Rationale and Objectives
Osteoarthritis (OA) is a common disease, and early diagnosis is essential for preventing further cartilage destruction and decreasing severe complications. Ultrasound biomicroscopy (UBM) is sensitive for detecting minute lesions in tissue because of its higher resolution, but its B-mode characterization of the early stage of OA has not been widely studied. The aim of this study was to determine the usefulness of UBM for detecting the early stage of OA using a rabbit model of early OA.
Materials and Methods
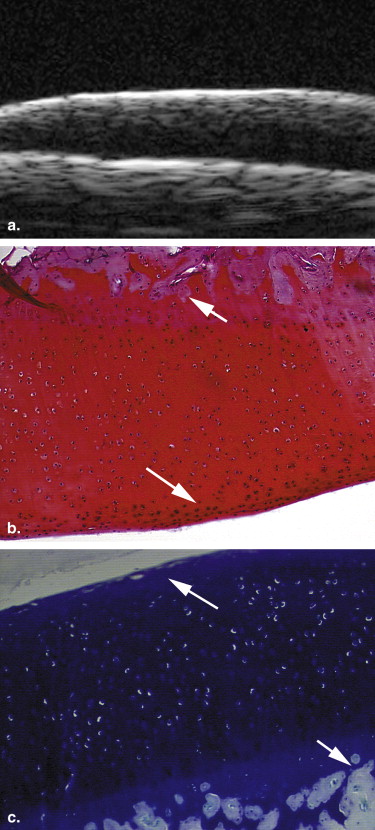
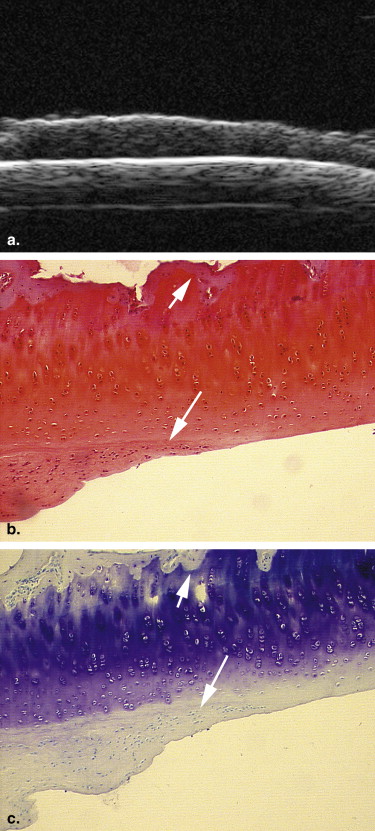
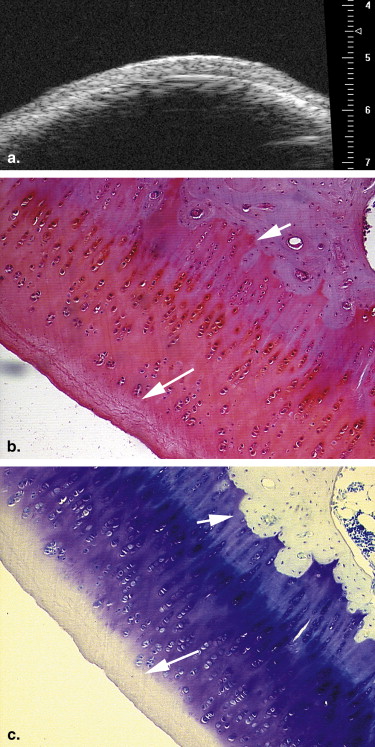
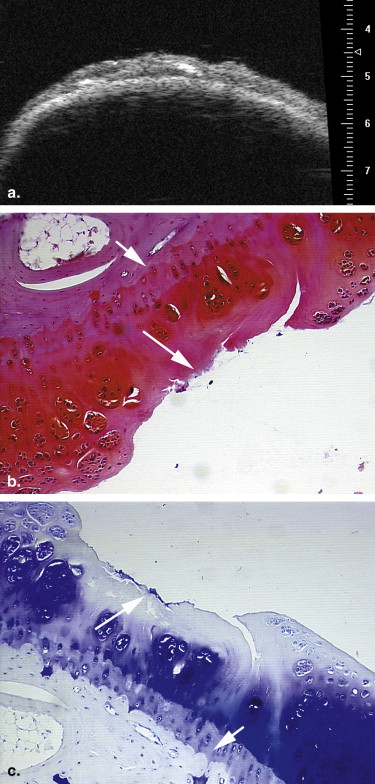
Eighteen adult New Zealand White female rabbits were used in this study, which included 12 rabbits that underwent transections of the left anterior cruciate ligament and six control rabbits. At 2, 4, and 6 weeks after surgery, four experimental rabbits and two control rabbits were euthanized. UBM was performed to evaluate the articular cartilage surfaces of the left knee, using a 55-MHz transducer. All the articular cartilage surfaces were independently assessed in blinded fashion by two radiologists for the severity of OA. The value of UBM, interobserver reliability, and the concordance between UBM and pathologic grades were determined.
