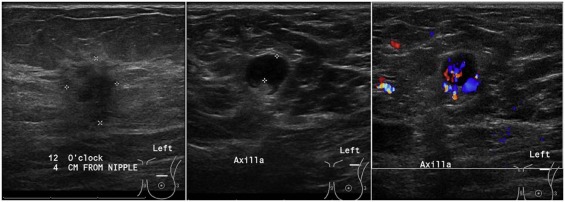
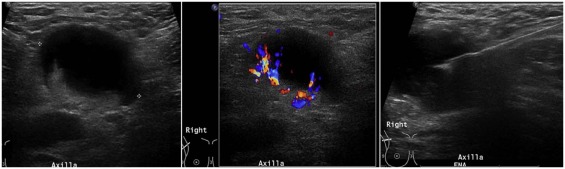
Management of breast cancer has been evolving over the last few decades. Less invasive procedures with lower morbidity have been shown to be equally effective with no adverse impact on outcomes . Metastatic involvement of the axilla is an important prognostic indicator on the outcome in patients with breast cancer. In stage I-II breast cancers, tumor grade, size, receptor status, and lymphovascular invasion of the tumor increase the odds for involvement of the axilla . The imaging modality of choice for evaluation of the axilla is ultrasound (US). When suspicious lymph nodes are identified on US, fine-needle aspiration biopsy (FNA) or core-needle biopsy typically follows, both to establish a histological diagnosis and to guide further management. A number of studies have outlined the benefits of US and FNA in the evaluation of axillary lymph nodes .
The publication of the American College of Surgeons Oncology Group Z11 trial, however, resulted in a paradigm shift in the way early T1 and T2 breast cancers are managed at most institutions . The study showed no benefit for axillary lymph node dissection (ALND) over surgical sentinel node biopsy (SNB) for patients with low burden of axillary disease who underwent breast conserving therapy and received whole breast irradiation. It established SNB as the standard of care . As a consequence, the value of imaging and interventional procedures of axillary lymph nodes has been questioned . To remain relevant and continue to contribute meaningfully to patient care, it is important for breast imagers to adapt to these changes.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Methods and Materials
Get Radiology Tree app to read full this article<
Human Subjects
Get Radiology Tree app to read full this article<
Table 1
Distribution of Tumor Stage, Grade, and the Axillary Surgery Following FNA in Subjects with Stage T2 or Lower Cancers
n__Tumor stage T1a 7 T1b 14 T1c 48 T2 59Tumor grade I 17 II 58 III 53Axillary surgery SNB 54 ALND 65 SNB followed by ALND 9
ALND, axillary lymph node dissection; FNA, fine-needle aspiration biopsy; SNB, sentinel node biopsy.
Get Radiology Tree app to read full this article<
Ultrasound Imaging and FNA
Get Radiology Tree app to read full this article<
Retrospective Review
Get Radiology Tree app to read full this article<
Reference Standard
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Ultrasound Imaging Features
Get Radiology Tree app to read full this article<
Table 2
Odds Ratio and 95% Confidence Intervals of Ultrasound Imaging Features Predictive of a Positive Result From Ultrasound-guided Fine-needle Aspiration Biopsy (FNA)
Ultrasound Imaging Feature Odds Ratio Estimate 95% Confidence Intervals_P_ Value_All breast cancer stages_ ( n = 150) Cortical thickness in mm 1.268 1.075 1.496 .0048 Hilum (abnormal vs. normal) 5.271 2.217 12.530 .0002Stage T2 or lower ( n = 128) Cortical thickness in mm 1.258 1.042 1.520 .0172 Hilum (abnormal vs. normal) 4.509 1.735 11.718 .0020
Other than cortical thickness in mm and hilum status categorized as normal or abnormal, none of the other ultrasound imaging features and measurements were significantly ( P > .3769) associated with a positive FNA.
