Rationale and Objectives
The vascular permeability of tumors can be changed by transarterial infusion heat, but the mechanisms remain unknown. The aim of this study was to analyze the underlying causes of changes in tumor vascular permeability after heated perfusion via two different modes.
Materials and Methods
Thirty rabbits with VX2 hepatic tumors were randomly divided into three groups of 10 rabbits each. The hepatic artery was selectively catheterized via a femoral approach, and unheated saline (control group) or heated saline (60°C) was then injected in either a continuous (transcatheter arterial continuous perfusion [TACP]) or a pulsed (transcatheter arterial pulsed perfusion [TAPP]) manner. Changes in vascular permeability in the tumors were assessed using the following markers and methods: (1) qualitative assessment by visual estimation on digital subtraction angiography performed after the heat infusion procedure on live animals and quantitative assessment by spectrophotometry using Evans blue dye extravasation on tumor and liver tissue after animals were sacrificed and (2) kinase domain receptor or vascular endothelial growth factor (VEGF), expressed in vascular endothelial cells, assessed by immunohistochemical staining, Western blot analysis, and reverse transcription polymerase chain reaction.
Results
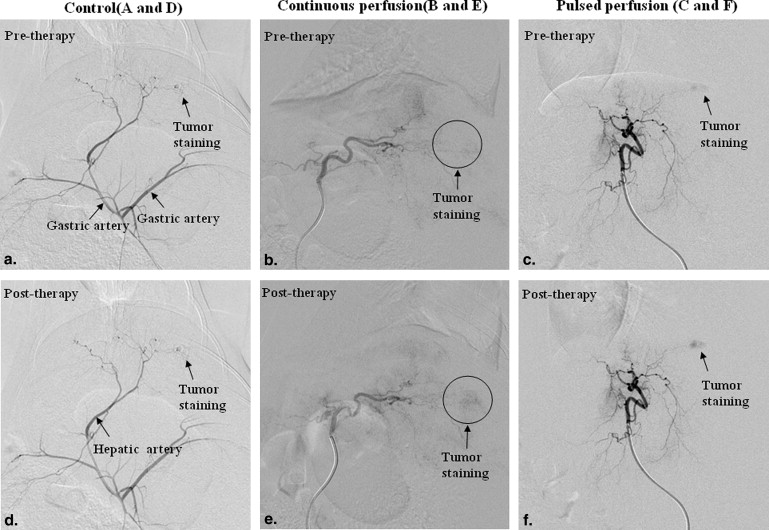
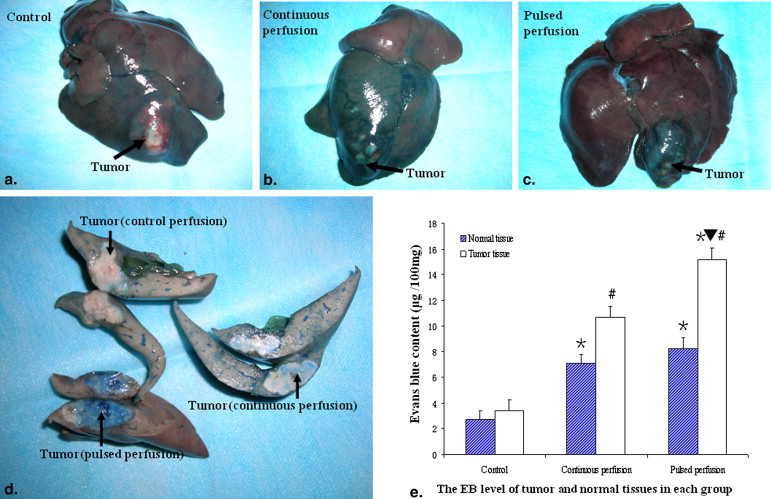
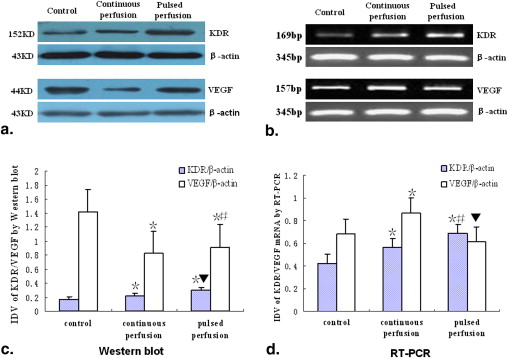
Tumor staining increased in the TAPP group more than in the TACP group, but not in the control group, assessed on digital subtraction angiography. Extracted dye was higher in tumors in the TAPP group than in those in the TACP group; extracted dye in both groups was higher than in the control group. Kinase domain receptor protein and messenger ribonucleic acid expression were both higher in the TAPP group than in the TACP and control groups. VEGF protein expression was lower in the TAPP and TACP groups than in the control group, but VEGF messenger ribonucleic acid expression was higher in the TACP group than in the TAPP and control groups, and VEGF messenger ribonucleic acid expression was lower in the TAPP group than in the control group.