Results
For the first radiologist, the sensitivity, specificity, positive predictive value, and negative predictive value of UBM for the diagnosis of OA were 91%, 83%, 89%, and 86%, respectively. For the second radiologist, the sensitivity, specificity, positive predictive value, and negative predictive value of UBM were 93%, 86%, 91%, and 89%, respectively. The concordance between UBM and pathologic grades for both radiologists was high (κ = 0.72 and 0.76), and the interobserver agreement was high (κ = 0.80).
Conclusions
UBM can be used to evaluate cartilage defects in an animal model, and further study is needed to determine whether this technique can be valuable for detecting early OA in humans.
Osteoarthritis (OA) is a common disease that can cause a loss of function and disability in adults . Early diagnosis is essential for preventing further cartilage destruction and decreasing the severe complications of this disease. Unfortunately, early changes in cartilage cannot be determined with radiography. Arthroscopy offers information only on the surface of articular cartilage, while possible degeneration of the deep cartilage and subchondral bone remains undetected. Ultrasound has been widely adopted to assess the articular cartilage, because of its noninvasiveness, easy access, and low cost . However, because the articular cartilage is a very thin tissue, it brings an inherent constraint between imaging spatial resolution and the penetration depth of conventional ultrasound, which is performed externally from the joint surface. Therefore, more and more researchers have become interested in a minimally invasive but more direct measurement of the articular cartilage using arthroscopy-based techniques . Quantitative high-resolution arthroscopic ultrasound imaging could provide a potential technique for the early diagnosis of OA. However, an uncontrolled, nonperpendicular angle of an ultrasound beam and the natural curvature of the cartilage surface may jeopardize the reliability of the ultrasound measurements .
Ultrasound biomicroscopy (UBM) has been shown to be sensitive for detecting minute lesions in tissue because of its higher resolution . But its B-mode characterization of the early stage of OA has not been widely studied . We hypothesized that B-mode UBM could be used for diagnosing early OA. The aim of this study was to determine the usefulness of UBM for detecting the early stage of OA using a standard surgical model of OA in rabbits.
Materials and methods
Animals and OA Model
Get Radiology Tree app to read full this article<
UBM
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Reference Standard
Get Radiology Tree app to read full this article<
Statistical Analysis
Get Radiology Tree app to read full this article<
Results
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 1
Detection of OA in Rabbits on Ultrasound Biomicroscopy
First Radiologist Second Radiologist Pathology OA Lesions Normal OA Lesions Normal OA lesions 39 4 40 3 Normal 5 24 4 25
Data are the number of articular cartilage surfaces.
OA, osteoarthritis.
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Table 2
Distribution of Ultrasound Biomicroscopy Grades and Pathologic Grades of Articular Cartilage Surfaces
First Radiologist Second Radiologist OARSI Grade Normal Grade 1 Grade 2 Grade 3 Normal Grade 1 Grade 2 Grade 3 Normal 24 5 0 0 25 4 0 0 Grade 1 4 7 3 0 3 9 2 0 Grade 2 0 2 21 0 0 3 20 0 Grade 3 0 0 0 6 0 0 0 6
Data are the number of articular cartilage surfaces.
OARSI, Osteoarthritis Research Society International.
Get Radiology Tree app to read full this article<
Discussion
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
Get Radiology Tree app to read full this article<
References
1. Arden N., Nevitt M.C.: Osteoarthritis: epidemiology. Best Pract Res Clin Rheumatol 2006; 20: pp. 3-25.
2. Aisen A.M., McCune W.J., MacGuire A., et. al.: Sonographic evaluation of the cartilage of the knee. Radiology 1984; 153: pp. 781-784.
3. Grassi W., Lamanna G., Farina A., et. al.: Sonographic imaging of normal and osteoarthritic cartilage. Semin Arthritis Rheum 1999; 28: pp. 398-403.
4. Friedman L., Finlay K., Jurriaans E.: Ultrasound of the knee. Skeletal Radiol 2001; 30: pp. 361-377.
5. Möller I., Bong D., Naredo E., et. al.: Ultrasound in the study and monitoring of osteoarthritis. Osteoarthritis Cartilage 2008; 16: pp. S4-S7.
6. Huang Y.P., Zheng Y.P.: Intravascular ultrasound (IVUS): a potential arthroscopic tool for quantitative assessment of articular cartilage. Open Biomed Eng J 2009; 26: pp. 13-20.
7. Virén T., Saarakkala S., Kaleva E., et. al.: Minimally invasive ultrasound method for intra-articular diagnostics of cartilage degeneration. Ultrasound Med Biol 2009; 35: pp. 1546-1554.