Get Radiology Tree app to read full this article<
Axillary Surgery
Get Radiology Tree app to read full this article<
Table 3
Odds Ratio and 95% Confidence Intervals of the Predictor(s) Associated with Axillary Lymph Node Metastasis From Pathology Analysis of Sentinel Node Biopsy (SNB) or Axillary Lymph Node Dissection (ALND)
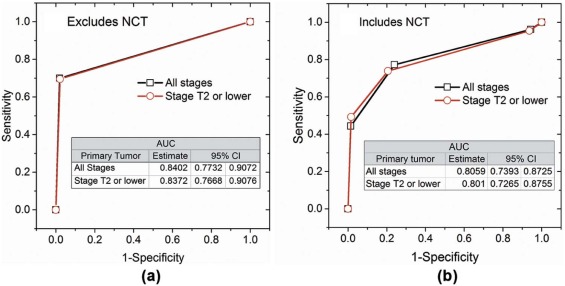
Predictor Odds Ratio Estimate 95% Confidence Intervals_P_ Value Cohort: Excludes Subjects Who Underwent NCT (Univariate Analysis)All breast cancer stages (n = 101) FNA (positive vs. negative) 116.646 14.724 924.084 <.0001Stage T2 or lower (n = 93) FNA (positive vs. negative) 105.133 13.158 840.046 <.0001
Cohort: Includes Subjects Who Underwent NCT (Multivariable Analysis)All breast cancer stages ( n = 150) FNA (positive vs. negative) 25.533 8.209 79.418 <.0001 NCT (yes vs. no) 0.224 0.07 0.722 .0122Stage T2 or lower ( n = 128) FNA (positive vs. negative) 22.672 7.205 71.342 <.0001 NCT (yes vs. no) 0.226 0.066 0.768 .0172
Some of the subjects underwent interim neoadjuvant chemotherapy (NCT) between the time of ultrasound-guided fine-needle aspiration biopsy (FNA) and the surgical procedure, and was included in the model. Hence, the results are presented for the cohort of subjects who did not undergo NCT, and for the entire study sample that includes subjects who underwent NCT.
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusion
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Black D.M., Mittendorf E.A.: Landmark trials affecting the surgical management of invasive breast cancer. Surg Clin North Am 2013; 93: pp. 501-518.
2. Rivadeneira D.E., Simmons R.M., Christos P.J., et. al.: Predictive factors associated with axillary lymph node metastases in T1a and T1b breast carcinomas: analysis in more than 900 patients. J Am Coll Surg 2000; 191: pp. 1-6. discussion-8
3. Hieken T.J., Trull B.C., Boughey J.C., et. al.: Preoperative axillary imaging with percutaneous lymph node biopsy is valuable in the contemporary management of patients with breast cancer. Surgery 2013; 154: pp. 831-838. discussion 8–40
4. Cools-Lartigue J., Sinclair A., Trabulsi N., et. al.: Preoperative axillary ultrasound and fine-needle aspiration biopsy in the diagnosis of axillary metastases in patients with breast cancer: predictors of accuracy and future implications. Ann Surg Oncol 2013; 20: pp. 819-827.
5. Koelliker S.L., Chung M.A., Mainiero M.B., et. al.: Axillary lymph nodes: US-guided fine-needle aspiration for initial staging of breast cancer—correlation with primary tumor size. Radiology 2008; 246: pp. 81-89.
6. Joh J.E., Han G., Kiluk J.V., et. al.: Indications for axillary ultrasound use in breast cancer patients. Clin Breast Cancer 2012; 12: pp. 433-437.
7. Leenders M.W., Broeders M., Croese C., et. al.: Ultrasound and fine needle aspiration cytology of axillary lymph nodes in breast cancer. To do or not to do?. Breast 2012; 21: pp. 578-583.
8. Park S.H., Kim M.J., Park B.W., et. al.: Impact of preoperative ultrasonography and fine-needle aspiration of axillary lymph nodes on surgical management of primary breast cancer. Ann Surg Oncol 2011; 18: pp. 738-744.
9. Jung J., Park H., Park J., et. al.: Accuracy of preoperative ultrasound and ultrasound-guided fine needle aspiration cytology for axillary staging in breast cancer. ANZ J Surg 2010; 80: pp. 271-275.
10. Sauer T., Karesen R.: The value of preoperative ultrasound guided fine-needle aspiration cytology of radiologically suspicious axillary lymph nodes in breast cancer. Cytojournal 2014; 11: pp. 26.
11. MacNeill M., Arnott I., Thomas J.: Fine needle aspiration cytology is a valuable adjunct to axillary ultrasound in the preoperative staging of breast cancer. J Clin Pathol 2011; 64: pp. 42-46.
12. Mainiero M.B., Cinelli C.M., Koelliker S.L., et. al.: Axillary ultrasound and fine-needle aspiration in the preoperative evaluation of the breast cancer patient: an algorithm based on tumor size and lymph node appearance. AJR Am J Roentgenol 2010; 195: pp. 1261-1267.
13. Chang M.C., Crystal P., Colgan T.J.: The evolving role of axillary lymph node fine-needle aspiration in the management of carcinoma of the breast. Cancer Cytopathol 2011; 119: pp. 328-334.
14. Rao R., Lilley L., Andrews V., et. al.: Axillary staging by percutaneous biopsy: sensitivity of fine-needle aspiration versus core needle biopsy. Ann Surg Oncol 2009; 16: pp. 1170-1175.
15. Krishnamurthy S., Sneige N., Bedi D.G., et. al.: Role of ultrasound-guided fine-needle aspiration of indeterminate and suspicious axillary lymph nodes in the initial staging of breast carcinoma. Cancer 2002; 95: pp. 982-988.
