Rationale and Objectives
Studies reporting the safety of magnetic resonance imaging (MRI) in patients with a cardiac implantable electronic device (CIED) have mostly excluded examinations with the device in the magnet isocenter. The purpose of this study was to describe the safety of cardiac and thoracic spine MRI in patients with a CIED.
Materials and Methods
The medical records of patients with a CIED who underwent a cardiac or thoracic spine MRI between January 2011 and December 2014 were reviewed. Devices were interrogated before and after imaging with reprogramming to asynchronous pacing in pacemaker-dependent patients. The clinical interpretability of the MRI and peak and average specific absorption rates (SARs, W/kg) achieved were determined.
Results
Fifty-eight patients underwent 51 cardiac and 11 thoracic spine MRI exams. Twenty-nine patients had a pacemaker and 29 had an implantable cardioverter defibrillator. Seventeen percent ( n = 10) were pacemaker dependent. Fifty-one patients (89%) had non-MRI-conditional devices. There were no clinically significant changes in atrial and ventricular sensing, impedance, and threshold measurements. There were no episodes of device mode changes, arrhythmias, therapies delivered, electrical reset, or battery depletion. One study was prematurely discontinued due to a patient complaint of chest pain of which the etiology was not determined. Across all examinations, the average peak SAR was 2.0 ± 0.85 W/kg with an average SAR of 0.35 ± 0.37 W/kg. Artifact significantly limiting the clinical interpretation of the study was present in 33% of cardiac MRI studies.
Conclusions
When a comprehensive CIED magnetic resonance safety protocol is followed, the risk of performing 1.5-T magnetic resonance studies with the device in the magnet isocenter, including in patients who are pacemaker dependent, is low.
Introduction
Magnetic resonance imaging (MRI) has been considered to be a contraindication in patients with a cardiac implantable electronic device (CIED) due to potential life-threatening interactions with incompatible CIED systems . In recent years, an increasing number of MRI-conditional pacemakers have become available . These systems contain specially developed components that are tested and approved for usage in an MRI environment. While the routine implantation of MRI-conditional devices and leads may become standard practice, there is currently a large population of patients with previously implanted cardiac devices that require MRI scanning. In addition, until recently, there was no Food and Drug Administration approved MRI-conditional device approved to be scanned in the isocenter of the magnet precluding cardiac magnetic resonance (MR) and thoracic spine MR exams .
The largest, prospective multicenter data come from the recently completed MagnaSafe Registry trial, which included 1500 patients undergoing MRI with a cardiac implantable device . However, patients with a CIED undergoing cardiac and thoracic spine MRI studies were explicitly excluded from the trial due to safety concerns with the device in the magnet isocenter during scanning. In addition, exams with the CIED in the magnetic isocenter are poorly represented in the literature and the energy deposition of the specific MR protocols is rarely reported. The purpose of the present study was to describe the safety of cardiac and thoracic spine MRI in patients with a CIED, including information on the energy deposition during scanning (specific absorption rate [SAR], W/kg, and whole-body radio frequency [RF], energy J/kg) achieved during MR scanning, using a multidisciplinary MRI safety protocol.
Methods
Study Design and Data Collection
Get Radiology Tree app to read full this article<
MRI SARs
Get Radiology Tree app to read full this article<
MRI RF Energy Deposition
Get Radiology Tree app to read full this article<
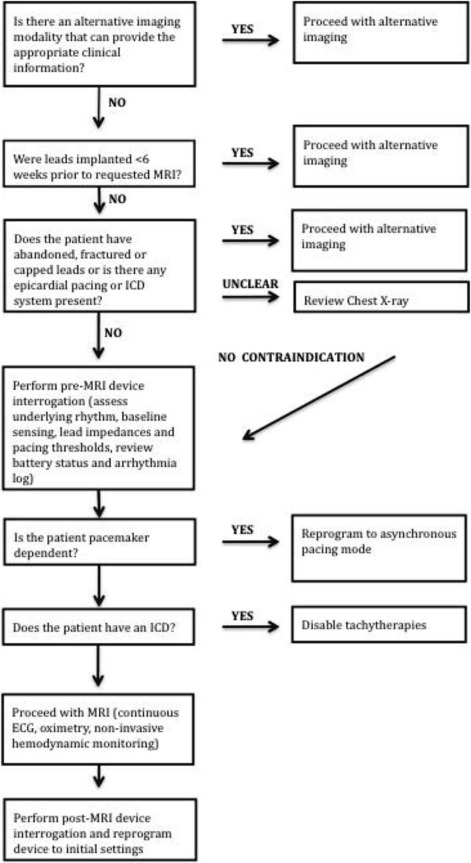
Comprehensive Safety Protocol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
MRI Protocol
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Patient and Device Characteristics ( Table 1 )
Get Radiology Tree app to read full this article<
Table 1
Patient and Device Characteristics
Variable Age, years (SD) 58.5(14.3) Male gender, n (%) 40(65) Pacemaker, n (%) 29(50) ICD, n (%) 29(50) Dual-chamber device, n (%) 38(62) Biventricular device, n (%) 7(11) MRI-conditional device, n (%) 7(11) Pacemaker dependent, n (%) 10(17)
ICD, implantable cardioverter defibrillator; MRI, magnetic resonance imaging; SD, standard deviation.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Mean Device Measurements
Baseline Lead Position Sensing, mV (SD) Impedance, ohm (SD) Threshold, V (SD) Right atrium 2.7(1.4) 489.3(96.0) 0.88(0.49) Right ventricle 9.1(4.2) 486.9(98.5) 0.96(0.58) Left ventricle 11.0(5.3) 618.6(221.5) 1.2(0.5)
Mean Difference Post-MRI Lead Position Sensing, mV (SD) Impedance, ohm (SD) Threshold, V (SD) Right atrium +0.03(0.22) −1.4(17.7) +0(0.1) Right ventricle +0.09(0.85) −4.1(32.7) +0(0.13) Left ventricle −0.33(0.65) −6.4(11.8) +0.02(0.04)
MRI, magnetic resonance imaging; SD, standard deviation.
