Rationale and Objectives
Improvements in imaging technology have led to the increased use of computed tomography (CT). For example, micro-CT and quantitative CT (QCT) are now often used in osteoporosis research, in which micro-CT is able to analyze small bones or bone samples with high spatial resolution. In contrast, QCT is able to investigate large samples with low spatial resolution. The aim of this study was to test the usefulness of flat-panel volumetric CT (fpVCT) in a rat model of osteopenia.
Material and Methods
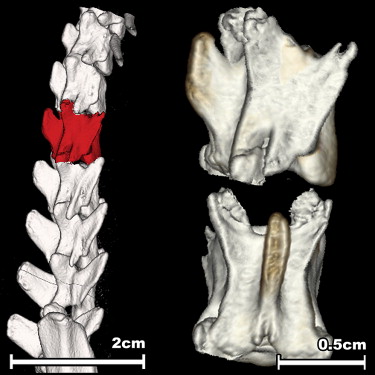
Twenty-two 3-month-old rats underwent ovariectomy and were either left untreated or supplemented with estradiol for 15 weeks. After sacrificing, the rats’ second lumbar vertebral body bone mineral density (BMD) was analyzed using fpVCT and ashing. The results were compared to those of a microstructural analysis of the first lumbar vertebrae and a biomechanical evaluation of the fourth lumbar vertebrae.
Results
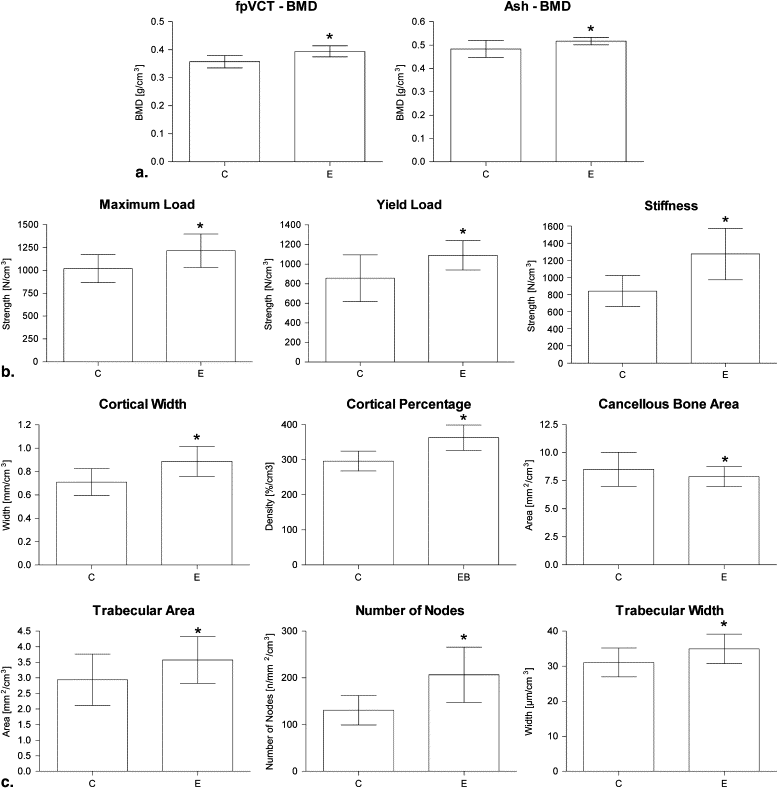
BMD measurements using both fpVCT (0.39 vs 0.35 mg/cm 3 ) and ashing (0.52 vs 0.48 mg/cm 3 ) demonstrated a significant improvement after estradiol supplementation. The correlation coefficient of the two methods was 0.858. After estradiol supplementation, the bone microstructural and bone biomechanical parameters were improved, compared to no treatment. The correlations of both the microstructural and the biomechanical evaluations were closer for BMD measured using fpVCT (r = 0.482–0.769) than on the basis of ashing (r = 0.345–0.573). FpVCT was not able to display the trabecular microstructure of the rat lumbar vertebrae.
Conclusion
The use of fpVCT demonstrated a close relationship between morphologic and biomechanical evaluations in a rat model of osteopenia. Because of its different proportions, fpVCT might be able to bridge the gap between micro-CT and QCT in analyzing larger animals.
The introduction of multidetector-row computed tomography (CT) has opened avenues for the examination of large body sections within short examination times, while maintaining high spatial resolution . A novel approach in hardware development, however, uses flat-panel detectors. Flat-panel detectors allow for greater spatial resolution, isotropic voxel imaging, and volumetric coverage than conventional CT . The flat-panel detectors used were initially developed for conventional radiographic applications and are known for offering excellent image quality in high-contrast structures . The main difference between flat-panel volumetric CT (fpVCT) and conventional multidetector-row CT is that the rows of detector elements (usually four, 16, or 64) are replaced by an area detector . Currently, only a few prototypes for fpVCT exist, and data on the diagnostic benefit of these devices are scarce. Therefore, the aim of this study was to evaluate the possibilities of fpVCT for use in osteoporosis research.