16. Baruah B.P., Goyal A., Young P., et. al.: Axillary node staging by ultrasonography and fine-needle aspiration cytology in patients with breast cancer. Br J Surg 2010; 97: pp. 680-683.
17. Giuliano A.E., Hunt K.K., Ballman K.V., et. al.: Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 2011; 305: pp. 569-575.
18. Giuliano A.E., McCall L., Beitsch P., et. al.: Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: the American College of Surgeons Oncology Group Z11 randomized trial. Ann Surg 2010; 252: pp. 426-432. discussion 32-3
19. Cody H.S., Houssami N.: Axillary management in breast cancer: what’s new for 2012?. Breast 2012; 21: pp. 411-415.
20. Humphrey K.L., Saksena M.A., Freer P.E., et. al.: To do or not to do: axillary nodal evaluation after ACOSOG Z11 Trial. Radiographics 2014; 34: pp. 1807-1816.
21. Ainsworth R.K., Kollias J., Le Blanc A., et. al.: The clinical impact of the American College of Surgeons Oncology Group Z-0011 trial—results from the BreastSurgANZ National Breast Cancer Audit. Breast 2013; 22: pp. 733-735.
22. Ecanow J.S., Abe H., Newstead G.M., et. al.: Axillary staging of breast cancer: what the radiologist should know. Radiographics 2013; 33: pp. 1589-1612.
23. Abe H., Schmidt R.A., Sennett C.A., et. al.: US-guided core needle biopsy of axillary lymph nodes in patients with breast cancer: why and how to do it. Radiographics 2007; 27: pp. S91-S99.
24. Lee B., Lim A.K., Krell J., et. al.: The efficacy of axillary ultrasound in the detection of nodal metastasis in breast cancer. AJR Am J Roentgenol 2013; 200: pp. W314-W320.
25. Zhu Y., Zhou W., Zhou J.Q., et. al.: Axillary staging of early-stage invasive breast cancer by ultrasound-guided fine-needle aspiration cytology: which ultrasound criteria for classifying abnormal lymph nodes should be adopted in the post-ACOSOG Z11 trial era?. J Ultrasound Med 2016; 35: pp. 885-893.
26. Saffar B., Bennett M., Metcalf C., et. al.: Retrospective preoperative assessment of the axillary lymph nodes in patients with breast cancer and literature review. Clin Radiol 2015; 70: pp. 954-959.
27. Amonkar S.J., Oates E., McLean L., et. al.: Pre-operative staging of the axilla in primary breast cancer. By redefining the abnormal appearing node can we reduce investigations without affecting overall treatment?. Breast 2013; 22: pp. 1114-1118.
28. Rautiainen S., Masarwah A., Sudah M., et. al.: Axillary lymph node biopsy in newly diagnosed invasive breast cancer: comparative accuracy of fine-needle aspiration biopsy versus core-needle biopsy. Radiology 2013; 269: pp. 54-60.
29. Fleissig A., Fallowfield L.J., Langridge C.I., et. al.: Post-operative arm morbidity and quality of life. Results of the ALMANAC randomised trial comparing sentinel node biopsy with standard axillary treatment in the management of patients with early breast cancer. Breast Cancer Res Treat 2006; 95: pp. 279-293.
30. Purushotham A.D., Upponi S., Klevesath M.B., et. al.: Morbidity after sentinel lymph node biopsy in primary breast cancer: results from a randomized controlled trial. J Clin Oncol 2005; 23: pp. 4312-4321.
31. Hindie E., Groheux D., Brenot-Rossi I., et. al.: The sentinel node procedure in breast cancer: nuclear medicine as the starting point. J Nucl Med 2011; 52: pp. 405-414.
32. Boughey J.C., Moriarty J.P., Degnim A.C., et. al.: Cost modeling of preoperative axillary ultrasound and fine-needle aspiration to guide surgery for invasive breast cancer. Ann Surg Oncol 2010; 17: pp. 953-958.
33. Yip C.H., Taib N.A., Tan G.H., et. al.: Predictors of axillary lymph node metastases in breast cancer: is there a role for minimal axillary surgery?. World J Surg 2009; 33: pp. 54-57.
34. Wada N., Imoto S., Yamauchi C., et. al.: Predictors of tumour involvement in remaining axillary lymph nodes of breast cancer patients with positive sentinel lymph node. Eur J Surg Oncol 2006; 32: pp. 29-33.
35. Boland M.R., Prichard R.S., Daskalova I., et. al.: Axillary nodal burden in primary breast cancer patients with positive pre-operative ultrasound guided fine needle aspiration cytology: management in the era of ACOSOG Z011. Eur J Surg Oncol 2015; 41: pp. 559-565.