Get Radiology Tree app to read full this article<
Complications
Get Radiology Tree app to read full this article<
SAR Data
Get Radiology Tree app to read full this article<
Table 3
Exam Level-Specific Adsorption Ratio Data
Exam Subjects, n Peak SAR (W/kg) SAR (W/kg) Max Average Median SD Average Median SD Brain MRI 3 0.08 0.07 0.07 0.00 0.03 0.03 0.01 Cardiac MRI 51 3.65 2.03 1.97 0.91 0.25 0.19 0.30 Cervical spine MRI 7 1.49 0.94 0.92 0.35 0.50 0.45 0.16 Thoracic spine MRI 10 2.18 1.68 1.82 0.39 0.94 0.88 0.23 Lumbar spine MRI 7 2.21 1.81 2.00 0.46 1.24 1.26 0.36 Pelvis MRI 1 2.00 N/A N/A N/A 0.93 0.66 0.74
MRI, magnetic resonance imaging; N/A, not applicable; SAR, specific absorption rate; SD, standard deviation.
Get Radiology Tree app to read full this article<
Whole-Body RF Energy
Get Radiology Tree app to read full this article<
Patients with Multiple MRI Exams
Get Radiology Tree app to read full this article<
Clinical Interpretability
Get Radiology Tree app to read full this article<
Discussion
Main Findings
Get Radiology Tree app to read full this article<
Comparison to Prior Studies
Get Radiology Tree app to read full this article<
Risks of MRI in the Presence of a CIED
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Limitations
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Langman D.A., Goldberg I.B., Finn J.P., et. al.: Pacemaker lead tip heating in abandoned and pacemaker-attached leads at 1.5 Tesla MRI. J Magn Reson Imaging 2011; 33: pp. 426-431.
2. Kalin R., Stanton M.S.: Current clinical issues for MRI scanning of pacemaker and defibrillator patients. Pacing Clin Electrophysiol 2005; 28: pp. 326-328.
3. Pavlicek W., Geisinger M., Castle L., et. al.: The effects of nuclear magnetic resonance on patients with cardiac pacemakers. Radiology 1983; 147: pp. 149-153.
4. Wilkoff B.L., Bello D., Taborsky M., et. al.: Magnetic resonance imaging in patients with a pacemaker system designed for the magnetic resonance environment. Heart Rhythm 2011; 8: pp. 65-73.
5. Gold M.R., Sommer T., Schwitter J., et. al.: Full-body MRI in patients with an implantable cardioverter-defibrillator: primary results of a randomized study. J Am Coll Cardiol 2015; 65: pp. 2581-2588.
6. Russo R.J.: Determining the risks of clinically indicated nonthoracic magnetic resonance imaging at 1.5 T for patients with pacemakers and implantable cardioverter-defibrillators: rationale and design of the MagnaSafe Registry. Am Heart J 2013; 165: pp. 266-272.
7. Nazarian S., Halperin H.R.: How to perform magnetic resonance imaging on patients with implantable cardiac arrhythmia devices. Heart Rhythm 2009; 6: pp. 138-143.
8. Nazarian S., Beinart R., Halperin H.R.: Magnetic resonance imaging and implantable devices. Circ Arrhythm Electrophysiol 2013; 6: pp. 419-428.
9. Nazarian S., Hansford R., Roguin A., et. al.: A prospective evaluation of a protocol of a protocol for magnetic resonance imaging of patients with implanted cardiac devices. Ann Intern Med 2011; 155: pp. 415-424.