Conclusions
The vascular permeability of rabbit VX2 tumors significantly increased after arterial pulsed heated infusion, and the protein kinase domain receptor may play a key role in this increase of tumor vascular permeability.
Therapies using hyperthermia have gained acceptance for the treatment of hepatocellular carcinoma, such as radiofrequency ablation , microwave coagulation therapy , laser-induced interstitial thermotherapy , and percutaneous hot saline injection therapy . These local thermotherapy methods have proven effective in patients with single, nodular-type hepatocellular carcinomas <5 cm, and they are designed to produce heat-induced coagulation necrosis . However, during treatment for patients with hepatocellular carcinomas >5 cm or with multifocal tumors, to reach all areas of the tumor and to induce complete necrosis, percutaneous puncture must be performed repeatedly, and the insertion site must be changed , so the rate of complications, such as arterioportal shunts and bile duct injury, may be up to 12% , and a needle-track tumor-seeding rate of 12.5% has been reported in patients after percutaneous radiofrequency ablation . In contrast, if the heat is distributed throughout the tumor by transhepatic arterial infusion, the insertion site need not be changed, and the complications of these local thermotherapy methods may be avoided. Thus, transarterial infusion with heated drugs may be a safe and promising new strategy that is able to achieve a better therapeutic effect in the treatment of both large and multifocal hepatic tumors.
Recently, regional hyperthermic hepatic perfusion has been used in the clinic in combination with chemotherapy to enhance the efficacy of cancer treatment . In vitro studies have demonstrated that the best anticancer effect is achieved at 42°C to 45°C for 30 minutes , and we have verified that the temperature of heated saline (60°C) that reaches the region of the tumor is not <42°C after injection and that perfusion of 60°C saline is safe and minimally damaging to the normal liver tissue of tumor-bearing rabbits . In vivo studies have also demonstrated that if heated drugs are infused directly into the target vessel of a tumor, the heat of the infusion can change the tumor’s vascular permeability , but the mechanism or mechanisms remain unknown. Because the vascular endothelial growth factor (VEGF) and kinase domain receptor (KDR) proteins are two important factors related to vascular permeability , they were observed and evaluated in this study. The aim of this study was to analyze the cause of the change in tumor vascular permeability after different modes of heated perfusion.