Osteoporosis in postmenopausal women mainly affects the trabecular bones of the body (eg, vertebral body, femoral neck, distal radius, or proximal humerus). Because of their high clinical relevance, lumbar vertebral bodies were chosen for this study. Vertebral fractures are an important clinical indicator of the progression of osteoporosis and the ongoing fracture risk for new osteoporotic fractures, independent of bone mineral density (BMD) ( ).
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Materials and methods
Animals and Substances
Get Radiology Tree app to read full this article<
fpVCT
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Ashing
Get Radiology Tree app to read full this article<
Biomechanical Testing
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Preparation for Microscopy and Microradiography
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Body Weight
Get Radiology Tree app to read full this article<
Table 1
Results of the Animal Trial
Variable Controls Estradiol-treated Rats Body weight After 15 weeks (g) 317.9 ± 26.1 246.5 ± 24.7 ∗ Vertebral body Volume (cm 3 ) 0.192 ± 0.014 0.165 ± 0.012 ∗ BMD (g/cm 3 ) fpVCT 0.357 ± 0.022 0.394 ± 0.020 ∗ Ashing 0.484 ± 0.034 0.517 ± 0.016 ∗ Biomechanical properties Maximum load (N/cm 3 ) 1020 ± 154 1217 ± 184 ∗ Yield load (N/cm 3 ) 856 ± 239 1090 ± 150 ∗ Young’s modulus (N/mm/cm 3 ) 841 ± 180 1274 ± 301 ∗ Microradiography Cortical percentage (%) 56.1 ± 3.7 59.7± 5.4 ∗ Cortical width (mm) 0.135 ± 0.019 0.146 ± 0.021 ∗ Cancellous area (mm 2 ) 1.56 ± 0.40 1.33 ± 0.18 ∗ Trabecular area (mm 2 ) 0.53 ± 0.12 0.62 ± 0.13 ∗ Number of nodes ( n /mm 2 ) 24.8 ± 6.1 34.1 ± 10.2 ∗ Trabecular width (μm) 5.88 ± 0.64 5.74 ± 0.65
BMD, bone mineral density; fpVCT, flat-panel volumetric computed tomography.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
fpVCT
Get Radiology Tree app to read full this article<
Ashing
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Biomechanical Testing
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Microradiographic Evaluation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Pearson’s r and R 2 Values of the Different Correlations Between the BMD Measurements and the Biomechanical and Structural Evaluations
fpVCT Ashing Variable_r__R_ 2 r__R 2 Biomechanical properties Maximum load (N/cm 3 ) 0.769 0.591 0.573 0.328 Yield load (N/cm 3 ) 0.523 0.274 0.473 0.227 Young’s modulus (N/mm/cm 3 ) 0.482 0.232 0.516 0.266 Microradiography Cortical percentage (%) 0.515 0.265 0.426 0.181 Cancellous area (mm 2 ) 0.353 0.125 0.272 0.074 Trabecular area (mm 2 ) 0.445 0.198 0.345 0.119 Number of nodes ( n /mm 2 ) 0.511 0.261 0.419 0.175 Trabecular width (μm) 0.486 0.236 0.436 0.190
BMD, bone mineral density; fpVCT, flat-panel volumetric computed tomography.
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusion
Get Radiology Tree app to read full this article<
Acknowledgments
Get Radiology Tree app to read full this article<
References
1. Flohr T., Stierstorfer K., Bruder H., et. al.: New technical developments in multislice CT—part 1: approaching isotropic resolution with sub-millimeter 16-slice scanning. Rofo 2002; 174: pp. 839-845.
2. Völk M., Hamer O.W., Feuerbach S., et. al.: Dose reduction in skeletal and chest radiography using a large-area flat-panel detector based on amorphous silicon and thallium-doped cesium iodide: technical background, basic image quality parameters, and review of the literature. Eur Radiol 2004; 14: pp. 827-834.
3. Groh B.A., Siewerdsen J.H., Drake D.G., et. al.: A performance comparison of flat-panel imager-based MV and kV cone-beam CT. Med Phys 2002; 29: pp. 967-975.
4. Ludwig K., Henschel A., Bernhardt T.M., et. al.: Performance of a flat-panel detector in the detection of artificial erosive changes: comparison with conventional screen-film and storage-phosphor radiography. Eur Radiol 2003; 13: pp. 1316-1323.
5. Nikolaou K., Flohr T., Stierstorfer K., et. al.: Flat panel computed tomography of human ex vivo heart and bone specimens: initial experience. Eur Radiol 2005; 15: pp. 329-333.
6. Siris E., Adachi J.D., Lu Y., et. al.: Effects of raloxifene on fracture severity in postmenopausal women with osteoporosis: results from the MORE study. Osteoporos Int 2002; 13: pp. 907-913.