8. Laasanen M.S., Töyräs J., Hirvonen J., et. al.: Novel mechano-acoustic technique and instrument for diagnosis of cartilage degeneration. Physiol Meas 2002; 23: pp. 491-503.
9. Hattori K., Takakura Y., Ishimura M., et. al.: Quantitative arthroscopic ultrasound evaluation of living human cartilage. Clin Biomech (Bristol, Avon) 2004; 19: pp. 213-216.
10. Kaleva E., Saarakkala S., Jurvelin J.S., et. al.: Effects of ultrasound beam angle and surface roughness on the quantitative ultrasound parameters of articular cartilage. Ultrasound Med Biol 2009; 35: pp. 1344-1351.
11. Yao B.Q., Wu L.L., Zhang C., et. al.: Ultrasound biomicroscopic features associated with angle closure in fellow eyes of acute primary angle closure after laser iridotomy. Ophthalmology 2009; 116: pp. 444-448.
12. Weisbrod D.J., Pavlin C.J., Xu W., et. al.: Long-term follow-up of 42 patients with small ciliary body tumors with ultrasound biomicroscopy. Am J Ophthalmol 2010; 149: pp. 616-622.
13. Spriet M.P., Girard C.A., Foster S.F., et. al.: Validation of a 40 MHz B-scan ultrasound biomicroscope for the evaluation of osteoarthritis lesions in an animal model. Osteoarthritis Cartilage 2005; 13: pp. 171-179.
14. Altman R.D., Gold G.E.: Atlas of individual radiographic features in osteoarthritis, revised. Osteoarthritis Cartilage 2007; 15: pp. A1-A56.
15. Saïed A., Chérin E., Gaucher H., et. al.: Assessment of articular cartilage and subchondral bone: subtle and progressive changes in experimental osteoarthritis using 50 MHz echography in vitro. J Bone Miner Res 1997; 12: pp. 1378-1386.
16. Disler D.G., Raymond E., May D.A., et. al.: Articular cartilage defects: in vitro evaluation of accuracy and interobserver reliability for detection and grading with US. Radiology 2000; 215: pp. 846-851.
17. Pritzker K.P., Gay S., Jimenez S.A., et. al.: Osteoarthritis cartilage histopathology: grading and staging. Osteoarthritis Cartilage 2006; 14: pp. 13-29.
18. Minns R.J., Steven F.S., Hardinge K.: Osteoarthrotic articular cartilage lesions of the femoral head observed in the scanning electron microscope. J Pathol 1977; 122: pp. 63-70.
19. Arokoski J.P., Jurvelin J.S., Väätäinen U., et. al.: Normal and pathological adaptations of articular cartilage to joint loading. Scand J Med Sci Sports 2000; 10: pp. 186-198.
20. Buckwalter J.A., Mankin H.J.: Articular cartilage, part II: degeneration and osteoarthritis, repair, regeneration, and transplantation. J Bone Joint Surg Am 1997; 79: pp. 612-632.
21. Freeman M.A.: Is collagen fatigue failure a cause of osteoarthrosis and prosthetic component migration? A hypothesis. J Orthop Res 1999; 17: pp. 3-8.
22. Yoon C.H., Kim H.S., Ju J.H., et. al.: Validity of the sonographic longitudinal sagittal image for assessment of the cartilage thickness in the knee osteoarthritis. Clin Rheumatol 2008; 27: pp. 1507-1516.
23. Iagnocco A., Filippucci E., Ossandon A., et. al.: High resolution ultrasonography in detection of bone erosions in patients with hand osteoarthritis. J Rheumatol 2005; 32: pp. 2381-2383.
24. Saarakkala S., Julkunen P., Kiviranta P., et. al.: Depth-wise progression of osteoarthritis in human articular cartilage: investigation of composition, structure and biomechanics. Osteoarthritis Cartilage 2010; 18: pp. 73-81.
25. Saarakkala S., Toyras J., Hirvonen J., et. al.: Ultrasonic quantitation of superficial degradation of articular cartilage. Ultrasound Med Biol 2004; 30: pp. 783-792.
26. Toyras J., Rieppo J., Nieminen M.T., et. al.: Characterization of enzymatically induced degradation of articular cartilage using high frequency ultrasound. Phys Med Biol 1999; 44: pp. 2723-2733.
27. Julkunen P., Iivarinen J., Brama P.A., et. al.: Maturation of collagen fibril network structure in tibial and femoral cartilage of rabbits. Osteoarthritis Cartilage 2010; 18: pp. 406-415.
28. Rieppo J., Hyttinen M.M., Halmesmaki E., et. al.: Remodelation of collagen network architecture during cartilage maturation [abstract]. Trans Orthop Res Soc 2004; 29: pp. 549.
29. Hollander A.P., Heathfield T.F., Webber C., et. al.: Increased damage to type II collagen in osteoarthritic articular cartilage detected by a new immunoassay. J Clin Invest 1994; 93: pp. 1722-1732.