Materials and methods
Animal Models
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Experimental Grouping
Get Radiology Tree app to read full this article<
Arterial Catheterization
Temperature Probe Placement
Get Radiology Tree app to read full this article<
Hepatic Artery Catheterization
Get Radiology Tree app to read full this article<
Digital Subtraction Angiography
Get Radiology Tree app to read full this article<
Artery Perfusion
Get Radiology Tree app to read full this article<
Evans Blue (EB) Dye Injection
Get Radiology Tree app to read full this article<
Catheter Removal
Get Radiology Tree app to read full this article<
Animal Sacrifice
Get Radiology Tree app to read full this article<
Extravasation of EB Dye
Get Radiology Tree app to read full this article<
Hematoxylin and Eosin (H&E) and Immunohistochemistry
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Get Radiology Tree app to read full this article<
Western Blot Analysis
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Heating Efficiency
Get Radiology Tree app to read full this article<
Changes in Tumor Vascular Permeability
Digital Subtraction Angiographic Findings
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
EB Dye Extravasation
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
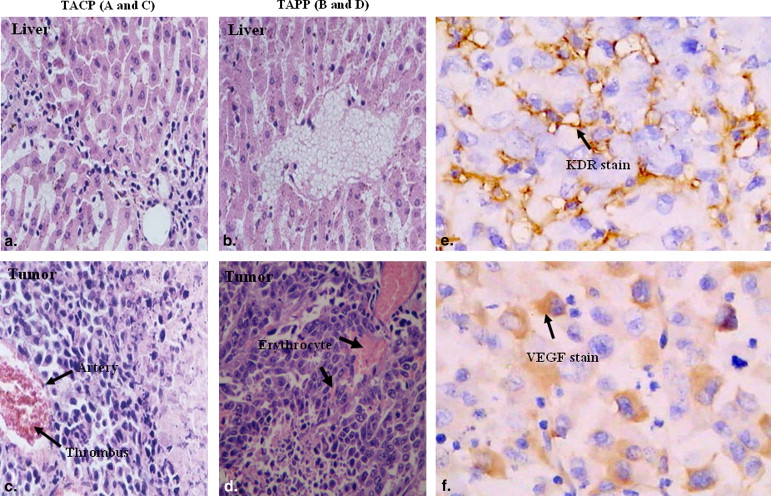
H&E Staining
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
KDR Protein and Messenger Ribonucleic Acid (mRNA) in Tumor Tissue
Immunohistochemical Staining
Get Radiology Tree app to read full this article<
Table 1
Comparisons of KDR and VEGF Protein Levels between Groups After Perfusion
Group KDR Protein Level ∗ VEGF Protein Level † ± + ++ 0 1+ 2+ 3+ Control (37°C) ( n = 10) 4 6 0 0 4 3 3 TACP (60°C) ( n = 10) 0 7 3 1 7 2 0 TAPP (60°C) ( n = 10) 0 2 8 0 8 2 0
KDR, kinase domain receptor; TACP, transcatheter arterial continuous perfusion; TAPP, transcatheter arterial pulsed perfusion; VEGF, vascular endothelial growth factor.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Western Blot Analysis
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
RT-PCR
Get Radiology Tree app to read full this article<
VEGF Protein and mRNA in Tumor Tissue
Immunohistochemical Staining
Get Radiology Tree app to read full this article<
Western Blot Analysis
Get Radiology Tree app to read full this article<
RT-PCR
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Conclusions
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Lee J.M., Lee Y.H., Kim Y.K., et. al.: Combined treatment of radiofrequency ablation and acetic acid injection: an in vivo feasibility study in rabbit liver. Eur Radiol 2004; 14: pp. 1303-1310.
2. Denys A.L., De Baere T., Kuoch V., et. al.: Radio-frequency tissue ablation of the liver: in vivo and ex vivo experiments with four different systems. Eur Radiol 2003; 13: pp. 2346-2352.
3. Buscarini E., Buscarini L.: Radiofrequency thermal ablation with expandable needle of focal liver malignancies: complication report. Eur Radiol 2004; 14: pp. 31-37.
4. Inokuchi R., Seki T., Ikeda K., et. al.: Percutaneous microwave coagulation therapy for hepatocellular carcinoma: increased coagulation diameter using a new electrode and microwave generator. Oncol Rep 2010; 24: pp. 621-627.
5. Nikfarjam M., Christophi C.: Interstitial laser thermotherapy for liver tumours. Br J Surg 2003; 90: pp. 1033-1047.
6. Araki Y., Hukano M., Urabe M., et. al.: Hepatocellular carcinoma treated by percutaneous hot saline injection. Oncol Rep 2004; 12: pp. 569-571.
7. Ng K.K., Lam C.M., Poon R.T., et. al.: Thermal ablative therapy for malignant liver tumors: a critical appraisal. J Gastroenterol Hepatol 2003; 18: pp. 616-629.