7. Lindsay R., Gallagher J.C., Kleerekoper M., et. al.: Bone response to treatment with lower doses of conjugated estrogens with and without medroxyprogesterone acetate in early postmenopausal women. Osteoporos Int 2005; 16: pp. 372-379.
8. Genant H.K., Delmas P.D., Chen P., et. al.: Severity of vertebral fracture reflects deterioration of bone microarchitecture. Osteoporos Int 2007; 18: pp. 69-76.
9. Erlandsson M.C., Jonsson C.A., Lindberg M.K., et. al.: Raloxifene- and estradiol-mediated effects on uterus, bone and B lymphocytes in mice. J Endocrinol 2002; 175: pp. 319-327.
10. Rossouw J.E., Anderson G.L., Prentice R.L., et. al.: Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 2002; 288: pp. 321-333.
11. Stürmer E.K., Seidlová-Wuttke D., Sehmisch S., et. al.: Standardized bending and breaking test for the normal and osteoporotic metaphyseal tibias of the rat: effect of estradiol, testosterone, and raloxifene. J Bone Miner Res 2006; 21: pp. 89-96.
12. Stürmer K.M., Schmit-Neuerburg K.P.: Quantitative determination of the effect of induced alternating currents on the integration of autologous spongiosa transplants. Unfallchirurgie 1985; 11: pp. 168-173.
13. Bitterling H., Vogel T., Dobler T., et. al.: Role of osteoporosis in trauma diagnostics. Rofo 2005; 177: pp. 1663-1669.
14. Guglielmi G., Lang T.F.: Quantitative computed tomography. Semin Musculoskelet Radiol 2002; 6: pp. 219-227.
15. Prevrhal S., Genant H.K.: Quantitative computer tomography. Radiologe 1999; 39: pp. 194-202.
16. Raisz L.G.: Clinical practice. Screening for osteoporosis. N Engl J Med 2005; 353: pp. 164-171.
17. Engelke K., Karolczak M., Lutz A., et. al.: Micro-CT. Technology and application for assessing bone structure. Radiologe 1999; 39: pp. 203-212.
18. Felsenberg D., Boonen S.: The bone quality framework: determinants of bone strength and their interrelationships, and implications for osteoporosis management. Clin Ther 2005; 27: pp. 1-11.
19. Martin-Badosa E., Amblard D., Nuzzo S., et. al.: Excised bone structures in mice: imaging at three-dimensional synchrotron radiation micro CT. Radiology 2003; 229: pp. 921-928.
20. Obert M., Ahlemeyer B., Baumgart-Vogt E., et. al.: Flat-panel computed tomography: a new method for visualizing fine bone detail in living mice. J Comput Assist Tomogr 2005; 29: pp. 560-565.
21. Kuiper J.W., van Kuijk C., Grashuis J.L., et. al.: Accuracy and the influence of marrow fat on quantitative CT and dual-energy x-ray absorptiometry measurements of the femoral neck in vitro. Osteoporosis Int 1996; 6: pp. 25-30.
22. Glüer C.C., Genant H.K.: Impact of marrow fat in accuracy of quantitative CT. J Comput Assist Tomogr 1989; 13: pp. 1023-1035.
23. Gupta R., Bartling S.H., Basu S.K., et. al.: Experimental flat-panel high-spatial-resolution volume CT of the temporal bone. AJNR Am J Neuroradiol 2004; 25: pp. 1417-1424.
24. Taschereau R., Chow P.L., Chatziioannou A.F.: Monte Carlo simulations of dose from microCT imaging procedures in a realistic mouse phantom. Med Phys 2006; 33: pp. 216-224.
25. Missbach-Guentner J., Dullin C., Zientkowska M., et. al.: Flat-panel detector-based volume computed tomography: a novel 3D imaging technique to monitor osteolytic bone lesions in a mouse tumor metastasis model. Neoplasia 2007; 9: pp. 755-765.
26. Missbach-Guentner J., Dullin C., Kimmina S., et. al.: Morphological changes of mammary carcinomas in mice over time as monitored by flat-panel detector volume computed tomography. Neoplasia 2008; 10: pp. 663-673.
27. Greschus S., Kiessling F., Lichy M.P., et. al.: Potential applications of flat-panel volumetric CT in morphologic and functional small animal imaging. Neoplasia 2005; 7: pp. 730-740.
28. Kiessling F., Greschus S., Lichy M.P., et. al.: Volumetric computed tomography (VCT): a new technology for noninvasive, high-resolution monitoring of tumor angiogenesis. Nat Med 2005; 10: pp. 1133-1138.
29. Reichardt B., Sarwar A., Bartling S.H., et. al.: Musculoskeletal applications of flat-panel volume CT. Skeletal Radiol 2008; 37: pp. 1069-1076.