10. Higgins J.V., Sheldon S.H., Watson R.E., et. al.: “Power-on resets” in cardiac implantable electronic devices during magnetic resonance imaging. Heart Rhythm 2015; 12: pp. 540-544.
11. Russo R.J.: Determining the risks of magnetic resonance imaging at 1.5 Tesla for patients with non-MRI conditional pacemakers and implantable cardioverter defibrillators: Final results of the MagnaSafe Registry. American Heart Association Scientific Sessions. Chicago, IL. Oral Presentation2014.
12. Fetter J., Aram G., Holmes D.R., et. al.: The effects of nuclear magnetic resonance imagers on external and implantable pulse generators. Pacing Clin Electrophysiol 1984; 7: pp. 720-727.
13. Gimbel J.R., Johnson D., Levine P.A., et. al.: Safe performance of magnetic resonance imaging on five patients with permanent cardiac pacemakers. Pacing Clin Electrophysiol 1996; 19: pp. 913-919.
14. Fontaine J.M., Mohamed F.B., Gottlieb C., et. al.: Rapid ventricular pacing in a pacemaker patient undergoing magnetic resonance imaging. Pacing Clin Electrophysiol 1998; 21: pp. 1336-1339.
15. Irnich W., Irnich B., Bartsch C., et. al.: Do we need pacemakers resistant to magnetic resonance imaging?. Europace 2005; 7: pp. 353-365.
16. Sommer T., Vahlhaus C., Lauck G., et. al.: MRI and cardiac pacemakers: in-vitro evaluation and in vivo studies in 51 patients at 0.5 T. Radiology 2000; 215: pp. 869-879.
17. Gold M.R., Kanal E., Schwitter J., et. al.: Preclinical evaluation of implantable cardioverter-defibrillator developed for magnetic resonance imaging use. Heart Rhythm 2015; 12: pp. 631-638.
18. van der Graaf A.W., Bhagirath P., Gotte M.J.: MRI and cardiac implantable electronic devices; current status and required safety conditions. Neth Heart J 2014; 22: pp. 269-276.
19. Faris O.P., Shein M.J.: Government viewpoint: US Food and Drug Administration: pacemakers, ICDs, and MRI. Pacing Clin Electrophysiol 2005; 28: pp. 268-269.
20. Levine G.N., Gomes A.S., Arai A.E., et. al.: Safety of magnetic resonance imaging in patients with cardiovascular devices: an American Heart Association Scientific Statement from the Committee on Diagnostic and Interventional Cardiac Catheterization, Council on Clinical Cardiology, and the Council on Cardiovascular Radiology and Intervention: endorsed by the American College of Cardiology Foundation, the North American Society for Cardiac Imaging, and the Society for Cardiovascular Magnetic Resonance. Circulation 2007; 116: pp. 2878-2891.
21. Brignole M., Auricchio A., Baron-Esquivias G., et. al.: 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy: the Task Force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Eur Heart J 2013; 34: pp. 2281-2329.
22. Verma A., Ha A.C., Dennie C.: Canadian Heart Rhythm Society and Canadian Association of Radiologists consensus statement on magnetic resonance imaging with cardiac implantable electronic devices. Can Assoc Radiol J 2014; 65: pp. 290-300.
23. Mollerus M., Albin G., Lipinski M., et. al.: Magnetic resonance imaging of pacemakers and implantable cardioverter-defibrillators without specific rate restrictions. Europace 2010; 12: pp. 947-951.
24. Baker K.B., Tkach J.A., Nyenhuis J.A., et. al.: Evaluation of specific absorption rate as a dosimeter of MRI-related implant heating. J Magn Reson Imaging 2004; 20: pp. 315-320.
25. Rashid S., Rapacchi S., Shivkumar K., et. al.: Modified wideband three-dimensional late gadolinium enhancement MRI for patients with implantable cardiac devices. Magn Reson Med 2016; 75: pp. 572-584. 2015 March 13 [Epub]
26. Rashid S., Rapacchi S., Vaseghi M., et. al.: Improved late gadolinium enhancement MR imaging for patients with implanted cardiac devices. Radiology 2014; 270: pp. 269-274.
27. Stevens S.M., Tung R., Rashid S., et. al.: Device artifact reduction for magnetic resonance imaging of patients with implantable cardioverter-defibrillators and ventricular tachycardia: late gadolinium enhancement correlation with electroanatomic mapping. Heart Rhythm 2014; 11: pp. 289-298.
28. Ranjan R., McGann C.J., Jeong E.K., et. al.: Wideband late gadolinium enhanced magnetic resonance imaging for imaging myocardial scar without image artefacts induced by implantable cardioverter-defibrillator: a feasibility study at 3 T. Europace 2015; 17: pp. 483-488.