8. Guan Y.S., Liu Y.: Interventional treatments for hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int 2006; 5: pp. 495-500.
9. Llovet J.M., Vilana R., Bru C., et. al.: Increased risk of tumor seeding after percutaneous radiofrequency ablation for single hepatocellular carcinoma. Hepatology 2001; 33: pp. 1124-1129.
10. Feldman E.D., Wu P.C., Beresneva T., et. al.: Treatment of patients with unresectable primary hepatic malignancies using hyperthermic isolated hepatic perfusion. J Gastrointest Surg 2004; 8: pp. 200-207.
11. Harada M., Yamashita Y., Hirai T., et. al.: Intravascular hyperthermia: experimental study of transcatheter treatment. Acad Radiol 1995; 2: pp. 475-483.
12. Cao W., Wan Y., Liang Z.H., et. al.: Heated lipiodol as an embolization agent for transhepatic arterial embolization in VX2 rabbit liver cancer model. Eur J Radiol 2010; 73: pp. 412-419.
13. Cao W, Li JH, Feng DY, et al. Effect of transarterial pulsed perfusion with heated saline on tumor vascular permeability in a rabbit VX2 liver tumor model. Eur J Radiol. In press.
14. Baban D.F., Seymour L.W.: Control of tumour vascular permeability. Adv Drug Deliv Rev 1998; 34: pp. 109-119.
15. Feng D., Nagy J.A., Brekken R.A., et. al.: Ultrastructural localization of the vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) receptor-2 (FLK-1, KDR) in normal mouse kidney and in the hyperpermeable vessels induced by VPF/VEGF-expressing tumors and adenoviral vectors. J Histochem Cytochem 2000; 48: pp. 545-556.
16. Liu L.B., Xue Y.X., Liu Y.H.: Bradykinin increases the permeability of the blood-tumor barrier by the caveolae-mediated transcellular pathway. J Neurooncol 2010; 99: pp. 187-194.
17. Xia C.Y., Zhang Z., Xue Y.X., et. al.: Mechanisms of the increase in the permeability of the blood-tumor barrier obtained by combining low-frequency ultrasound irradiation with small-dose bradykinin. J Neurooncol 2009; 94: pp. 41-50.
18. Pietro L., Daher S., Rudge M.V., et. al.: Vascular endothelial growth factor (VEGF) and VEGF-receptor expression in placenta of hyperglycemic pregnant women. Placenta 2010; 31: pp. 770-780.
19. Liang B., Zheng C.S., Feng G.S., et. al.: Correlation of hypoxia-inducible factor 1alpha with angiogenesis in liver tumors after transcatheter arterial embolization in an animal model. Cardiovasc Intervent Radiol 2010; 33: pp. 806-812.
20. Gaber M.H., Wu N.Z., Hong K., et. al.: Thermosensitive liposomes: extravasation and release of contents in tumor microvascular networks. Int J Radiat Oncol Biol Phys 1996; 36: pp. 1177-1187.
21. Huang S.K., Stauffer P.R., Hong K., et. al.: Liposomes and hyperthermia in mice: increased tumor uptake and therapeutic efficacy of doxorubicin in sterically stabilized liposomes. Cancer Res 1994; 54: pp. 2186-2191.
22. Nesher E., Greenberg R., Avital S., et. al.: Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy in peritoneal carcinomatosis. Isr Med Assoc J 2007; 9: pp. 787-790.
23. Muraoka A., Takeda S., Matsui M., et. al.: Experimental study of a novel thermotherapy for hepatocellular carcinoma using a magnesium ferrite complex powder that produces heat under a magnetic field. Hepatogastroenterology 2004; 51: pp. 1662-1666.
24. Schuh A.C., Faloon P., Hu Q.L., et. al.: In vitro hematopoietic and endothelial potential of flk-1(-/-) embryonic stem cells and embryos. Proc Natl Acad Sci U S A 1999; 96: pp. 2159-2164.